Abstract
Craniopharyngioma accounts for 2~5% of all primary intracranial neoplasms. It may present with a variety of manifestations including neurological, visual, and/or hypothalamic-pituitary dysfunction. Treatment options include radical surgery or radiotherapy, or a combination of these modalities. Craniopharyngioma ablation results in anterior and/or posterior pituitary hormone deficits. Slipped capital femoral epiphysis (SCFE), in which the femoral head slips downward and backward on the femoral neck at the epiphyseal plate, most commonly occurs during the rapid growth phase of puberty. Its actual cause is unknown, but the clinical association between SCFE and endocrine disorders is well known. We report a case of an adult male patient who developed SCFE in association with panhypopituitarism after treatment of a craniopharyngioma.
Figures and Tables
Figure 1
Frontal view of external genitalia of the patient showed hypoplastic genitalia with no pubic hair. Penis was 3 cm in length. The volume of right testis was 2 cc, and that of left was 1 cc.

Figure 2
A. Preoperative both hip A-P view showed downward and posterior slipping of the left femoral epiphysis. B. Postoperative both hip A-P view showed internal fixation with cannulated screw of both femoral epiphysis.
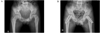
Figure 3
Hand X-ray showed bone age corresponding with 12 years. All carpal bones were shown. The epiphyseal growth plates of metacarpal, phalangeal, distal ulnar and radial bone were not closed.
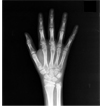
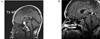
Figure 4
A. Preoperative sagittal T1 - weighted sellar MRI showed 1.8 cm sized well circumscribed mass with suprasellar extension. B. 12 years later, sagittal T1 - weighted sellar MRI showed empty sella with no remnant mass.
References
1. Karavitaki N, Cudlip S, Adams CB, Wass JA. Craniopharyngiomas. Endocr Rev. 2006. 27:371–397.
2. Chung JH, Lee HC, Yoo NC, Chung YS, Lim SK, Kim KR, Huh KB. Clinical aspects on craniopharyngioma. J Kor Soc Endocrinol. 1991. 6:163–169.
3. Paja M, Lucas T, García-Uría J, Salamé F, Barceló B, Estrada J. Hypothalamic-pituitary dysfunction in patients with craniopharyngioma. Clin Endocrinol (Oxf). 1995. 42:467–473.
4. Karavitaki N, Brufani C, Warner JT, Adams CB, Richards P, Ansorge O, Shine B, Turner HE, Wass JA. Craniopharyngiomas in children and adults: systematic analysis of 121 cases with long-term follow-up. Clin Endocrinol (Oxf). 2005. 62:397–409.
5. Loder RT, Wittenberg B, DeSilva G. Slipped capital femoral epiphysis associated with endocrine disorders. J Pediatr Orthop. 1995. 15:349–356.
6. Harris WR. The endocrine basis of slipping of the upper femoral epiphysis. An experimental study. J Bone and Joint Surg. 1950. 32:5.
7. Blethen SL, Rundle AC. Slipped capital femoral epiphysis in children treated with growth hormone. A summary of the National Cooperative Growth Study experience. Horm Res. 1996. 46:113–116.
8. Jeon HJ, Ko BS, Kim DH, Bae JH, Oh TG, Kang SB. Kallmann's syndrome associated with slipped capital femoral epiphysis. J Kor Soc Endocrinol. 1996. 11:318–323.
9. Wilcox PG, Weiner DS, Leighley B. Maturation factors in slipped capital femoral epiphysis. J Pediatr Orthop. 1988. 8:196–200.
10. Rappaport EB, Fife D. Slipped capital femoral epiphysis in growth hormone-deficient patients. Am J Dis Child. 1985. 139:396–399.
11. Ippolito E, Mickelson MR, Ponseti IV. A histochemical study of slipped capital femoral epiphysis. J Bone Joint Surg Am. 1981. 63:1109–1113.
12. Hochberg Z. Clinical physiology and pathology of the growth plate. Best Pract Res Clin Endocrinol Metab. 2002. 16:399–419.
13. Alini M, Kofsky Y, Wu W, Pidoux I, Poole AR. In serum-free culture thyroid hormones can induce full expression of chondrocyte hypertrophy leading to matrix calcification. J Bone Miner Res. 1996. 11:105–113.
14. Dearden LC. Enhanced mineralization of the tibial epiphyseal plate in the rat following propylthiouracil treatment: a histochemical, light, and electron microscopic study. Anat Rec. 1974. 178:671–689.
15. Weise M, De-Levi S, Barnes KM, Gafni RI, Abad V, Baron J. Effects of estrogen on growth plate senescence and epiphyseal fusion. Proc Natl Acad Sci U S A. 2001. 98:6871–6876.
16. Isaksson OG, Lindahl A, Nilsson A, Isgaard J. Mechanism of the stimulatory effect of growth hormone on longitudinal bone growth. Endocr Rev. 1987. 8:426–438.
17. Wells D, King JD, Roe TF, Kaufman FR. Review of slipped capital femoral epiphysis associated with endocrine disease. J Pediatr Orthop. 1993. 13:610–614.
18. Hägglund G, Hansson LI, Ordeberg G, Sandström S. Bilaterality in slipped upper femoral epiphysis. J Bone Joint Surg Br. 1988. 70:179–181.
19. Bunin GR, Surawicz TS, Witman PA, Preston-Martin S, Davis F, Bruner JM. The descriptive epidemiology of craniopharyngioma. J Neurosurg. 1998. 89:547–551.
20. Van Effenterre R, Boch AL. Craniopharyngioma in adults and children: a study of 122 surgical cases. J Neurosurg. 2002. 97:3–11.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download