Abstract
Androgen insensitivity syndrome (AIS) is a hereditary disorder that's characterized by the female phenotype in spite of the 46, XY karyotype, and this is caused by mutation of the androgen receptor gene. We experienced a case of the complete type of AIS. A 20-yr-old woman was evaluated for primary amenorrhea. The patient had external genitalia of the female phenotype, but she had no ovaries or uterus. The abdominal computed tomography scan revealed suspected testes in the pelvic cavity. The chromosome analysis was reported as 46, XY. We identified an androgen receptor gene novel mutation, including CAT deletion at the position 1925~1927 and AG deletion at the position 2129~2130 of exon 5, in both the proband and her sister. The patient underwent laparoscopic gonadectomy due to the possibility of malignant tumor developing in the testes. The subject is now on estrogen supplementation and she is under regular follow-up; she is in a good condition.
Figures and Tables
Fig. 2
Computed tomography of abdomen showed bilateral pelvic cavity masses suspected with testicular tumors (arrows).
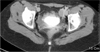
Fig. 4
Scheme of the androgen receptor gene mutation. The underlines indicate the position of nucleotide deletions. The arrow indicates the position of frame-shift. Amino acids are shown with a three letter code.

Fig. 5
Gross sections of the resected gonads. A. Right gonad was surrounded by whitish tunica and had yellow-gray nodule. B. Left gonad was composed of gray parenchyme and had multiple nodules.

Fig. 6A
Histology of the resected gonads. A hamartomatous nodule was encapsulated by thin connective tissue and was composed of compacted seminiferous tubules with little stromal Leydig cells (H&E stain ×100).
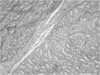
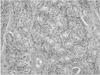
Fig. 6B
Histology of the resected gonads. Surrounding parenchyme consisted of tubules and proliferated stromal Leydig cells (H&E stain ×200).
References
1. Morris JM. The syndrome of testicular feminization in male pseudohermaphrodites. Am J Obstet Gynecol. 1953. 65:1192–1211.
2. French FS, Van Wyk JJ, Baggett B, Easterling WE, Talbert LM, Johnston FR, Forchielli E, Dey AC. Further evidence of a target organ defect in the syndrome of testicular feminization. J Clin Endocrinol Metab. 1966. 26:493–503.
3. Dewhurst CJ, Ferreira HP, Gillett PS. Gonadal malignancy in XY females. J Obstet Gynaecol Br Commonw. 1971. 78:1077–1083.
4. Gottlieb B. Androgen receptor gene mutations database.
www.mcgill.ca/androgendb
.
5. Mashchak CA, Kletzky OA, Davajan V, Mishell DR Jr. Clinical and laboratory evaluation of patients with primary amenorrhea. Obstet Gynecol. 1981. 57:715–721.
6. Rhee JH. Clinical investigation of primary amenorrhea. Korean J Obstet Gynecol. 2002. 45:1045–1051.
7. Morris JM, Mahesh VB. Further observations on the syndrome, "Testicular feminization". Am J Obstet Gynecol. 1963. 87:731–748.
8. Schmitt S, Knorr D, Schwartz HP. Gonadotropin regulation during puberty in complete androgen insensitivity syndrome with testicles in situ. Horm Res. 1994. 42:253–256.
9. Connell MJ, Ramsey HE, Whang-Peng J. Testicular feminization syndrome in three sibs: emphasis on gonadal neoplasia. Am J Med Sci. 1973. 265:321–333.
10. Bangsbll S, Qvist I, Lebech P. Testicular feminization syndrome and associated gonadal tumors in Denmark. Acta Obstet Gynecol Scand. 1992. 71:63–66.
11. Damjanov I, Nesbitt KA, Reardon MP. Giant sertoli cell adenoma in testicular feminization syndrome. Obstet Gynecol. 1976. 48:624–627.
12. Jo JH, Park YB, Park TH, Jang WY, Kim DG, Kim KW, Kim DK, Yoon HW, Kim DH, Kim MW, Park ST. A case report of completed testicular feminization syndrome with rudimentary salpinx. Korean J Obstet Gynecol. 2001. 44:986–989.
13. Mizunuma H, Soda M, Okano H, Kagami I, Miyamoto S, Ohsawa M, Ibuki Y. Changes in bone mineral density after orchidectomy and hormone replacement therapy in individuals with androgen insensitivity syndrome. Hum Reprod. 1998. 13:2816–2818.
14. Weidemann W, Linck B, Haupt H, Mentrup B, Romalo G, Stockklauser K, Brinkmann AO, Schweikert HU, Spindler KD. Clinical and biochemical investigations and molecular analysis of subjects with mutations in the androgen receptor gene. Clin Endocrinol (Oxf). 1996. 45:733–739.
15. Lee HJ, Mowszowicz I, Chang C. The first detection of complete androgen insensitivity with no mutation in the coding sequence of the androgen receptor gene. Front Biosci. 1996. 1:a34–a38.
16. Park SY, Choi YM, Park SH, Yang SW, Ku SY, Kim SH, Kim SW, Paik JS, Yang DH, Choi DS, Kwon HC, Choi DH, Lee SH. Analysis of androgen receptor gene in Korean patients with androgen insensitivity syndrome. J Korean Obstet Gynecol. 2001. 44:655–662.
17. Griffin JE, Wilson JD. The syndromes of androgen resistance. N Engl J Med. 1980. 302:198–209.
18. McPhaul MJ, Marcelli M, Zoppi S, Griffin JE, Wilson JD. Genetic basis of endocrine disease 4. The spectrum of mutations in the androgen receptor gene that cause androgen resistance. J Clin Endocrinol Metab. 1993. 76:17–23.
19. Quigley CA, De Bellis A, Marschke KB, el-Awady MK, Wilson EM, French FS. Androgen receptor defects: historical, clinical, and molecular perspectives. Endocr Rev. 1995. 16:271–321.
20. Kim HJ, Ko DS, Lee JY, Hong SY, Shin JH, Park CH, Park EJ, Seo YS, Park WI. Complete androgen insensitivity syndrome due to androgen receptor gene mutation in the amino acid position 607 which bilateral testes situated in the normal ovary sites. Korean J Obstet Gyn. 2006. 49:1795–1802.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download