Abstract
A small number of viable tuberculosis bacilli can reside in an individual with latent tuberculosis infection (LTBI) without obvious clinical symptoms or abnormal chest radiographs. Diagnosis and treatment for LTBI are important for tuberculosis (TB) control in public and private health, especially in high-risk populations. The updated 2014 Korean guidelines for TB recommend that tuberculin skin tests, interferon-gamma release assays, or a combination of the two can be used for LTBI diagnosis according to age and immune status of the host as well as TB contact history. The regimens for LTBI treatment include isoniazid, rifampicin, or isoniazid/rifampicin. However, results of drug susceptibility test from the index case must be considered in selecting the appropriate drug for recent contacts. Standardized LTBI diagnosis and treatment based on the new 2014 guidelines will contribute to the effective TB control in Korea as well as to the establishment of updated guidelines.
When humans are infected with Mycobacterium tuberculosis, tuberculosis (TB) only develops in 10% of those infected. TB develops in 5% of those infected, mostly within 1-2 years of M. tuberculosis infection1.
Latent tuberculosis infection (LTBI) is the state in which human are infected with M. tuberculosis without any clinical symptoms, radiological abnormality, or microbiological evidence2.
The TB burden of South Korea is intermediate, where the annual incidence of TB in 2013 was reported as 97/100,0003. LTBI treatment was not emphasized until 2011, when Korean TB guidelines included aggressive LTBI treatment. Because of frequent TB outbreaks in schools and increasing number of immune-compromised patients including the elderly, guidelines for LTBI were enhanced in 20114 and updated in 20145. In this review, we will focus on the main points and revised contents of the 2014 Korean guidelines for TB.
The LTBI rate in Korea, estimated by tuberculin skin test (TST) among 20-24 years old in 1985 was 74.5%, and it was 59.3%6 in 1995. The TST positive rate among 15-20 years old and young military soldiers in the mid 2000 was 20%-30%7,8; but the rate in the late 2000 and in the early 2010, in which most subjects did not receive bacillus Calmette-Guérin (BCG) revaccination, were decreased to 14%-16% in Koreans in their late teens and early twenties9.
LTBI diagnosis is based on the targeted TST strategy, which recommends a test for the subjects who have risk factors for developing active TB from LTBI10. In the case of suspicious new infection for subjects who had been treated for LTBI or active TB, LTBI treatment can be done considering the risk of active TB progression.
For LTBI diagnosis, exclusion of active TB is essential, and the old healed TB without TB treatment history among the bellowed risk groups for TB development is considered as LTBI, even without an LTBI test11. A TST test using RT-23 2TU and an interferon-gamma release assay (IGRA) test are used to diagnose LTBI. There are commercial IGRA tests such as QuantiFERON-TB Gold In Tube (QFT-IT; Qiagen, Valencia, CA, USA) and T-SPOT.TB (Oxford Immunotec, Abingdon, UK). However, these diagnostic tools do not differentiate LTBI from active TB infection.
TST is the basic method to diagnose LTBI in immune-competent subjects10,12, but IGRA alone can be used13. Furthermore, a TST/IGRA two-step strategy in which the initial TST is followed by confirmatory IGRA for TST positive subjects can be used, because false positivity due to BCG vaccination or nontuberculous mycobacteria (NTM) infection can be reduced by additional IGRA test with high specificity14,15. The diagnostic approach algorithm is presented in Figure 1.
IGRA alone or TST combined with IGRA can be used, but exclusion of LTBI using TST-negative result alone is not recommended. Either TST-positive or IGRA positive is regarded as LTBI in TST/IGRA combined method. The TST/IGRA two-step strategy is not recommended in immune-compromised subjects16 in contrast to immune-competent subjects17. Old spontaneously healed TB lesion without TB treatment history is regarded as LTBI, even when the LTBI test is negative. An indeterminate result on the initial IGRA test in immune-compromised subjects is frequently repeated on subsequent IGRA test18, so LTBI confirmation can be made based on TST result. The diagnostic approach algorithm is presented in Figure 2.
A serial test refers to a TST test following an initial TST in contact investigation to exclude a false-negative result during the period of 8 weeks after the last contact with an infectious TB patient (window period); this is when the TST does not lead to complete response due to delayed hypersensitivity19. On the other hand, a two-step TST, which is usually conducted for healthcare workers, means subsequent TST 1-4 weeks after the initial TST20. This is because delayed response triggered by initial TST can appear due to recall immune phenomenon, in the subjects who are previously infected with latent TB (booster effect)19. In the subjects whose basal initial TST result is negative, TST conversion (from negative to positive) after several months can strongly suggest a new TB infection19.
Serial TST is recommended in high/moderate risk groups (Table 1) and children ≤18 years of age. In the subjects with TST <5 mm, TST conversion is confirmed with a subsequent TST ≥10 mm. In the subjects with TST 5-9 mm, TST conversion is confirmed with subsequent TST increase ≥6 mm19. This rule applies to regular follow-up checks for healthcare workers. But in contact investigation, a TST increase of ≥6 mm from the initial TST result (e.g., 3 mm to 9 mm) indicates conversion under the following conditions: highly infectious index case, close TB contacts, long duration of contact, contacts <5 years, or immune-compromised contacts19.
LTBI diagnosis is based upon the premise that the subjects with a positive LTBI test must be treated. The candidates for LTBI diagnosis is categorized into two groups: those with contact with infectious TB patients and those without contact with infectious TB patients.
The candidates for LTBI diagnosis can be classified as high risk groups and moderate risk groups22. For the subjects with LTBI-positive in the high risk group, LTBI treatment must be conducted; but for those with LTBI-positive in the moderate risk group, LTBI treatment can be considered (Table 1). In 2014 Korean new guideline, head and neck cancer and hematologic malignancy were included in the moderate risk group24.
In contact investigation condition, candidates for LTBI screening is restricted to those who are ≤35 years old, because risk owing to hepatotoxicity by anti-TB drugs exceed the benefit of LTBI treatment. For those who are ≤35 years old with LTBI-positive, LTBI treatment is obligatory17. For LTBI-positive subjects in the above mentioned high/moderate risk groups, LTBI treatment is necessary for those that had contact with an infectious TB patient (Table 2). The patients with head and neck cancer or hematologic malignancy were added for this group in the 2014 Korean guidelines.
A serial repeated TST must be performed for people in the high-moderate risk group, who had an initial negative TST result after 8 weeks since their last contact with an infectious TB patients25. LTBI treatment must be maintained during the window period even though the initial TST result was negative for contacts <2 years old17, those on immunosuppressive agents who received a transplant, or those on tumor necrosis factor (TNF) α blockers.
Based on the repeated TST result after 8 weeks of window period, LTBI treatment must be continued for the contact with a positive result, but LTBI treatment can be stopped for those with a negative result. However, for the human immunodeficiency virus-positive, LTBI treatment must be continued even after a repeated TST-negative result. For the contacts with initial TST-negative result in the moderate risk group, the decision for LTBI treatment must be delayed until a repeated TST is taken after 8 weeks of window period. The risk groups are as follows: between 2-18 years of age; taking long-term steroids or scheduled to take long-term steroids; having silicosis, end stage renal failure, diabetes mellitus, head and neck cancer, or hematologic malignancy; and gastrectomy/jejunoileal bypass or scheduled for one. For contacts with TST conversion (from TST-negative to TST-positive), LTBI treatment must be performed; but for people without TST conversion (from TST-negative to TST-negative), LTBI treatment is unnecessary25.
The drug susceptibility test (DST) from the index case must be considered when selecting the appropriated drug for recent TB contacts, and the LTBI treatments are as follows.
Isoniazid (5 mg/kg/day, maximum 300 mg/day) for 9 months (9H) is recommended, but isoniazid for 6 months (6H) can be reasonable considering the cost-effectiveness26.
Isoniazid/rifampicin combination regimen for 3 month (3HR) is recommended by the National Institutes for Health and Clinical Excellence (NICE) guideline17. This regimen has excellent treatment compliance rate28 and was reported to be superior to 9H regimen in treatment efficacy29. Moreover, isoniazid / rifapentine combination regimen (once a week for 12 weeks) was recently introduced as a new regimen30, but rifapentine is not available yet in Korea.
Basal complete blood count, aspartate aminotransferase/alanine aminotransferase, and bilirubin must be checked before LTBI treatment. A liver function test must be checked monthly for the subjects with basal abnormal liver function test results or with risk factors. There is no method to verify complete eradication of LTBI, so TST or IGRA result does not verify complete LTBI treatment.
The 9H regimen is recommended, and pyridoxine must be supplied to infant whose mother is being treated with isoniazid for LTBI. The 4R regimen can be given to pregnant women, but further investigation is needed for this regimen.
Generally, LTBI treatment is not recommended due to lack of data, and follow-up for 2 years is recommended10.
The guidelines for LTBI diagnosis for these subjects is same for the guidelines for immune-compromised patients. IGRA alone or TST/IGRA combination tests can be used, but LTBI diagnosis with TST alone is not recommended. In TST/IGRA combination tests, LBTI can be confirmed by either TST-positive or IGRA-positive31. A TST-positive criterion is induration size ≥10 mm. Two-step TST can be considered, if applicable, to increase the sensitivity of LTBI diagnosis in immune-compromised subjects32.
Active TB detected by basal examination must be treated, and TNF blockers are recommended to be started after the completion of active TB treatment. But, TNF blockers can be started after 2 months of intensive treatment for active TB33, under the following conditions: the disease is not severe, responses to anti TB drugs are favorable, and the strain is drug-susceptible. LTBI detected by basal examination must be treated. TNF blockers are recommended to be started 3 weeks after the initial LTBI treatment34, but TNF blockers treatment and LTBI treatment may be initiated at the same time35.
Prompt examination reacting to the symptoms suggesting TB is preferred to regular chest radiographs screening for TB development. The incidences of extrapulmonary tuberculosis, NTM disease, or severe TB are high during the uses of TNF blockers, so particular attention must be paid.
The examinations for active TB and LTBI must be performed for patients with organ transplants, who are taking or will be taking immunosuppressive agents. LTBI diagnosis comply with the guidelines for immune-compromised subjects. LTBI screening is preferably recommended before immunosuppressive agents are begun, to decrease the indeterminate result of IGRAs.
In a Korean study where TST and T-SPOT.TB tests for LTBI diagnosis and treatment were performed for 312 patients with kidney transplants, T-SPOT.TB tests for the patients with TST-negative results were useful in predicting active TB development36. Based on either TST-positive or IGRA-positive after excluding active TB, LBTI is confirmed because the concordance rate is not so high between TST and IGRAs for the patients waiting for kidney transplants37,38. In the same context, TST-positive or IGRA-positive can confirm the LTBI after excluding active TB in the subjects with organ transplants or waiting for organ transplants. The 9H regimen for LTBI treatment is recommended, but 4R or 3HR can be considered to complete the LTBI treatment before transplantation.
The followings are modifications in the 2014 Korean guidelines for TB, from the 2011 guidelines. Head and neck cancer and hematologic malignancy were added to the risk group for LTBI diagnosis and treatment. For non-contacts with infectious TB patients, the candidates for LTBI diagnosis and treatment were classified into a high risk group and moderate risk group. For LTBI treatment, the results of the DST from the index case must be considered in selecting the appropriate drug for recent TB contacts. The guidelines expressing IGRA's important role for the patients with organ transplants were added. In immune-compromised patients including TNF blocker users, LTBI can be diagnosed with either TST-positive or IGRA-positive, but cannot be excluded with TST-negative alone. In the public sector, LTBI screening for contacts to infectious TB patients are actively progressing with government-centered policy. In the private sector, clinicians are faced with the need to actively treat growing number of immune-compromised patients with LTBI, including the patients with organ transplants and those taking immunosuppressive agents. Conclusively, the 2014 Korean guidelines for TB, based on standardized LTBI diagnosis and treatment, is expected to be upgraded in the near future, supplementing the LTBI guidelines for growing foreign residents in an era of internationalization.
Figures and Tables
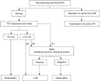 | Figure 1Latent tuberculosis infection (LTBI) diagnosis in immune-competent subjects. Appropriate method according to situations can be selected from the followings: TST alone (A), TST/IGRA two step test (B), or IGRA test alone (C)5. TB: tuberculosis; Hx: history; P.Ex: physical examination; CXR: chest radiographs. |
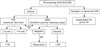 | Figure 2Latent tuberculosis infection (LTBI) diagnosis in immune-compromised subjects. Appropriate method according to situations can be tuberculin skin test (TST)/interferon-gamma release assays (IGRA) combination test (A) or IGRA test alone (B). TST-negative result alone cannot exclude LTBI, but TST-positive result alone can indicate LTBI5. TB: tuberculosis; Hx: history; P.Ex: physical examination; CXR: chest radiographs. |
Table 1
Summary for indication of LTBI treatment in Korean TB guideline5 (non-contacts to an infectious TB patient)

Table 2
Summary for the indications of LTBI treatment in Korean TB guideline5 (contacts to an infectious TB patient)

References
1. Sutherland I. Recent studies in the epidemiology of tuberculosis, based on the risk of being infected with tubercle bacilli. Adv Tuberc Res. 1976; 19:1–63.
2. Frieden TR, Sterling TR, Munsiff SS, Watt CJ, Dye C. Tuberculosis. Lancet. 2003; 362:887–899.
3. World Health Organization. Global tuberculosis report 2014. Geneva: World Health Organization;2014.
4. Joint Committee for the Development of Korean Guidelines for Tuberculosis, Korean Centers for Disease Control and Prevention. Korean guidelines for tuberculosis. Seoul: Korean Centers for Disease Control and Prevention;2011.
5. Joint Committee for the Revision of Korean Guidelines for Tuberculosis, Korean Centers for Disease Control and Prevention. Korean guidelines for tuberculosis. 2nd ed. Seoul and Cheongwon: Joint Committee for the Revision of Korean Guidelines for Tuberculosis, Korea Centers for Disease Control and Prevention;2014.
6. Hong YP, Kim SJ, Lew WJ, Lee EK, Han YC. The seventh nationwide tuberculosis prevalence survey in Korea, 1995. Int J Tuberc Lung Dis. 1998; 2:27–36.
7. Choi CM, Kang CI, Kim DH, Kim CH, Kim HJ, Lee CH, et al. The role of TST in the diagnosis of latent tuberculosis infection among military personnel in South Korea. Int J Tuberc Lung Dis. 2006; 10:1342–1346.
8. Lee JY, Choi HJ, Park IN, Hong SB, Oh YM, Lim CM, et al. Comparison of two commercial interferon-gamma assays for diagnosing Mycobacterium tuberculosis infection. Eur Respir J. 2006; 28:24–30.
9. Lee SW, Oh SY, Lee JB, Choi CM, Kim HJ. Tuberculin skin test distribution following a change in BCG vaccination policy. PLoS One. 2014; 9:e86419.
10. Targeted tuberculin testing and treatment of latent tuberculosis infection. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. This is a Joint Statement of the American Thoracic Society (ATS) and the Centers for Disease Control and Prevention (CDC). This statement was endorsed by the Council of the Infectious Diseases Society of America. (IDSA), September 1999, and the sections of this statement. Am J Respir Crit Care Med. 2000; 161(4 Pt 2):S221–S247.
11. Grzybowski S, Fishaut H, Rowe J, Brown A. Tuberculosis among patients with various radiologic abnormalities, followed by the chest clinic service. Am Rev Respir Dis. 1971; 104:605–608.
12. Diagnostic Standards and Classification of Tuberculosis in Adults and Children. This official statement of the American Thoracic Society and the Centers for Disease Control and Prevention was adopted by the ATS Board of Directors, July 1999. This statement was endorsed by the Council of the Infectious Disease Society of America, September 1999. Am J Respir Crit Care Med. 2000; 161(4 Pt 1):1376–1395.
13. Kang YA, Lee HW, Yoon HI, Cho B, Han SK, Shim YS, et al. Discrepancy between the tuberculin skin test and the whole-blood interferon gamma assay for the diagnosis of latent tuberculosis infection in an intermediate tuberculosis-burden country. JAMA. 2005; 293:2756–2761.
14. Song S, Jeon D, Kim JW, Kim YD, Kim SP, Cho JS, et al. Performance of confirmatory interferon-gamma release assays in school TB outbreaks. Chest. 2012; 141:983–988.
15. Kim HJ, Lee GH, Ryoo S, Oh SY, Lee JB, Kim JH, et al. Role of confirmatory interferon-gamma release assays in school outbreaks of tuberculosis in South Korea. Int J Tuberc Lung Dis. 2015; Forthcoming.
16. Solovic I, Sester M, Gomez-Reino JJ, Rieder HL, Ehlers S, Milburn HJ, et al. The risk of tuberculosis related to tumour necrosis factor antagonist therapies: a TBNET consensus statement. Eur Respir J. 2010; 36:1185–1206.
17. National Institute for Health and Care Excellence. Clinical guideline 33. Tuberculosis: clinical diagnosis and management of tuberculosis, and measures for its prevention and control [Internet]. London: National Institute for Health and Care Excellence;2011. cited 2014 Dec 12. Available from: http://www.nice.org.uk/CG117.
18. Metcalfe JZ, Cattamanchi A, McCulloch CE, Lew JD, Ha NP, Graviss EA. Test variability of the QuantiFERON-TB gold in-tube assay in clinical practice. Am J Respir Crit Care Med. 2013; 187:206–211.
19. Menzies D. Interpretation of repeated tuberculin tests. Boosting, conversion, and reversion. Am J Respir Crit Care Med. 1999; 159:15–21.
20. Kim SY, Park MS, Kim YS, Kim SK, Chang J, Yong D, et al. Tuberculin skin test and boosted reactions among newly employed healthcare workers: an observational study. PLoS One. 2013; 8:e64563.
21. World Health Organization. Guidance for National Tuberculosis Programmes on the Management of Tuberculosis in Children. 2nd ed. Geneva: World Health Organization;2014.
22. Canadian Thoracic Society, The Pulblic Health Agency of Canada and Licensors. Canadian tuberculosis standards. 7th ed. Ottawa: Pulblic Health Agency of Canada;2014.
23. American Academy of Pediatrics. Tuberculosis. In : Pickering LK, Baker CJ, Kimberlin DW, Long SS, editors. Red Book: 2012 Report of the Committee on Infectious Disease. Elk Grove Village, IL: American Academy of Pediatrics;2012. p. 736–759.
24. Mazurek GH, Jereb J, Vernon A, LoBue P, Goldberg S, Castro K, et al. Updated guidelines for using interferon gamma release assays to detect Mycobacterium tuberculosis infection: United States, 2010. MMWR Recomm Rep. 2010; 59:1–25.
25. National Tuberculosis Controllers Association. Centers for Disease Control and Prevention (CDC). Guidelines for the investigation of contacts of persons with infectious tuberculosis. Recommendations from the National Tuberculosis Controllers Association and CDC. MMWR Recomm Rep. 2005; 54:1–47.
26. Lobue P, Menzies D. Treatment of latent tuberculosis infection: an update. Respirology. 2010; 15:603–622.
27. Lee SH, Yim JJ, Kim HJ, Shim TS, Seo HS, Cho YS, et al. Adverse events and development of tuberculosis after 4 months of rifampicin prophylaxis in a tuberculosis outbreak. Epidemiol Infect. 2012; 140:1028–1035.
28. Yun JW, Lim SY, Suh GY, Chung MP, Kim H, Kwon OJ, et al. Diagnosis and treatment of latent tuberculosis infection in arthritis patients treated with tumor necrosis factor antagonists in Korea. J Korean Med Sci. 2007; 22:779–783.
29. Spyridis NP, Spyridis PG, Gelesme A, Sypsa V, Valianatou M, Metsou F, et al. The effectiveness of a 9-month regimen of isoniazid alone versus 3- and 4-month regimens of isoniazid plus rifampin for treatment of latent tuberculosis infection in children: results of an 11-year randomized study. Clin Infect Dis. 2007; 45:715–722.
30. Sterling TR, Villarino ME, Borisov AS, Shang N, Gordin F, Bliven-Sizemore E, et al. Three months of rifapentine and isoniazid for latent tuberculosis infection. N Engl J Med. 2011; 365:2155–2166.
31. Jung YJ, Lee JY, Jo KW, Yoo B, Lee CK, Kim YG, et al. The 'either test positive' strategy for latent tuberculous infection before anti-tumour necrosis factor treatment. Int J Tuberc Lung Dis. 2014; 18:428–434.
32. Gomez-Reino JJ, Carmona L, Angel Descalzo M. Biobadaser Group. Risk of tuberculosis in patients treated with tumor necrosis factor antagonists due to incomplete prevention of reactivation of latent infection. Arthritis Rheum. 2007; 57:756–761.
33. British Thoracic Society Standards of Care Committee. BTS recommendations for assessing risk and for managing Mycobacterium tuberculosis infection and disease in patients due to start anti-TNF-alpha treatment. Thorax. 2005; 60:800–805.
34. Mariette X, Salmon D. French guidelines for diagnosis and treating latent and active tuberculosis in patients with RA treated with TNF blockers. Ann Rheum Dis. 2003; 62:791.
35. Shim TS. Diagnosis and treatment of latent tuberculosis infection due to initiation of anti-TNF therapy. Tuberc Respir Dis. 2014; 76:261–268.
36. Kim SH, Lee SO, Park JB, Park IA, Park SJ, Yun SC, et al. A prospective longitudinal study evaluating the usefulness of a T-cell-based assay for latent tuberculosis infection in kidney transplant recipients. Am J Transplant. 2011; 11:1927–1935.
37. Kim SY, Jung GS, Kim SK, Chang J, Kim MS, Kim YS, et al. Comparison of the tuberculin skin test and interferon-gamma release assay for the diagnosis of latent tuberculosis infection before kidney transplantation. Infection. 2013; 41:103–110.
38. Kim SH, Lee SO, Park IA, Park SJ, Choi SH, Kim YS, et al. Diagnostic usefulness of a T cell-based assay for latent tuberculosis infection in kidney transplant candidates before transplantation. Transpl Infect Dis. 2010; 12:113–119.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download