Abstract
Glomus tumors of the lung are rare benign neoplasm, originating from modified smooth muscle cells. The patients are usually presented with no or non-specific symptoms such as cough, dyspnea or hemoptysis. Although surgical treatment is considered as the treatment of choice, the endobronchial therapy can be applied to the patients who are unfit for surgical excision. Herein, we describe two rare cases of glomus tumor originated at large airway (trachea and main bronchus) without respiratory symptoms and review their characteristic radiologic, macroscopic and pathological features.
Glomus tumor (GT) is uncommon neoplasm, originating from the glomus cells surrounding an arteriovenous anastomosis and constituting less than 2% of soft tissue tumors1. They are usually found in the deep dermis of the extremities, especially in the subungal regions of finger tips, however can appear extracutaneously in the gastrointestinal tract2, bone, cervix3, ovary4, mediastinum, and respiratory tract5. Rare cases with primary GT in large airway, especially in trachea, have been reported in the English and Korean literature5-7. Herein, we describe two rare cases of primary pulmonary GT that developed in trachea and main bronchus without respiratory symptom and reviewed their characteristic radiologic, macroscopic and microscopic findings.
A 64-year old man, who had no clinical respiratory symptom and previous medical problem except hypertension, visited our hospital due to incidentally found tracheal mass on the examination of health care program using computed tomography (CT). CT revealed a highly enhanced 2.6 cm-sized, iceberg shaped mid-tracheal tumor involving the right-side posterolateral wall, suggesting the impression of tracheal paraganglioma (most likely), sclerosing hemangioma and leiomyoma (Figure 1A). On the bronchoscopic examination, a 1 cm-sized, vascular mass was noted 7 cm below the vocal cord (Figure 1B). However, pulmonary function test (PFT) showed no abnormal findings: forced vital capacity (FVC), 3.6 L; forced expiratory volume in 1 second (FEV1), 3.13 L; and FEV1/FVC, 87%. Tracheal resection and anastomosis was done. He is well without evidence of local recurrence or complication two years after operation.
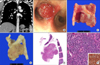
A 52-year-old women, who had no specific medical history except appendectomy and Cesarean section, was transferred due to incidentally found tracheobronchial nodule on the chest CT of health care program. Radiologically, a 1.6 cm-sized, highly enhanced nodule was found in right main bronchus near the carina suspicious of carcinoid, leiomyoma or other submucosal tumor, such as adenoid cystic carcinoma (Figure 2A). Although her PFT showed moderate obstructive pattern (FVC, 3.21 L; FEV1, 1.70 L; FEV1/FVC, 53%) and normal carbon monoxide diffusing capacity (DLco), she complained no associated respiratory symptom. Bronchoscopy revealed a sessile endobronchial mass with hyperemic appearance in carina to right main, suggesting of carcinoid tumor and squamous cell carcinoma (less likely) (Figure 2B). Resection of carina and both main bronchus with neocarina formation was done. She was well without local recurrence on 3 month of follow-up, although a bronchoscopic en bloc resection was done due to granulation tissue at the anastomosis site 2 months after operation.
Macroscopically, the mass was located at posterior membranous portion of trachea and showed erythematous mucosal surface (Figure 1C). On section, it discloses a pink, round, intraluminally protruding, well-demarcated mass showing meaty cut surface without necrosis or hemorrhage (Figure 1D). After the fixation by formalin, the mass showed similar consistency and appearance with leiomyoma of bronchus or the other organs (Figure 1E). Microscopically, it consisted of proliferation of epithelioid cells with abundant vascular channels (Figure 1F, G). The tumor cells had abundant eosinophilic or clear cytoplasm and bland oval nuclei (Figure 1H). These cells showed perivascular arrangements. No nuclear atypia, mitoses or necrosis were noted. These histologic findings were compatible with those of carcinoid tumor, hemangiopericytoma and GT. Immunohistochemically, the tumor cells showed positive finding for smooth muscle actin (1:1,000, mouse monoclonal, Dako, Glostrup, Denmark), confirming GT (Figure 1H, inset).
A mass was located at left main bronchus involving carina with intraluminally protruding appearance and erythematous mucosal surface. On section, it disclosed a pink, round, well demarcated mass showing flesh cut surface without necrosis or hemorrhage (Figure 2C). After the fixation by formalin, the mass showed gray solid cut surface with rubbery consistency (Figure 2D). Microscopically, the mass was well circumscribed and consisted of relatively uniform epithelioid cells with bland looking hyperchromatic nuclei (Figure 2E, F). The formation of vascular channels was less than case 1. For differential diagnosis with carcinoid, immunohistochemical stains with smooth muscle actin (1:1,000, mouse monoclonal, Dako), chromogranin (1:400, DAK-A3, Dako) and CD56 (1:50, 1B6, Novocastra, Newcastle upon Tyne, UK) were done, and the tumor cells showed positivity for only smooth muscle actin (Figure 2F, inset).
GT arising in large airway is very rare neoplasm consisting of several variant; solid GT (most common), glomangioma, glomangiomyoma, glomangiomatosis, symplastic GT and malignant GT (glomangiosarcoma), and most GTs follow a benign course except the case of glomangiosarcoma. Although the surgical treatment is considered as the treatment of choice, an endobronchial therapy can be applied to the patients unfit for surgical excision.
Many patients with respiratory GT showed no respiratory symptom5, as Ariizumi et al.6 reviewed that 42.4% of previously reported patients with respiratory GT showed no respiratory symptoms. However, most patients with GT in large airway (11/12 patients) showed respiratory symptom such as hemoptysis, hemosputum and cough. Our two cases were from large airway with no clinically symptom, incidentally found. It is notable that the patient of case 2 had not complained asthma-like symptom, although the polypoid mass partly occupied luminal space and the PFT showed moderate obstructive pattern. Therefore, it is supposed that considerately growing mass induce remodeling of pulmonary vasculature and delay an asthma-like symptom8 or that mass in left main bronchus just distal from carina could be insufficient to make associated symptom.
The differential diagnoses for large airway include squamous cell carcinoma, adenoid cystic carcinoma, carcinoid, and other tumors (squamous cell papilloma, mucoepidermoid carcinoma, hamartoma, lipoma, and leiomyoma)9,10. Although squamous cell carcinoma can be discriminated from GT bronchoscopically, that former shows an irregular surface with or without ill-defined margin and necrosis, the discrimination of tumors showing smooth luminal surface (like carcinoid, adenoid cystic carcinoma, mucoepidermoid carcinoma, lipoma, and hamartoma) can be helped by radiologic examination. Chest CT is the modality of choice on GT of pulmonary origin, even though GTs of other sites can be examined by various radiologic modalities such as ultrasonography, CT and magnetic resonance image. GTs of lung typically show marked contrast enhancements due to their rich vasculature11,12. As the same reason, they show hyperemic intraluminal surface bronchoscopically.
Grossly, the masses show meaty or flesh solid surface depending on the cellularity and vascularity, which is similar with those of leiomyoma. Microscopic findings also include many differential diagnoses such as carcinoid, myopericytoma, epithelioid leiomyoma and paraganglioma. In addition to similar cytomorphological features of tumor cells in both carcinoid and GT, proliferation of vascular channel and perivascular arrangement of tumor cells can mimic the trabecular arrangement of carcinoid. Solid and somewhat zellballen-like arrangement with less vasculature might remind paraganglioma as differential diagnosis. However, both carcinoid and paraganglioma show positivity for the neuroendocrine markers, such as, chomogranin, CD56 and neuron-specific enolase. Occasionally, irregularly-branched vascular proliferation of GT needs for differential diagnosis with myopericytoma (previous hemangiopericytoma). However, hemangiopericytoma is negative for smooth muscle actin, but diffuse positive staining for CD34. Although most smooth muscle neoplasm comprises spindle cells with a fascicular pattern, epithelioid leiomyoma can show similar histologic finding with GT and also express smooth muscle actin. However, epithelioid leiomyoma show more cellular appearance with less vascularity than GT and express other smooth muscle markers including desmin and caldesmon.
In summary, we here report two characteristic cases of rare primary GT arising in the large airway with no associated symptom and reviewed their radiologic or bronchoscopic findings and histologic differential diagnoses. Preoperative suspicion, careful morphologic observation and immunohistochemical staining with appropriate markers should enable to make the diagnosis.
Figures and Tables
Figure 1
(A) Computed tomography reveals a highly enhanced 2.6 cm-sized, iceberg shaped tracheal tumor involving the right-side posterolateral. (B) Bronchoscopy reveals a 1 cm-sized mass with erythematous mucosal surface 7 cm below the vocal cord. (C) Grossly, the mass is internally protruded and has a reddish mucosal surface. (D) On section, it discloses a pinkish, meaty and solid cut surface without necrosis or hemorrhage. (E) After the fixation by formalin, the mass shows similar consistency and appearance with leiomyoma. (F) At low power view, the mass is well circumscribed and has abundant vasculature (H&E stain, 1:1 scan view). (G) It consists of proliferation of epithelioid cells showing perivascular arrangements with abundant vascular channels (H&E stain, ×200). (H) The tumor cells have abundant eosinophilic or clear cytoplasm and bland oval nuclei and shows positivity for smooth muscle actin (SMA) (H&E stain, ×400; inset: SMA, ×400).
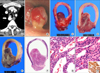
Figure 2
(A) The 1.6 cm-sized highly enhanced nodule is noted in right main bronchus near carina. (B) Bronchoscopy reveals a sessile endobronchial mass with hyperemic surface near the carina. (C) Grossly, the mass is intraluminal polypoid growing and has flesh cut surface. (D) After the fixation by formalin, the mass shows gray solid cut surface with rubbery consistency. (E) At low power view, it is a well circumscribed, and ovoid mass. (F) The tumor cells consist of relatively uniformed epithelioid cells with bland-looking hyperchromatic nuclei and shows positivity for smooth muscle actin (inset) (F, ×400).
References
1. Shugart RR, Soule EH, Johnson EW Jr. Glomus Tumor. Surg Gynecol Obstet. 1963; 117:334–340.
2. Miettinen M, Paal E, Lasota J, Sobin LH. Gastrointestinal glomus tumors: a clinicopathologic, immunohistochemical, and molecular genetic study of 32 cases. Am J Surg Pathol. 2002; 26:301–311.
3. Albores-Saavedra J, Gilcrease M. Glomus tumor of the uterine cervix. Int J Gynecol Pathol. 1999; 18:69–72.
4. Gokten N, Peterdy G, Philpott T, Maluf HM. Glomus tumor of the ovary: report of a case with immunohistochemical and ultrastructural observations. Int J Gynecol Pathol. 2001; 20:390–394.
5. Kim MJ, Sung WJ. Primary pulmonary glomus tumor, diagnosed by preoperative needle biopsy: report of one case and literature review. Korean J Pathol. 2008; 42:37–40.
6. Ariizumi Y, Koizumi H, Hoshikawa M, Shinmyo T, Ando K, Mochizuki A, et al. A primary pulmonary glomus tumor: a case report and review of the literature. Case Rep Pathol. 2012; 2012:782304.
7. Lee EW, Kim SO, Oh IJ, Ju JY, Cho GJ, Kim KS, et al. A case of bronchial glomus tumor. Tuberc Respir Dis. 2002; 53:445–449.
8. Jeffery PK. Remodeling in asthma and chronic obstructive lung disease. Am J Respir Crit Care Med. 2001; 164(10 Pt 2):S28–S38.
9. Ko JM, Jung JI, Park SH, Lee KY, Chung MH, Ahn MI, et al. Benign tumors of the tracheobronchial tree: CT-pathologic correlation. AJR Am J Roentgenol. 2006; 186:1304–1313.
10. Grillo HC, Mathisen DJ. Primary tracheal tumors: treatment and results. Ann Thorac Surg. 1990; 49:69–77.
11. Akata S, Yoshimura M, Park J, Okada S, Maehara S, Usuda J, et al. Glomus tumor of the left main bronchus. Lung Cancer. 2008; 60:132–135.
12. Glazebrook KN, Laundre BJ, Schiefer TK, Inwards CY. Imaging features of glomus tumors. Skeletal Radiol. 2011; 40:855–862.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download