Abstract
Purpose:
Phosphatase and tensin homolog (PTEN) is a novel tumor suppressor gene located at chromosome 10q23. Ki-67 antigen is a human nuclear protein that is expressed in all active parts of the cell cycle. We evaluated the significance of PTEN and Ki-67 expression in prostate cancer and investigated the relation of this expression with clinico- pathological factors in prostate cancer.
Materials and Methods:
Initially, we did two kinds of immunohistochemical staining for PTEN and Ki-67. Immunohistochemical staining was performed on 75 formalin-fixed paraffin-embedded cancer specimens. Staining on paraffin blocks from prostate carcinomas was compared with that for adjacent normal prostate. Stainings were considered positive if nuclear staining was seen. Positive stainings were analyzed with the patient's clinico-pathological findings. Statistical analysis was performed by using chi-square test with p<0.05 considered significant.
Results:
PTEN was expressed in 65 (86.6%) of 75 specimens. Ki-67 was expressed in 63 (84.0%) of 75 specimens. The staining scores of the tumor cells for PTEN and Ki-67 were higher than those of the adjacent normal cells (p<0.05). The staining scores for PTEN were negatively correlated with the serum prostate-specific antigen (PSA) level and Gleason score, but this was not statistically significant (p>0.05). PTEN expression was negatively correlated with lymph node or distant metastases (p<0.05). Ki-67 was positively correlated with the serum PSA level, the Gleason score, and metastases (p<0.05).
REFERENCES
1.Jemal A., Siegel R., Ward E., Murray T., Xu J., Smigal C, et al. Cancer statistics, 2006. CA Cancer J Clin. 2006. 56:106–30.

2.Cancer Registration and Biostatistics Branch, National Cancer Center. Cancer Statistics in Korea. 2006.
3.Javidan J., Deitch AD., Shi XB., de Vere White RW. The androgen receptor and mechanisms for androgen independence in prostate cancer. Cancer Invest. 2005. 23:520–8.

4.Steck PA., Pershouse MA., Jasser SA., Yung WK., Lin H., Ligon AH, et al. Identification of a candidate tumor suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet. 1997. 15:356–62.
5.Gray IC., Phillips SM., Lee SJ., Neoptolemos JP., Weissenbach J., Spurr NK. Loss of chromosomal region 10q23-25 in prostate cancer. Cancer Res. 1995. 55:4800–3.
6.Park JK., Jung MJ., Chun BK., Hur B. The relationship between PTEN tumor suppressor gene and vascular endothelial growth factor-mediated angiogenesis in breast cancer. Korean J Pathol. 2004. 38:100–5.
8.Liliental J., Moon SY., Lesche R., Mamillapalli R., Li D., Zheng Y, et al. Genetic deletion of the PTEN tumor suppressor gene promotes cell motility by activation of Rac1 and Cdc42 GTPases. Curr Biol. 2000. 10:401–4.

9.Koul D., Shen R., Garyali A., Ke LD., Liu TJ., Yung WK. MMAC/PTEN tumor suppressor gene regulates vascular endothelial growth factor-mediated angiogenesis in prostate cancer. Int J Oncol. 2002. 21:469–75.

10.Mazure NM., Chen EY., Laderoute KR., Giaccia AJ. Induction of vascular endothelial growth factor by hypoxia is modulated by a phosphatidylinositol 3-kinase/Akt signaling pathway in Ha-ras-transformed cells through a hypoxia inducible factor-1 transcriptional element. Blood. 1997. 90:3322–31.

11.Stattin P., Damber JE., Karlberg L., Bergh A. Cell proliferation assessed by Ki-67 immunoreactivity on formalin fixed tissues is a predictive factor for survival in prostate cancer. J Urol. 1997. 157:219–22.

12.Bubendorf L., Tapia C., Gasser TC., Casella R., Grunder B., Moch H, et al. Ki67 labeling index in core needle biopsies independently predicts tumor-specific survival in prostate cancer. Hum Pathol. 1998. 29:949–54.

13.Depowski PL., Rosenthal SI., Ross JS. Loss of expression of the PTEN gene protein product is associated with poor outcome in breast cancer. Mod Pathol. 2001. 14:672–6.

14.Rhei E., Kang L., Bogomolniy F., Federici MG., Borgen PI., Boyd J. Mutation analysis of the putative tumor suppressor gene PTEN/MMAC1 in primary breast carcinomas. Cancer Res. 1997. 57:3657–9.
15.Gibas P., Prout GR Jr., Connolly JG., Pontes JE., Sandberg AA. Nonrandom chromosomal changes in transitional cell carcinoma of the bladder. Cancer Res. 1984. 44:1257–64.
16.Cappellen D., Gil Diez de Medina S., Chopin D., Thiery JP., Radvanyi F. Frequent loss of heterozygosity on chromosome 10q in muscle-invasive transitional cell carcinomas of the blaader. Oncogene. 1997. 14:3059–66.
17.Cairns P., Evron E., Okami K., Halachmi N., Esteller M., Herman JG, et al. Point mutation and homozygous deletion of PTEN/MMAC1 in primary bladder cancers. Oncogene. 1998. 16:3215–8.

18.Shin DJ., Chung MJ., Kim HJ. Clinical significance of PTEN expression in bladder cancer. Korean J Urol. 2001. 42:594–7.
19.McMenamin ME., Soung P., Perera S., Kaplan I., Loda M., Sellers WR. Loss of PTEN expression in paraffin-embedded primary prostate cancer correlates with high Gleason score and advanced stage. Cancer Res. 1999. 59:4291–6.
20.Rubin MA., Gerstein A., Reid K., Bostwick DG., Cheng L., Parsons R, et al. 10q23.3 loss of heterozygosity is higher in lymph node-positive (pT2-3, N+) versus lymph node-negative (pT2-3, N0) prostate cancer. Hum Pathol. 2000. 31:504–8.
21.Fenic I., Franke F., Failing K., Steger K., Woenckhaus J. Expression of PTEN in malignant and non-malignant human prostate tissues: comparison with p27 protein expression. J Pathol. 2004. 203:559–66.

22.Gerdes J., Schwab U., Lemke H., Stein H. Production of a mouse monoclonal antibody reactive with a human nuclear antigen associated with cell proliferation. Int J Cancer. 1983. 31:13–20.

23.Burger PC., Shibata T., Kleihues P. The use of the monoclonal antibody Ki-67 in the identification of proliferating cells: application to surgical neuropathology. Am J Surg Pathol. 1986. 10:611–7.

24.Cohen MB., Waldman FM., Carroll PR., Kerschmann R., Chew K., Mayall BH. Comparison of five histopathologic methods to assess cellular proliferation in transitional cell carcinoma of the urinary bladder. Hum Pathol. 1993. 24:772–8.

25.Papadopoulos I., Weichert-Jacobsen K., Wacker HH., Sprenger E. Correlation between DNA ploidy, proliferation marker Ki-67 and early tumor progression in renal cell carcinoma. A prospective study. Eur Urol. 1997. 31:49–53.
26.Kim BH., Kim CI., Park CH. Caveolin-1 and Ki-67 expression as prognostic factors in clear cell carcinoma of the kidney. Korean J Urol. 2008. 49:99–106.

27.Delahunt B., Bethwaite PB., Thornton A., Ribas JL. Proliferation of renal cell carcinoma assessed by fixation-resistant poly-clonal Ki-67 antibody labeling. Correlation with clinical outcome. Cancer. 1995. 75:2714–9.

28.Chon WH., Lee SD., Lee JZ., Choi KW. The relationship of Clusterin expression and Ki-67 labeling index with clinicopathologic factors in human transitional cell carcinoma. Korean J Urol. 2008. 49:688–95.

29.Uzoaru I., Rubenstein M., Mirochnik Y., Slobodskoy L., Shaw M., Guinan P. An evaluation of the markers p53 and Ki-67 for their predictive value in prostate cancer. J Surg Oncol. 1998. 67:33–7.

30.Kim DG., Kim KK., Lee KS. Expressions of p53, bcl-2 protein and Ki-67 labelling index and their relationships with prognostic factors in prostate cancer. Korean J Urol. 2001. 42:828–33.
Fig. 1
(A, B) Absence of immunoreactivity for phosphatase and tensin homolog (PTEN) in normal prostate gland, (C, D) Numerous strongly positive nucleoli staining among cancer cells for PTEN (A, C: H&E x40, B, D: H&E x400).
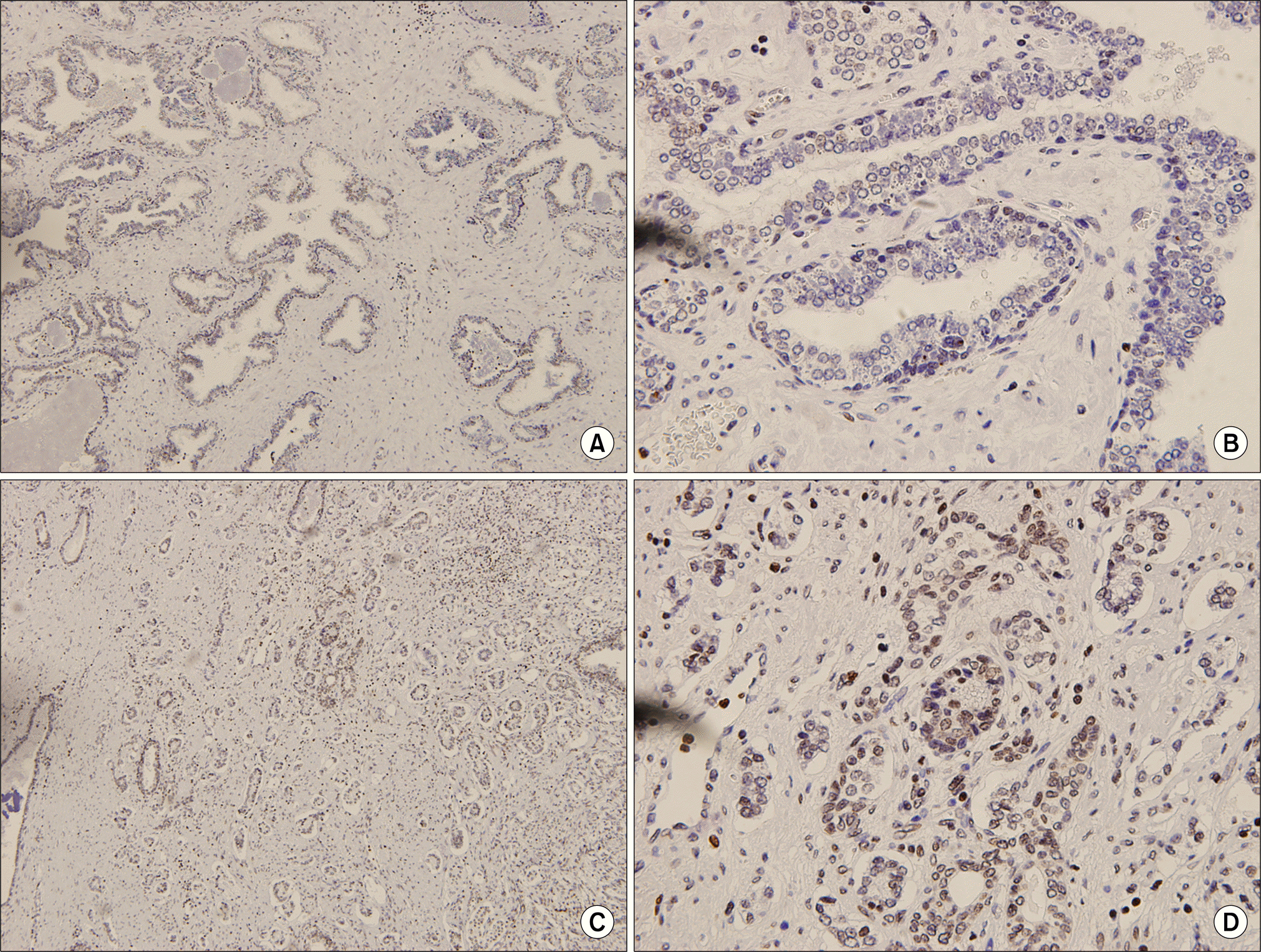
Fig. 2
(A, B) Absence of immunoreactivity for Ki-67 in normal prostate gland, (C, D) Discrete nuclear staining among cancer cells for Ki-67 (A, C: H&E x40, B, D: H&E x400).
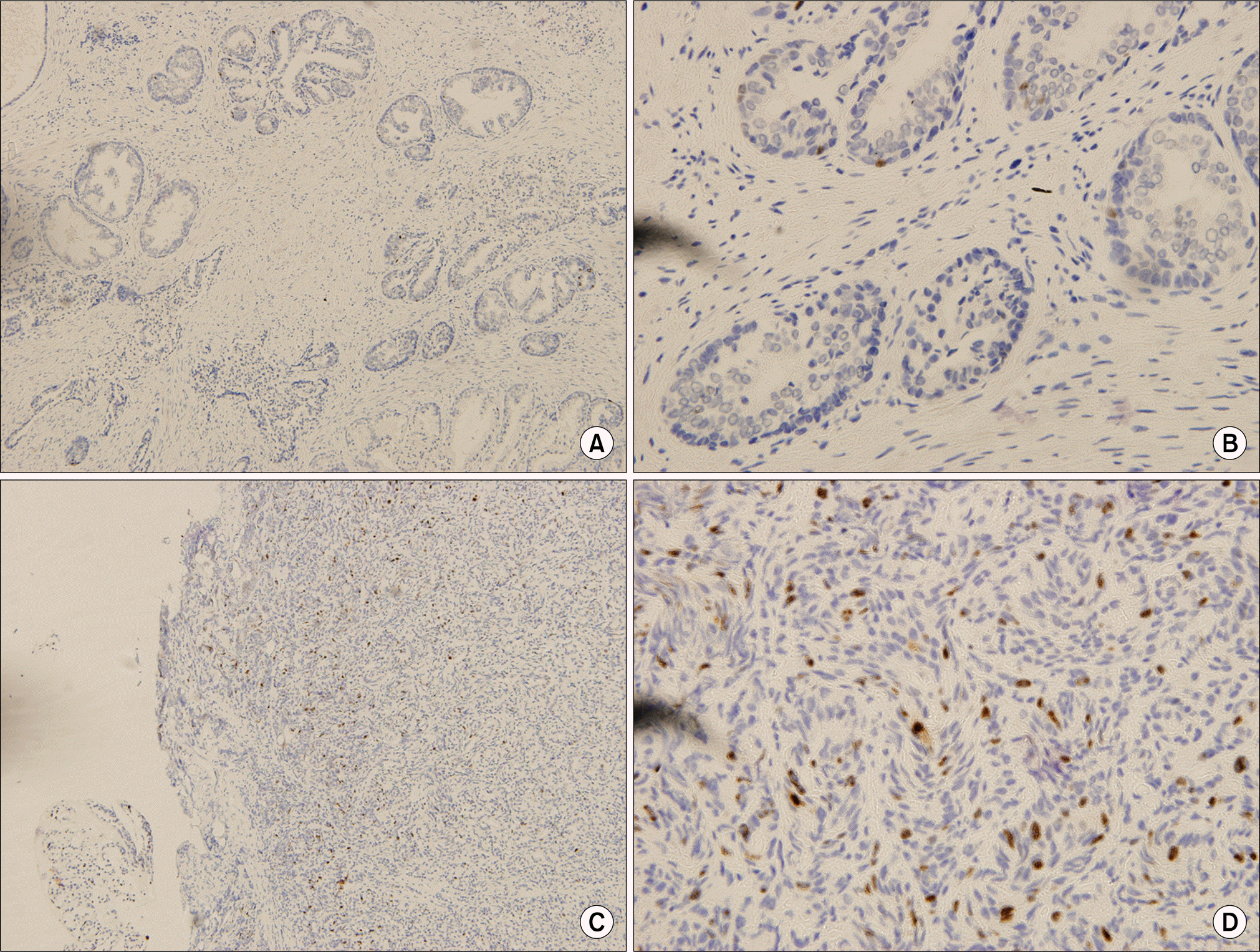
Fig. 3
The correlation between staining scores of phosphatase and tensin homolog (PTEN) and Ki-67 and the number of patients with metastases.
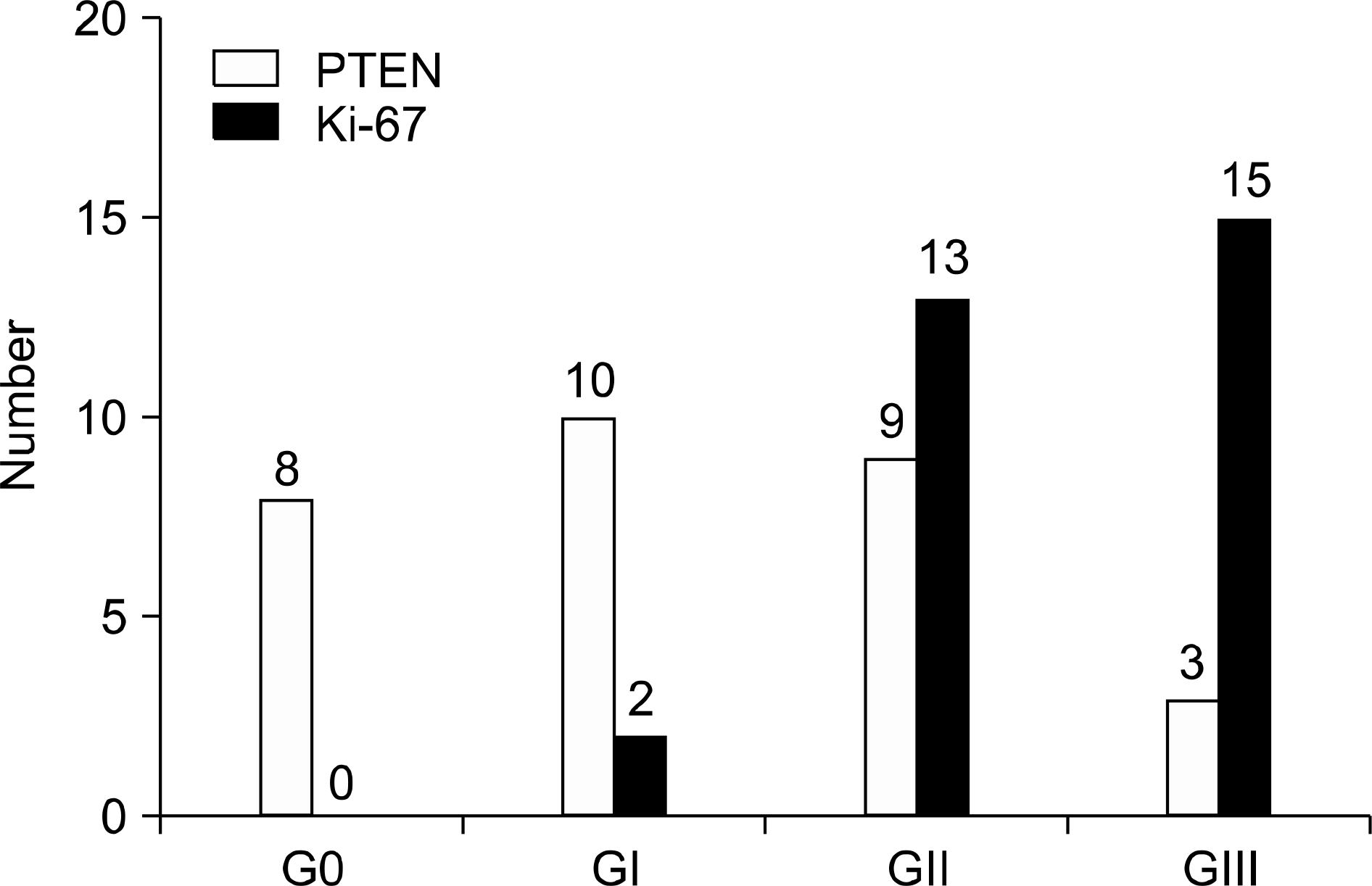
Table 1.
Comparison of clinicopathological characteristics according to phosphatase and tensin homolog (PTEN) expression
Table 2.
Comparison of clinicopathological characteristics accordingto Ki-67 expression




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download