Abstract
Objective
The objectives of this study were to determine the efficacy of AZD6244, a potent, selective MEK inhibitor, in epithelial ovarian cancer (EOC) cells and to determine the enhanced cell death by combined treatment of paclitaxel and AZD6244.
Methods
EOC cells were treated with tenfold dilutions of AZD6244 (0.1 to 10 µM) for 24, 48 and 72 hours. Cell viability was determined by the CellTiter 96 AQueous One Solution Cell Proliferation Assay. The apoptotic cascade was assessed by Caspase-Glo assays. ERK activation was evaluated by Western blot analyses. Cytokine profiling was performed from culture supernatants using the Luminex 200 system.
Results
In vitro cell viability showed that ovarian cancer cells with high p-ERK activities (A2780, R454, 01-28) exhibited significant growth inhibition. Cells with low p-ERK activities (R182, CP70), however, were resistant to AZD6244. Caspase-3 was not activated during AZD6244-induced growth inhibition. AZD6244 significantly inhibited p-ERK1/2 in both cell types regardless of constitutive levels of p-ERK. Proinflammatory cytokines including IL-6, IL-8, MCP-1 and GM-CSF were significantly decreased. Paclitaxel activated the p-ERK levels in paclitaxel-resistant R182 cells with low basal p-ERK level. MEK inhibition by AZD6244 enhanced paclitaxel-induced apoptosis significantly in R182 cells.
Conclusion
These results demonstrate that AZD6244 has a potent growth inhibitory effect in ovarian cancer cells with high p-ERK activities. In addition, targeted inhibition of the extracellular signal-regulated kinase pathway with AZD6244 can enhance the anti-tumor efficacy of the cytotoxic paclitaxel.
Figures and Tables
Figure 1
AZD6244 effectively inhibited MAPK/ERK pathway signaling in epithelial ovarian cancer cells. (A) EOC cells were treated with increasing concentrations of AZD6244 (0.1 to 10 µM) for 24 hours. MEK activity was determined by western blot for p-ERK1/2. Total ERK and β-actin were used for quantity control of proteins. (B) EOC cells were treated with with AZD6244 (10 µM) for different time points (0, 0.5, 1, 3, 24 hrs).

Figure 2
(A) AZD6244 inhibited the proliferation of EOC cells. Cells were exposed to increasing concentrations of AZD6244 (0.1 to 10 µM) for 24, 48 and 72 hours and the viability (in percentage, normalized to untreated cells) was measured. (B) Absorbance at 490 nm after AZD6244 for 24, 48 and 72 hours. Data were compiled from at least three independent experiments, each done in triplicate.
*P<0.05.

Figure 3
Relative caspase-3/7 activity in A2780 and R182 cells. Cells were exposed to AZD6244 for 24 hours. Caspase activity was measured as described in Materials and Methods. Y axis: caspase activity fold increase from control. The data were compiled from at least three independent experiments.

Figure 4
Cell cycle analysis in EOC cells. Cells were treated with AZD6244 (10 µM) for 48 hours. The cells stained with propidium iodide (PI) solution were subjected to flow cytometric analysis to determine the cell distribution at each phase of the cell cycle. The EOC cells treated with AZD6244 were found to undergo G0/G1-phase cell cycle arrest.

Figure 5
AZD6244 induced down-regulation of cyclin D1 in EOC cells. Cells were lysed for Western blot analyses after AZD6244 (0.1 to 10 µM) for 48 hours.
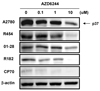
Figure 6
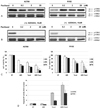
Expression of cytokines in MyD88-positive, low basal p-ERK R182 cells. Cells were treated with AZD6244 or paclitaxel for 48hrs, and the levels of cytokines /chemokines secreted were determined in the supernatants from EOC cells. Significant levels of pro-inflammatory cytokines were expressed in response to paclitaxel treatment. AZD6244 decreased pro-inflammatory cytokines in EOC cells. X-axis: AZD 6244(Taxol) dose.

Figure 7
Combination treatment of paclitaxel and AZD6244. (A) Effect of paclitaxel on the activation of p-ERK1/2 in epithelial ovarian cancer cells (A2780, R182). Cells were exposed to the indicated concentrations of paclitaxel for 24 hours. (B) Cells were treated with AZD6244 (10 µM) and increasing concentrations of paclitaxel (0.2 to 20 µM) for 24 hours and activation status of p-ERK1/2. (C) Cells were treated with AZD6244 (10 µM) and paclitaxel (20 µM) for 24, 48, and 72 hours and cell viability was determined (A2780, R182). (D) Cells were treated with AZD6244 (10 µM) and paclitaxel (20 µM) for 24 hours and activity of caspase-3 was measured. NT: non-treated. *P<0.05.
References
1. Aletti GD, Gallenberg MM, Cliby WA, Jatoi A, Hartmann LC. Current management strategies for ovarian cancer. Mayo Clin Proc. 2007. 82:751–770.

2. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008. 58:71–96.

3. Pouysségur J, Lenormand P. Fidelity and Spatio-temporal control in MAP kinase (ERKs) signaling. Eur J Biochem. 2003. 270:3291–3299.
4. Friday BB, Yu C, Dy GK, Smith PD, Wang L, Thibodeau SN, et al. BRAF V600E disrupts AZD6244-induced abrogation of negative feedback pathways between extracellular signal-regulated kinase and Raf proteins. Cancer Res. 2008. 68:6145–6153.

5. Montagut C, Settleman J. Targeting the RAFMEK-ERK pathway in cancer therapy. Cancer Lett. 2009. 283:125–134.

6. Beeram M, Patnaik A, Rowinsky EK. Raf: a strategic target for therapeutic development against cancer. J Clin Oncol. 2005. 23:6771–6790.

7. Allen LF, Sebolt-Leopold J, Meyer MB. CI-1040 (PD184352), a targeted signal transduction inhibitor of MEK (MAPKK). Semin Oncol. 2003. 30:5 suppl 16. 105–116.

8. Zeng P, Wagoner HA, Pescovitz OH, Steinmetz R. RNA interference (RNAi) for extracellular signal-regulated kinase 1 (ERK1) alone is sufficient to suppress cell viability in ovarian cancer cells. Cancer Biol Ther. 2005. 4:961–967.

9. Hsu CY, Bristow R, Cha MS, Wang BG, Ho CL, Kurman RJ, et al. Characterization of active mitogen-activated protein kinase in ovarian serous carcinomas. Clin Cancer Res. 2004. 10:6432–6436.

10. Kohno M, Pouyssegur J. Targeting the ERK signaling pathway in cancer therapy. Ann Med. 2006. 38:200–211.

11. Yoon S, Seger R. The extracellular signal-regulated kinase: multiple substrates regulate diverse cellular functions. Growth Factors. 2006. 24:21–44.

12. Nakayama N, Nakayama K, Yeasmin S, Ishibashi M, Katagiri A, Iida K, et al. KRAS or BRAF mutation status is a useful predictor of sensitivity to MEK inhibition in ovarian cancer. Br J Cancer. 2008. 99:2020–2028.

13. Sieben NL, Macropoulos P, Roemen GM, Kolkman-Uljee SM, Jan Fleuren G, Houmadi R, et al. In ovarian neoplasms, BRAF, but not KRAS, mutations are restricted to low-grade serous tumours. J Pathol. 2004. 202:336–340.
14. Cho YH, Kim DY, Kim JH, Kim YM, Kim KR, Nam JH, et al. Mutational analysis of KRAS, BRAF, and TP53 genes of ovarian serous carcinomas in Korean women. Yonsei Med J. 2009. 50:266–272.

15. Solit DB, Garraway LA, Pratilas CA, Sawai A, Getz G, Basso A, et al. BRAF mutation predicts sensitivity to MEK inhibition. Nature. 2006. 439:358–362.

16. Mayo MW, Norris JL, Baldwin AS. Ras regulation of NF-kappa B and apoptosis. Methods Enzymol. 2001. 333:73–87.
17. Tran SE, Holmstrom TH, Ahonen M, Kahari VM, Eriksson JE. MAPK/ERK overrides the apoptotic signaling from Fas, TNF, and TRAIL receptors. J Biol Chem. 2001. 276:16484–16490.

18. Kelly MG, Alvero AB, Chen R, Silasi DA, Abrahams VM, Chan S, et al. TLR-4 signaling promotes tumor growth and paclitaxel chemoresistance in ovarian cancer. Cancer Res. 2006. 66:3859–3868.

19. Dent P, Grant S. Pharmacologic interruption of the mitogen-activated extracellular-regulated kinase/mitogen-activated protein kinase signal transduction pathway: potential role in promoting cytotoxic drug action. Clin Cancer Res. 2001. 7:775–783.
20. Davies BR, Logie A, McKay JS, Martin P, Steele S, Jenkins R, et al. AZD6244 (ARRY-142886), a potent inhibitor of mitogen-activated protein kinase/extracellular signal-regulated kinase kinase 1/2 kinases: mechanism of action in vivo, pharmacokinetic/pharmacodynamic relationship, and potential for combination in preclinical models. Mol Cancer Ther. 2007. 6:2209–2219.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download