INTRODUCTION
Crossed renal ectopia is a rare congenital malformation in which one kidney is located on the side opposite from which its ureter inserts into the bladder. The majority of crossed ectopic kidneys are fused to the ipsilateral kidney. The incidence of congenital malformations involving these kidneys is increasing, and the ectopic kidney is more often involved [1]. The L-shaped kidney is one of the six forms of crossed renal ectopia with fusion; these include unilateral fused kidney (inferior ectopia), S-shaped kidney, lump kidney, L-shaped kidney, disc kidney, and unilateral fused kidney (superior ectopia) [2]. We describe a case of congenital pelvic ureteric junction obstruction (PUJO) involving an ectopically located crossed fused ectopic kidney and its management by laparoscopic pyeloplasty.
CASE REPORT
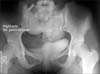
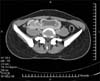
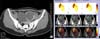
A 16-year-old female was admitted with complaints of right-side lower abdominal pain of a dull aching type lasting 6 months. Physical examination revealed no palpable lumps. On further evaluation, ultrasonography revealed bilateral kidneys absent in the renal fossa and found in the pelvis. These findings were confirmed with an intravenous pyelogram, which showed the right kidney low lying in the lumbar and pelvic region, malrotated, and normally excreting and the left kidney pelvic in location, grossly hydronephrotic, and crossing the midline (Fig. 1). A contrast-enhanced computed tomography (CT) scan showed a bilateral ectopic kidney with the L-type fusion, grossly dilated left renal pelvis, and the right ureter crossing over the left dilated pelvis (Fig. 2). Renal scintigraphy with LLEC (L-ethylcysteine) with single-photon emission CT (SPECT) (Fig. 3) showed left-sided crossed fused pelvic ectopic kidney with features suggestive of PUJO. The right kidney was also present in an ectopic location in the lumbar-pelvic region with an overall L-shape configuration. CT angiography revealed no crossing vessel as the cause of PUJO. Urine was sterile and the results of renal function tests were normal.
The patient was taken up for transperitoneal laparoscopic pyeloplasty under general anesthesia. The patient was initially placed in a lithotomy position for cystoscopy; the bilateral ureteric orifices were normally placed. Left-side retrograde pyelography was suggestive of PUJO, and a 6-Fr ureteric catheter was left in place. The right-side ureter was also stented by using a double J stent with a different color than the left ureteric catheter. The patient was placed supine with slight flexion of the table and the table in the 20-degree Trendelenburg position. Three ports were used: two 10 mm (periumbilical and right pararectal) and one 5 mm (left pararectal) in the lower abdomen.
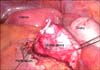
After creation of the pneumoperitoneum and with the ports in place, dissection was started and the dilated pelvis could be visualized through the peritoneal window. The peritoneum was incised and the dilated pelvis on the left side was reached. Through the left side ureteric catheter, dilute sterile methylene blue was instilled, which was aspirated percutaneously from the left renal pelvis under laparoscopic guidance to confirm the correct side. After further dissection, the classic Anderson-Hynes dismembered laparoscopic pyeloplasty was done by using 4 'O' polyglactin (Fig. 4). Minimal excision of the redundant pelvis was required and because the kidney was not mobilized, nephropexy was not required. Care was taken to avoid injury to the right ureter, which was coursing in close proximity to the left renal pelvis. Intraoperative antegrade stenting using a closed end 6-Fr/16-cm double J stent was done. Estimated blood loss during the procedure was around 50 mL, and the procedure was completed in 160 minutes without any intraoperative complications. The patient could tolerate oral feeding on the evening of the same day.
The periurethral catheter was removed on the second postoperative day, followed by drain removal and discharge on the third postoperative day. The double J stent was removed after 6 weeks and a renal nuclear scan was done after 3 and 12 months, which revealed decreased hydronephrosis and improved clearance (Fig. 5). At the 18-month follow-up, the patient was asymptomatic and doing well.
DISCUSSION
The incidence of PUJO in horseshoe and ectopic kidneys ranges from 25% to 33% and from 22% to 37%, respectively. No data are available in the literature regarding the incidence of PUJO in fusion anomalies other than horseshoe kidneys [1]. The embryological origin of crossed fused renal ectopia remains uncertain, but proposed causes include ureteric bud developmental abnormalities, undermining external pressures on the developing kidneys, and teratogenic factors [2]. In an L-shaped kidney, the fusion of the two kidneys is morphologically so severe that it results in the formation of a solitary kidney with two collecting systems. There have been reports of laparoscopic heminephrectomy for renal cell carcinoma in an L-shaped kidney [3] and laparoscopic pyelolithotomy in crossed fused ectopic kidney [4]. Romero et al. [5] reported accidental renal vein injury during laparoscopic heminephrectomy for renal cell carcinoma in a patient with a crossed fused kidney.
Laparoscopic pyeloplasty has rarely been performed in crossed fused kidneys. To the best of our knowledge, only one case of laparoscopic pyelolithotomy and pyeloplasty in a crossed fused ectopic kidney has been reported by Modi et al. [1]. However, no case has been reported of laparoscopic pyeloplasty in an L-shaped crossed fused ectopic kidney. In the hands of experienced surgeons, laparoscopic pyeloplasty offers a less invasive alternative to open surgery, with decreased morbidity, shorter hospital stay, and faster convalescence [6]. Moreover, we chose laparoscopic pyeloplasty over other minimally invasive treatment options such as endopyelotomy because laparoscopic pyeloplasty has been shown to offer better long-term success rates. Laparoscopic pyeloplasty in crossed fused ectopic kidneys poses a technical challenge to the surgeon performing a minimally invasive procedure. The specific problems are abnormal pelvic location of the kidneys (in close proximity to major vessels and pelvic viscera), abnormal vasculature supplying the kidneys, and proximity of the normal ureter to the dilated renal pelvis on the affected side.
We would like to emphasize the importance of a proper preoperative workup with a contrast-enhanced CT scan with CT angiography and a SPECT nuclear scan for detailed anatomical and functional study before proceeding to surgery. Preoperative retrograde pyelography and ureteral stenting on both sides should be considered to avoid iatrogenic injury to the contralateral ureter. Methylene blue instillation and aspiration in the ipsilateral pelvis may help in better identification of side. Attention to these details will help to avoid any possible mishaps. Laparoscopic pyeloplasty is a safe and feasible option in crossed fused renal ectopia that offers excellent patient outcomes with low morbidity. Judicious use of imaging, careful preoperative planning, and attention to details during surgery are necessary to avoid intraoperative surprises and mishaps.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download