Abstract
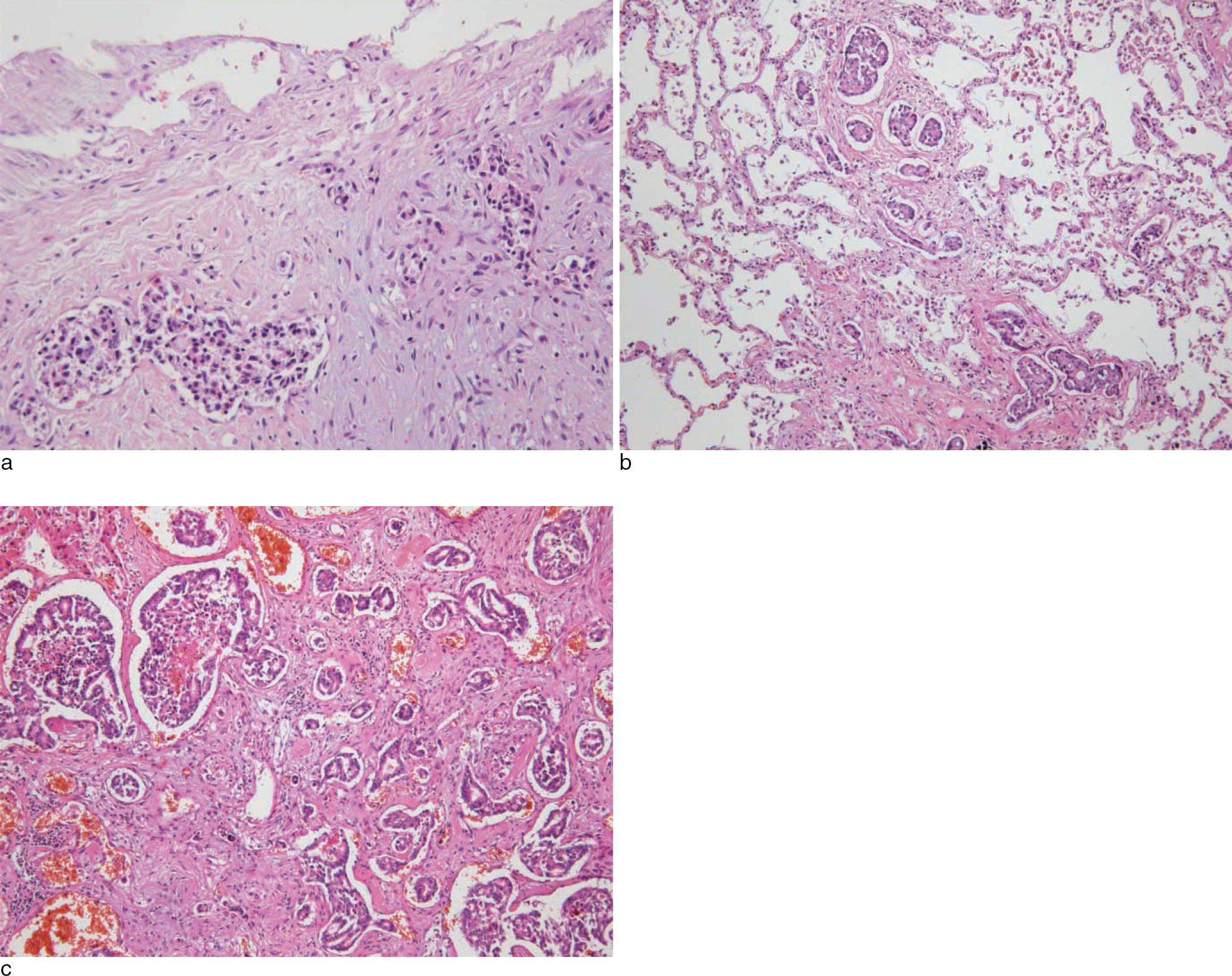
Dural metastasis associated with chronic subdural hematoma is very rare in patients with malignant neoplasms; it may be difficult to distinguish malignant neoplasms from chronic subdural hematoma. Chronic subdural hematoma is usually a late manifestation of malignant tumors, which contributes to the severity of the prognosis. The scans obtained by using magnetic resonance imaging or computed tomography may be misleading when a subdural hematoma masks the underlying tumor. Herein, we report a case of a subdural hematoma with dural metastasis of unknown origin in a 45-year-old woman; however, the neoplasm was not detected until autopsy.
REFERENCES
1. Tseng SH, Liao CC, Lin SM, et al. Dural metastasis in patients with malignant neoplasm and chronic subdural hematoma. Acta Neurol Scand. 2003; 108:43–6.

2. Posner JB. Intracranial metastases. Posner JB, editor. ed.Neurologic Complications of Cancer. Philadelphia: FA Davis;1995. p. 77–82.
3. Meyer PC, Reah TG. Secondary neoplasms of the central nervous system and meninges. Br J Cancer. 1953; 7:438–48.

4. Laigle-Donadey F, Taillibert S, Mokhtari K, et al. Dural metastasis. J Neurooncol. 2005; 75:57–61.
5. Kleinschmidt-DeMasters BK. Dural metastases: a retrospective surgical and autopsy series. Arch Pathol Lab Med. 2001; 125:880–7.
6. Nagayama I, Katoh H, Sakumoto M, et al. Subdural hematoma associated with dural metastasis from paranasal sinus cancer: a case report and review of literature. ORL J Otorhinolaryngol Relat Spec. 1993; 55:240–3.
7. Cheng CL, Greenberg J, Hoover LA. Prostatic adenomcarcinoma metastatic chronic subdural hematoma membrane. Case report. J Neurosurg. 1998; 68:642–4.
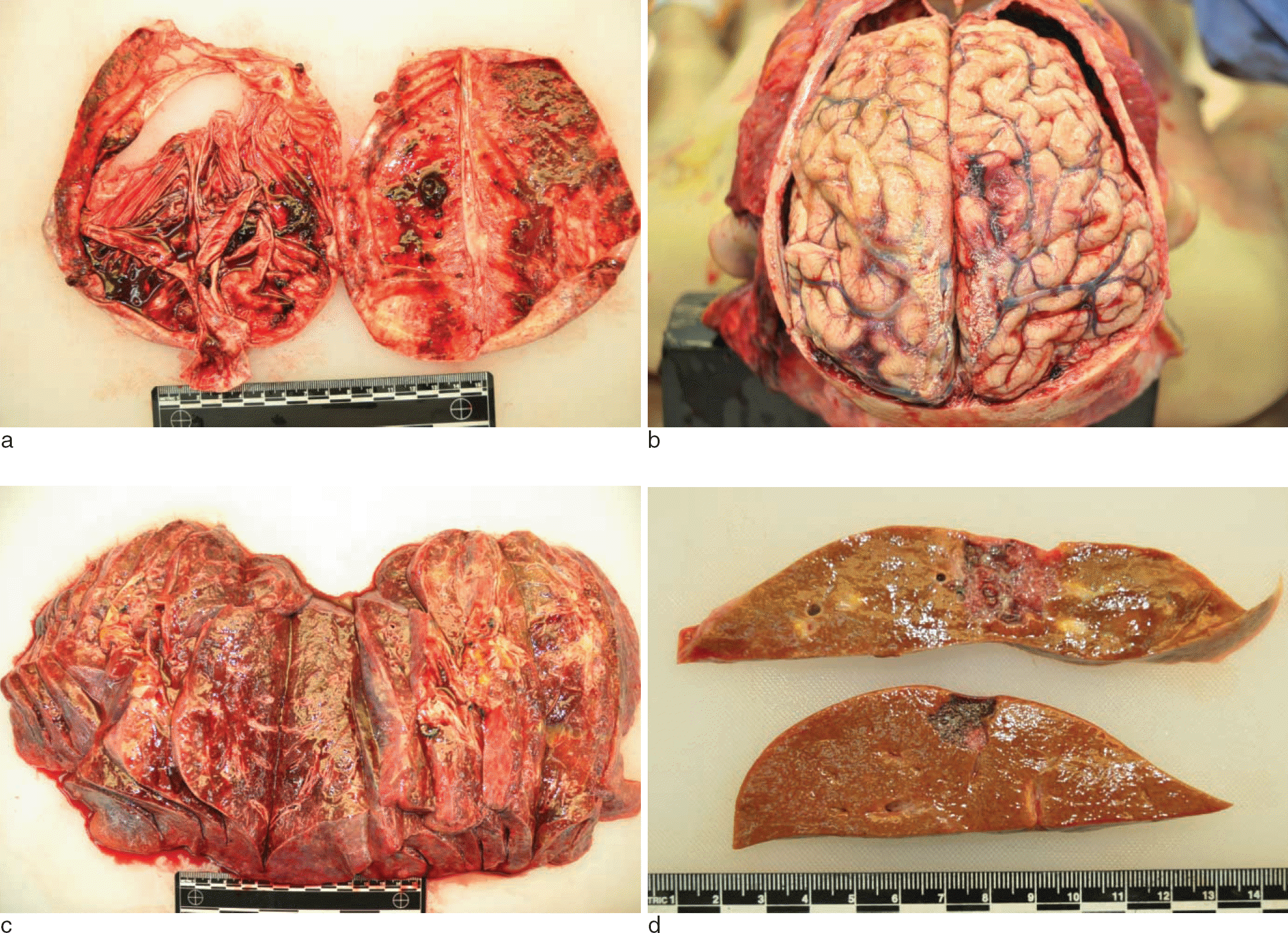
Fig. 1.
There was a small amount of mixed fresh and chronic hemorrhages in the subdural spaces of both sides (a). The underlying hemisphere was slightly edematous (b). Both lungs showed diffuse pneumonia patterns with occasional tiny nodules in all the lobes (c). The liver showed two hemorrhagic nodules. 3.0 × 2.0 cm in size in right lobe and 1.5 × 1.5 cm size in left lobe (d).




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download