Abstract
Objective:
Small interfering RNA (siRNA) triggers RNA interference in mammalian somatic cells. TNF-α is a proinflammatory cytokine implicated in the pathogenesis of inflammatory arthritis including rheumatoid arthritis (RA). This study was to use TNF receptor 1 (TNFRI)- specific siRNA to inhibit the TNF-α mediated signaling in RA fibroblast like synoviocytes (FLS) and chondrocytes.
Methods:
TNFRI specific siRNA was produced by targeting 3 nucleotide sequences at 474 ~ 494, 562~582 and 668~688. Reverse transcriptase polymerase chain reaction (RT-PCR) and Western blot were performed to optimize the silencing effects of TNFRI siRNA in cultured FLS and chondrocytes. The inhibition of TNF-α mediated signaling was determined by ELISA assay of metalloproteinase 1 secretion induced by TNF-α.
Go to : 
REFERENCES
1). Choy EH., Panayi GS. Cytokine pathways and joint inflammation in rheumatoid arthritis. N Engl J Med. 2001. 344:907–16.

2). Clark IA. How TNF was recognized as a key mechanism of disease. Cytokine Growth Factor Rev. 2007. 18:335–43.

3). Lipsky PE., van der Heijde DM., St ., Clair EW., Furst DE., Breedveld FC., Kalden JR, et al. Infliximaband methotrexate in the treatment of rheumatoid arthritis: anti-tumor necrosis factor trial in rheumatoid arthritis with concomitant therapy study group. N Engl J Med. 2000. 343:1594–602.
4). Weinblatt ME., Kremer JM., Bankhurst AD., Bulpitt KJ., Fleischmann RM., Fox RI, et al. A trial of etanercept, a recombinant tumor necrosis factor receptor: Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N Engl J Med. 1999. 340:253–9.
6). Elbashir SM., Harborth J., Lendeckel W., Yalcin A., Weber K., Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001. 411:494–8.

7). Harborth J., Elbashir SM., Bechert K., Tuschl T., Weber K. Identification of essential genes in cultured mammalian cells using small interfering RNAs. J Cell Sci. 2001. 114:4557–65.

8). Kim HA., Kim S., Chang SH., Hwang HJ., Choi YN. Anti-arthritic effect of ginsenoside Rb1 on collagen induced arthritis in mice. Int Immunopharmacol. 2007. 7:1286–91.

9). Bernstein E., Caudy AA., Hammond SM., Hannon GJ. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature. 2001. 409:363–6.

10). Manche L., Green SR., Schmedt C., Mathews MB. Interactions between double-stranded RNA regulators and the protein kinase DAI. Mol Cell Biol. 1992. 12:5238–48.

11). Saito F., Yokota H., Sudo Y., Yakabe Y., Takeyama H., Matsunaga T. Application of RNAi inducible TNFRI knockdown cells to the analysis of TNFalpha-induced cytotoxicity. Toxicol In Vitro. 2006. 20:1343–53.
12). Zhou HW., Lou SQ., Zhang K. Recovery of function in osteoarthritic chondrocytes induced by p16INK4a-specific siRNA in vitro. Rheumatology. 2004. 43:555–68.

13). Lianxu C., Hongti J., Changlong Y. NF-kappaB p65-specific siRNA inhibits expression of genes of COX-2, NOS-2 and MMP-9 in rat IL-1 beta-induced and TNF-alpha-induced chondrocytes. Osteoarthritis Cartilage. 2006. 14:367–76.
14). Klatt AR., Klinger G., Zech D., Paul-Klausch B., Renno JH., Schmidt J, et al. RNAi in primary human chondrocytes: Efficiencies, kinetics, and non-specific effects of siRNA-mediated gene suppression. Biologicals. 2007. 35:321–8.

15). Robbins PD., Evans CH., Chernajovsky Y. Gene therapy for arthritis. Gene Ther. 2003. 10:902–11.

16). Schiffelers RM., Xu J., Storm G., Woodle MC., Scaria PV. Effects of treatment with small interfering RNA on joint inflammation in mice with collagen-induced arthritis. Arthritis Rheum. 2005. 52:1314–8.

17). Chen LX., Lin L., Wang HJ., Wei XL., Fu X., Zhang JY, et al. Suppression of early experimental osteoarthritis by in vivo delivery of the adenoviral vector-mediated NF-kappaBp65-specific siRNA. Osteoarthritis Cartilage. 2007. doi: 10.1016/j.biologicals.2007.06.006.
Go to : 
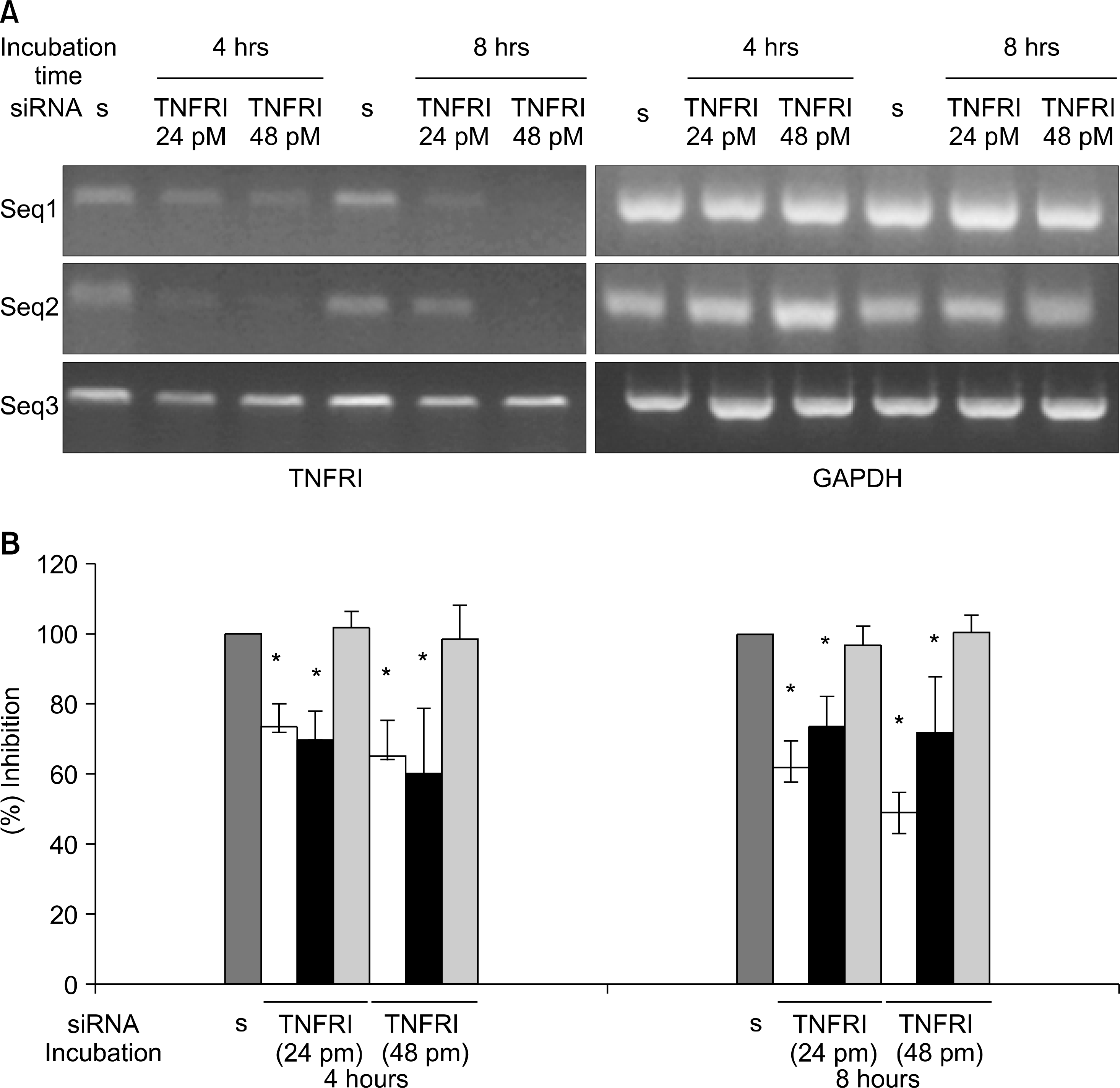 | Fig. 1.Inhibition of TNFRI mRNA by TNFRI specific siRNA in articular chondrocytes. (A) Expression of TNFRI mRNA in articular chondrocytes were detected by RT-PCR. Data represent samples from 3 different donors. (B) The band densities were quantified, the percent GAPDH density was calculated for TNFRI and the value for control was set at 100. ∗p<0.05 compared with control by Mann-Whitney U test. s: scrambled siRNA(negative control). |
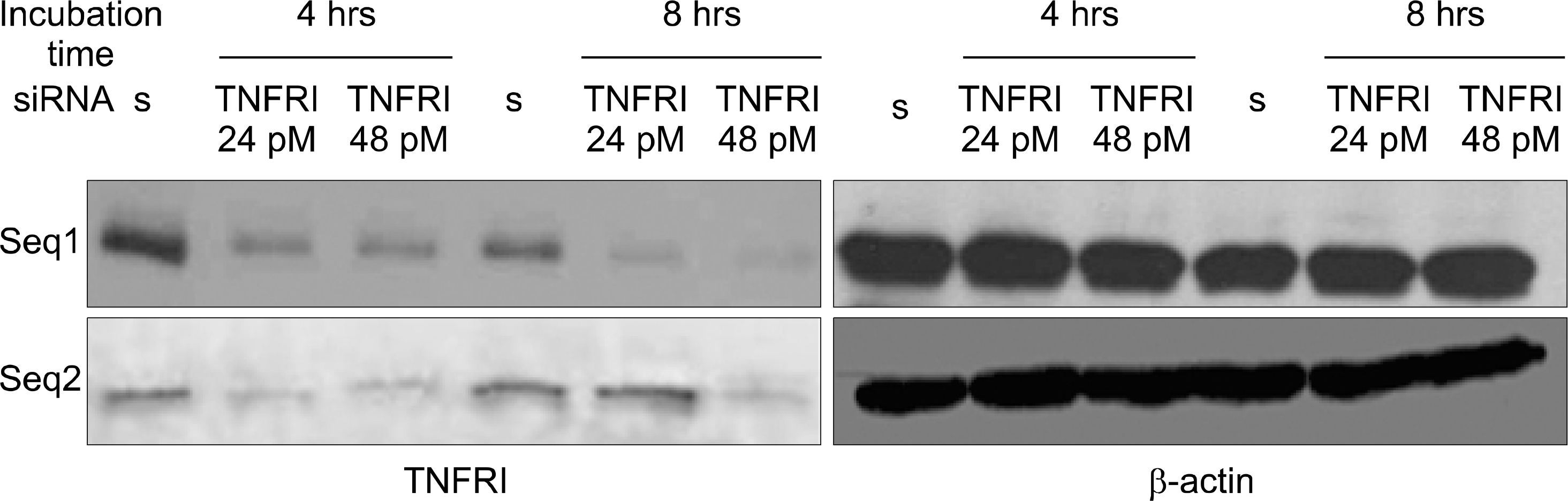 | Fig. 2.Inhibition of TNFRI protein by TNFRI specific siRNA in articular chondrocytes. Expression of TNFRI protein in articular chondrocytes was detected by Western blot. Data represent samples from 3 different donors. s: scrambled siRNA. |
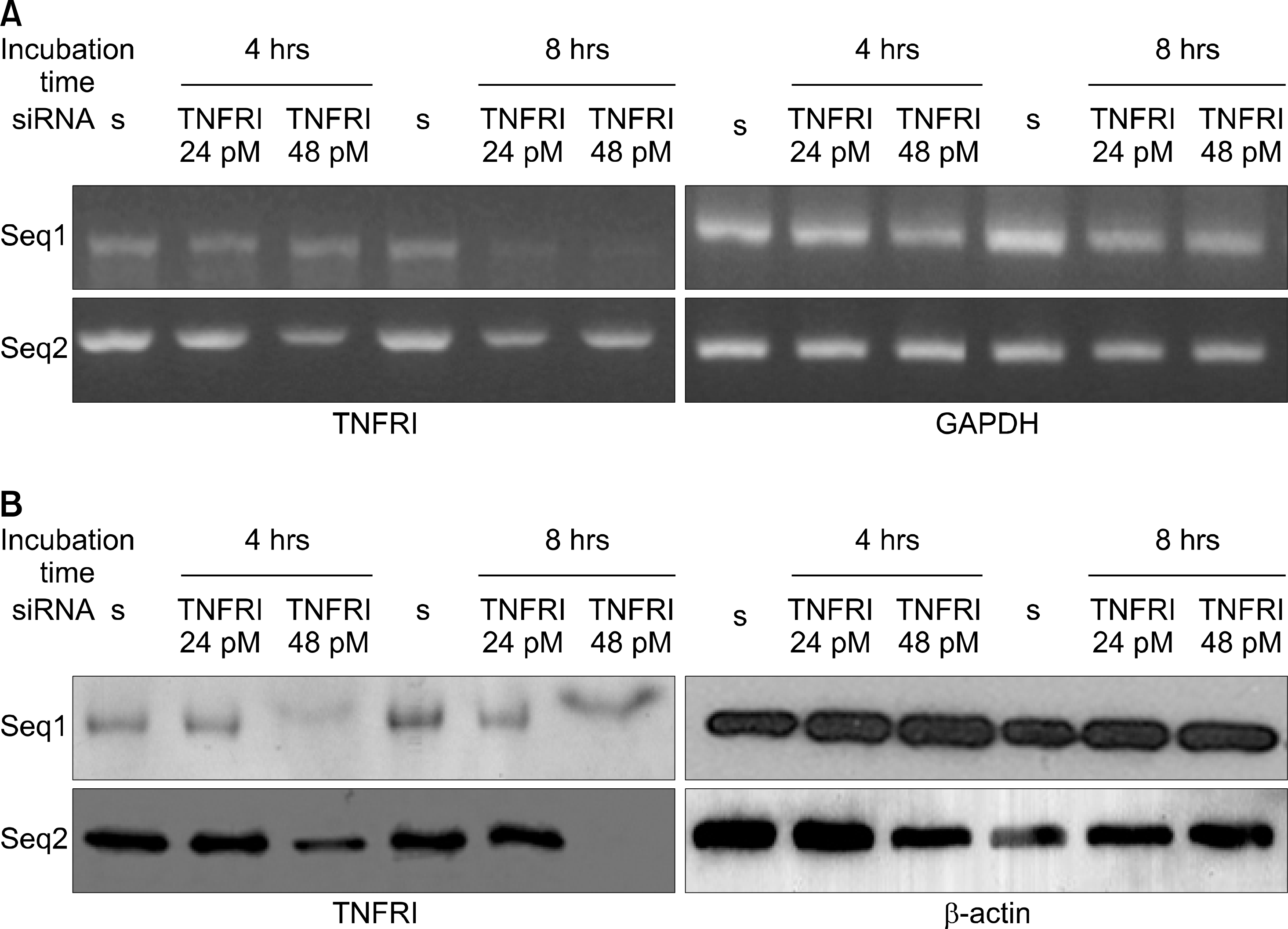 | Fig. 3.Inhibition of TNFRI mRNA and protein by TNFRI specific siRNA in RA fibroblast like synoviocytes(FLS). (A) Expression of TNFRI mRNA in RA FLS were detected by RT-PCR. Data represent samples from 5 different donors. (B) Expression of TNFRI protein in articular chondrocytes was detected by Western blot. Data represent samples from 4 different donors. s: scrambled siRNA. |
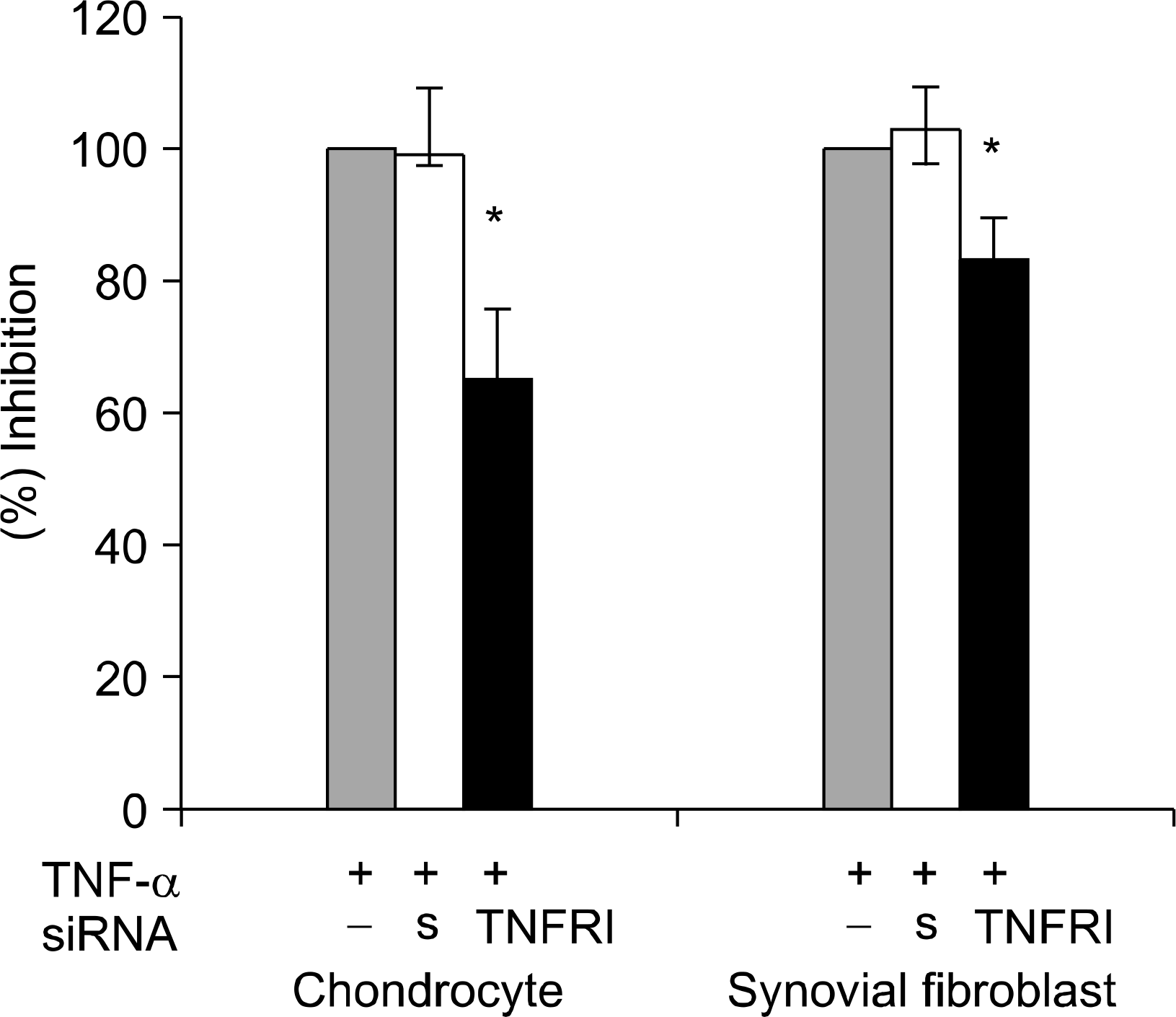 | Fig. 4.Inhibition of MMP-1 secretion induced with TNF- α by TNFRI siRNA. MMP-1 production was measured 24 hours after TNF- α treatment from the conditioned media by ELISA. Results are given as the mean and standard deviation of the mean percentage of TNF- α treatment alone (control, representative of duplicate experiments from 4 donors). ∗p<0.05 compared with control by Mann-Whitney U test. s: scrambled siRNA (negative control) |
Table 1.
The sequences of siRNA used in the experiment




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download