Abstract
Purpose
We have previously demonstrated that increased urethral resistance was more prominent in diabetic rats than in controls. This may result from a compressive obstruction such as damage of the urethral nerve containing nitric oxide. Another possible cause for urethral obstruction could be a constrictive obstruction such as a periurethral fibrosis. In the present study, we investigated the changes in the expression of nitric oxide synthase (NOS) isoforms (compressive obstruction) and collagen subtypes (constrictive obstruction) in the urethral tissues of non-insulin dependent diabetic rats.
Materials and Methods
Thirty-six male Sprague-Dawley rats (18 diabetic rats and 18 control rats), bred from birth, were included in this study. Diabetes mellitus was induced by intraperitoneal administration of streptozotocin (90mg/kg) on the second day after birth. Urethral tissues were harvested at 12, 24 and 36 weeks after induction of diabetes and were stained for neuronal NOS (nNOS) and Masson trichrome. We also performed reverse transcriptase-polymerase chain reaction or Western blot analysis to evaluate mRNA or protein expression of NOS isoforms and collagen subtypes in the urethral tissues.
Results
Immunohistochemical staining and Western blot analysis of nNOS revealed that the immunoreactivity and nNOS expression in the urethra was lower in the diabetic rats than in the controls. The Masson trichrome staining showed that there was urethral fibrosis in the diabetic rats. The mRNA or protein expression of collagen subtypes, especially type I collagen, were higher in the diabetic rat urethra than in the controls.
Conclusions
These data suggest that the increased urethral resistance in diabetic rats may be attributable to a decrease in the urethral nNOS expression and an increase in collagen content. Urethral dysfunction as well as a cystopathy may play an important role in the pathogenesis of diabetes-induced voiding dysfunction.
Figures and Tables
Fig. 1
Immunohistochemical staining for the nNOS in the urethra of control (A series) and diabetic (B series) rats at 24 weeks of age. The nNOS immunoreactivities in each area were lower in the diabetic rats than the activities in the controls. Arrows denote nNOS-positive nerve fibers. Left figures, lamina propria (×200); Middle figures, smooth muscle layer (×400); Right figures, adventitia (×400). NOS: nitric oxide synthase.

Fig. 2
Western blot for nNOS and eNOS. (A) Representative gel pictures showing the protein expression of nNOS and eNOS in rat urethral tissues. (B) The nNOS protein expression was lower in the diabetic rats than in the controls at 24 and 36 weeks of age. (C) The eNOS protein expression was lower in diabetic rats than in the controls at 24 weeks of age. β-actin was used as a loading control. CON: control group, STZ: streptozotocin-induced diabetic group, NOS: nitric oxide synthase.
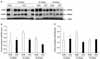
Fig. 3
Masson trichrome staining in 12-week-old control rats (A), 12-week-old diabetic rats (B), 24-week-old control rats (C), 24-week-old diabetic rats (D), 36-week-old control rats (E), and 36-week-old diabetic rats (F). Urethral fibrosis as evidenced by increase of collagen content compared to control rats was noted in diabetic rats.
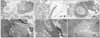
Fig. 4
RT-PCR for type I and type III collagen subtypes. (A) Representative gel pictures showing the gene expression of type I and type III collagen in rat urethral tissues. (B) The gene expression of type I collagen was higher in the diabetic rats than in the controls at 12 and 24 weeks of age. (C) No significant difference was found in the gene expression of type III collagen between the groups. GAPDH was used as an internal control for the RT-PCR. CON: control group, STZ: streptozotocin-induced diabetic group, GAPDH: glyceraldehyde-3-phosphate dehydrogenase, RT-PCR: reverse transcript-polymerase chain reaction.
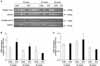
Fig. 5
Western blot analysis for type I collagen. (A) Representative gel pictures showing the protein expression of type I collagen in rat urethral tissues. (B) The expression of type I collagen was higher in the diabetic rats than in controls at 12 and 24 weeks of age. β-actin was used as a loading control. CON: control group, STZ: streptozotocin-induced diabetic group.
References
1. Kim SA, Park WS, Ohrr HC, Kang HY, Lee DH, Yi SW, et al. Prevalence and management status of diabetes mellitus in Korea. Korean J Med. 2005. 68:10–17.
2. Bryfogle JW, Bradley RF. The vascular complications of diabetes mellitus: a clinical study. Diabetes. 1957. 6:159–167.
3. Vinik AI, Holland MT, Le Bear JM, Liuzze FJ, Stansberry KB, Colen LB. Diabetic neuropathies. Diabetes Care. 1992. 15:1926–1975.
4. Frimodt-Moller C. Diabetic cystopathy: epidemiology and related disorders. Ann Intern Med. 1980. 92:318–321.
5. Motzkin D. The significance of deficient bladder sensation. J Urol. 1968. 100:445–450.
6. Ellenberg M. Development of urinary bladder dysfunction in diabetes mellitus. Ann Intern Med. 1980. 92:321–323.
7. Mumtaz FH, Sullivan ME, Thompson CS, Dashwood MR, Naseem KM, Bruckdorfer KR, et al. Alterations in the nitric oxide synthase binding sites and non-adrenergic, non-cholinergic mediated smooth muscle relaxation in the diabetic rabbit bladder outlet: possible relevance to the pathogenesis of diabetic cystopathy. J Urol. 1999. 162:558–566.
8. Derek JG. Pressure-flow studies of micturiation. Urol Clin North Am. 1996. 23:279–297.
9. Schafer W. Hinman F, editor. The contribution of the bladder outlet to the relation between pressure and flow rate during micturition. Benign prostatic hypertrophy. 1983. New York: Springer-Verlag;470.
10. Eika B, Levin RM, Longhurst PA. Collagen and bladder function in streptozotocin-diabetic rats: effects of insulin and aminoguanidine. J Urol. 1992. 148:167–172.
11. Koo HP, Santarosa RP, Buttyan R, Shabsigh R, Olsson CA, Kaplan SA. Early molecular changes associated with streptozotocin-induced diabetic bladder hypertrophy in the rat. Urol Res. 1993. 21:375–381.
12. Hellweg R, Hartung HD. Endogenous levels of nerve growth factor (NGF) are altered in experimental diabetes mellitus: a possible role for NGF in the pathogenesis of diabetic neuropathy. J Neurosci Res. 1990. 26:258–267.
13. Kim JC, Seo SL, Park YH, Hwang TK. Changes of detrusor contractility and growth factors in streptozotocin-induced NIDDM rat. Korean J Urol. 2000. 41:615–621.
14. Chung JS, Yoo HS, Lee T. Urethral dysfunction in a non-insulin dependent diabetes mellitus rat model. Korean J Urol. 2006. 47:1007–1012.
15. Weir GC, Clore ET, Zmachinski CJ, Bonner-Weir S. Islet secretion in a new experimental model for non-insulin-dependent diabetes. Diabetes. 1981. 30:590–595.
16. Sasaki K, Chancellor MB, Phelan MW, Yokoyama T, Fraser MO, Seki S, et al. Diabetic cystopathy correlates with a long-term decrease in nerve growth factor levels in the bladder and lumbosacral dorsal root Ganglia. J Urol. 2002. 168:1259–1264.
17. Low PA, Tuck RR, Dyck PJ, Schmelzer JD, Yao JK. Prevention of some electrophysiologic and biochemical abnormalities with oxygen supplementation in experimental diabetic neuropathy. Proc Natl Acad Sci USA. 1984. 81:6894–6898.
18. Thrainsdottir S, Malik RA, Dahlin LB, Wiksell P, Eriksson KF, Rosen I, et al. Endoneurial capillary abnormalities presage deterioration of glucose tolerance and accompany peripheral neuropathy in man. Diabetes. 2003. 52:2615–2622.
19. Buck AC, Reed PI, Siddiq YK, Chisholm GD, Fraser TR. Bladder dysfunction and neuropathy in diabetes. Diabetologia. 1976. 12:251–258.
20. Sima AA, Nathaniel V, Bril V, McEwen TA, Greene DA. Histopathological heterogeneity of neuropathy in insulin-dependent and non-insulin-dependent diabetes, and demonstration of axo-glial dysjunction in human diabetic neuropathy. J Clin Invest. 1988. 81:349–364.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download