Abstract
Background
Asian dust storms can be transported across eastern Asia. In vitro, Asian dust particle-induced inflammation and enhancement of the allergic reaction have been observed. However, the fibrotic effects of Asian dust particles are not clear. Production of transforming growth factor β1 (TGF-β1) and fibronectin were investigated in the bronchial epithelial cells after exposure to Asian dust particulate matter (AD-PM10).
Methods
During Asian dust storm periods, air samples were collected. The bronchial epithelial cells were exposed to AD-PM10 with and without the antioxidant, N-acetyl-L-cysteine (NAC). Then TGF-β1 and fibronectin were detected by Western blotting. The reactive oxygen species (ROS) was detected by the measurement of dicholorodihydrofluorescin (DCF), using a FACScan, and visualized by a confocal microscopy.
Fibrosis is an important process in the development of several pulmonary diseases. Airway remodeling in asthma and chronic obstructive pulmonary disease (COPD) is the result of a fibrotic reaction surrounding the airways of the lung, such as basement membrane thickening and peribronchiolar fibrosis1. The etiologic factors of COPD are known to include not only cigarette smoke, but also pollutants1. Furthermore, idiopathic pulmonary fibrosis (IPF) is a fatal lung disease with irreversible fibrosis2. Although the diagnosis of IPF requires 'exclusion of other known causes of interstitial lung diseases, such as drug environmental exposure, medication or systemic disease,' there are several pieces of evidence that environmental agents may have an etiologic role in pulmonary fibrosis2. Inhaled environmental agents, such as smoke, dust, and fumes, have been proposed to be involved in the development of pulmonary fibrosis3. Some epidemilogic studies showed that metal exposure and wood dust exposure are increased in patients with IPF4. Other studies have shown that exposure to metal dust, such as cobalt, aluminum, zinc, cadmium, and mercury are associated with the development of pulmonary fibrosis5.
Air pollutants have been recognized as a major problem for human health. Many epidemiologic studies have correlated episodes of elevated PM10 levels with increased mortality and morbidity6-8. Air pollution is associated with a variety of cellular toxicities, including inflammation, DNA damage, and fibrosis8. Exposure to air pollution particulates has been associated with airway fibrosis other than fibrotic reactions of the lung parenchyma9. Asian dust is the long-range transport of atmospheric pollutants during dust events in eastern Asia, which originate in the Chinese and Mongolian deserts during spring season10. Recently, there have been possible adverse effects of these dust events, as the dust storms pass through industrialized areas, such as northeastern China, which increases the probability that Asian dust contains combustion-source particles10. Asian dust storm events are associated with an increase in daily mortality in Seoul, Korea, and Taipei, Taiwan11,12. The dust also causes aggravation of respiratory symptoms of patients with airway diseases, neutrophilic airway inflammation in mice, and allergic reactions13-16. There are several evidences that Asian sand particles could enhance inflammation by respiratory pathogens17,18. However, there are no investigations regarding the fibrotic effect of the Asian sand particles. The present study was designed to investigate whether Asian dust particles induce production of transforming growth factor-β1 (TGF-β1) and fibronectin through oxidative pathways in bronchial epithelial cells.
Air samples were collected over the course of 2 years between 2004 and 2005 in Incheon City of South Korea. Asian dust particles (AD-PM10) were sampled using a high volume air sampler (HV500F; Sibata, Tokyo, Japan) with airflow at 500 L/min for at least 5 hours. For sample collection, glass microfiber filters (Millipore, Bedford, MA, USA) with a pore size of 0.25 µm were used. Filters were stored at 4℃ until use. Particles were suspended in phosphate buffered saline (PBS) and sonicated the particles for 3 minutes at maximal watt, after which the materials were sieved through filters, 10 µm in size (Mitex membrane filters; Millipore). All AD-PM10 suspensions were sterilized for removal of microorganisms. Endotoxin was measured by a Limulus amebocyte lysate assay kit (BioWhittaker, Walkersville, MD, USA), according to the manufacturer's specifications. Endotoxin levels in AD-PM10 samples analyzed within 24 hours of collection gave results similar to those obtained in stored samples and after sterilization.
Chemical composition of AD-PM10 was analyzed at the Korea Institute of Ceramic Engineering and Technology. AD-PM10 contained 48% SiO2, 12% Al2O3, 5% Fe2O3, and 1% TiO2 (Figure 1).
Normal bronchial epithelial cells (WI-26VA4 cell line; KCLB, Seoul, Korea) were cultured in Dubelcco's modified Eagle medium ((DMEM; Gibco BRL, Gaithersburg, MD, USA) containing 10% fetal bovine serum (FBS). Cells were incubated in 5% CO2 at 37°. Cells were cultured in 6-well plates and allowed to grow to confluence. Near-confluent cells were incubated with DMEM containing 0.5% FBS for 24 hours. After this time period, cells were treated with prepared AD-PM10. Cells were incubated with each concentration (10, 50, 100, 250, and 500 µg/mL) of suspension for 24 hours. In some experiments, cells were pretreated for 1 hour with the antioxidant, 5 mM N-acetyl-L-cysteine (NAC; Sigma chemical Co., St. Louis, MO, USA).
Cells were washed with PBS and suspended in a lysis buffer (PRO-PREP protein extraction solution; Intron Biotechnology, Inc., Seoul, Korea). The harvested cells were incubated at -20℃ and the lysates were clarified by centrifugation at 13,000 g for 10 minutes. After protein concentration determination of cell lysates with the Bradford assay, 30 g of protein of each lysate was mixed with sample buffer. Samples were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, and transferred to a polyvinylidene difluoride Western blotting membrane (Roche, Indianapolis, IN, USA). Immunoblot sequential incubation with primary antibody overnight at 4℃ and secondary antibody (anti-rabbit IgG; Amersham Biosciences, Little Chalfort, UK) was added for 1 hour. The membranes were incubated with enhanced chemiluminescence detection reagents (WEST-SOL plus; Intron Biotechnology, Inc.). Membranes were probed with antibodies for actin as a loading control. Primary antibodies were obtained from Santa Cruz Biotechnology (TGF-β1, fibronectin, and actin; Santa Cruz, CA, USA). Immunoreactive bands were quantified densitometrically and compared to the controls.
Bronchial epithelial cells in the presence or absence of AD-PM10 grown on a cover glass were incubated in serum-free media containing 5-(and-6)-choloromethyl-2',7'-dicholorodihydrofluorescin diacetate (CM-H2DCFDA; Molecular Probes Inc., Eugene, OR, USA) for 20 minutes at a final concentration of 5.0 µmol/L. After washing twice with PBS, cells were placed on a glass slide. Images were collected with a confocal laser scanning microscope (IX81; Olympus, Tokyo, Japan). The generation of ROS was evaluated as the fluorescence intensity of dicholorodihydrofluorescin (DCF).
The cell-permeable fluorogenic probe, 2',7'-dichlordihydrofluorescin diacetate (DCF-DA), was used to detect oxidative stress in bronchial epithelial cells. To analyze the intracellular ROS by FACScan flow cytometer, AD-PM10-exposed cells were washed twice with PBS and incubated in serum-free media containing 5 mM 5-(and-6)-choloromethyl-2',7'-dicholorodihydrofluorescin diacetate (CM-H2DCFDA; Molecular Probes Inc.) for 20 minutes at 37℃ in a CO2 incubator. The intracellular fluorescence intensity was measured in the cells by using a flow cytometer (FACSFlow; BD Biosciences, San Jose, CA, USA). For each analysis, 20,000 events were recorded.
All values are expressed as the mean±standard error. Significant differences between the multiple groups were analyzed using one-way ANOVA or the non-parametric Kruskal-Wallis test. The post hoc analysis was applied using the Scheffe test. Differences between the two groups were compared by a non-parametric two-independent sample Mann-Whitney U test. All statistical analyses were performed with SPSS ver. 12.0 (SPSS Inc., Chicago, IL, USA). A value of p<0.05 was accepted as statistically significant and 0.05≤p<0.1 indicated a trend.
Cell exposure to AD-PM10 induced the time-dependent production of TGF-β1. TGF-β1 was increased over the first 48 hours and peaked at 96 hours (Figure 2A). To study the dose dependence of TGF-β1 production, WI-26VA4 cells were treated with increasing concentrations of AD-PM10 at 48 hours. Maximum production of TGF-β1 occurred at 50 µg/mL of AD-PM10. The levels of TGF-β1 production with 50 and 100 µg/mL of AD-PM10 were significantly higher compared with control values. The production of fibronectin was enhanced over the first 24 hours and peaked at 96 hours (Figure 2B). To study the concentration dependence of fibronectin production, cells were incubated with increasing doses of AD-PM10 at 48 hours. The peak production of fibronectin occurred at 100 µg/mL of AD-PM10. The levels of fibronectin production with 10, 100, and 500 µg/mL of AD-PM10 were significantly higher compared to control values.
Figure 3 shows DCF-sensitive ROS production in normal bronchial epithelial cells incubated with 0, 10, 50, 100, 250, and 500 µg/mL of a AD-PM10 suspension. The DCF-sensitive ROS in AD-PM10-exposed bronchial epithelial cells showed a significantly higher expression compared with controls (Figure 3B). The level of ROS production was peak at 50 µg/mL of AD-PM10. Concentration-dependent, significant differences did not occur between cells exposed to variable concentrations of AD-PM10. We demonstrated that the antioxidant, NAC, attenuates the expression of intracellular ROS. As another method, intracellular ROS expression was examined using confocal microscopy (Figure 3A). The confocal microscopic findings showed a high expression of intracellular ROS in AD-PM10-exposed bronchial epithelial cells.
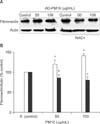
Figure 4 presents the expression of TGF-β1 using Western blotting by normal bronchial epithelial cells incubated with medium alone (control); and 10, 50, 100, 250, and 500 µg/mL of AD-PM10 suspension. The levels of TGF-β1 production with 50 and 100 µg/mL of AD-PM10 were significantly higher compared with control values. Also, the expression of TGF-β1 in AD-PM10-exposed cells was inhibited by the antioxidant, NAC. Figure 4 shows that NAC effectively blocked the expression of TGF-β1 in bronchial epithelial cells with the AD-PM10 suspension. The Western blotting for the analysis of fibronectin expression was performed in the bronchial epithelial cells incubated with 0 (medium alone, control), 50, and 100 µg/mL of AD-PM10 for 24 hours (Figure 5). As the result of densitometric analysis, the values of fibronectin production was stronger in the cells exposed to AD-PM10 than control (Figure 4B). Otherwise NAC downregulated the expression of fibronection exposed to AD-PM10.
The results showed that AD-PM10 collected during Asian dust periods induced the production of TGF-β1, and fibronectin in normal bronchial epithelial cells. This increase in the protein production of TGF-β1, and fibronectin was induced by an intracellular ROS.
Particulate matter is a mixture of inorganic and organic components that vary in size, origin, and composition19. Particle size and composition in particular influence cellular toxicity. PM10, with an aerodynamic diameter <10 µm, is known to induce adverse respiratory effects19. Asian dust particles in the size range of 1~10 µm are deposited in areas as far away as Korea over a distance of 1,500~2,000 km10. We prepared Asian dust particles to PM10 using filters, 10 µm in size. Much of the information on the component-specific effects of PM comes from recently conducted in vitro and in vivo toxicologic studies using different compositions, such as residual oil fly ashes20, metals21, diesel22, and ambient PM from different sources23. The exposure to variable dusts, including asbestos, coal, silica, silicates, iron dioxide, aluminum oxide, and mineral dusts, induce fibrosis of the small airways21. In the present study, AD-PM10 have fibrotic potentials in the bronchial epithelial cells. We analyzed the chemical composition of AD-PM10 used in this study. The results demonstrated that the main components of AD-PM10 are SiO2, and metals such as Al2O3, Fe2O3, and TiO2. These components are known as fibrogenic dusts. We demonstrated that the fibrogenic mediator, TGF-β1, and fibronectin, one of the matrix components, were induced by AD-PM10. TGF-β is a key regulator of both normal wound repair and the aberrant repair mechanisms characteristic of many fibrotic diseases, including pulmonary fibrosis24. Furthermore, TGF-β is a mediator of matrix, particularly collagen and fibronectin. Several components of particles have been known to the effect with fibrosis, particularly in the small airway. Gursinsky et al.20 reported that fly ash particles induce TGF-β1 and collagen 1 in primary fibroblasts and Dai et al.21 showed that mineral dusts produce fibrogenic mediators, such as TGF-β1 and platelet-derived growth factor (PDGF)-A in rat tracheal explants. There was a study that urban ambient particles upregulate the PDGF receptor system in lung myofibroblasts25. Our results show that Asian dust particles might have fibrotic effects in the bronchial epithelial cells.
According to results of this study, ROS increased in bronchial epithelial cells with AD-PM10 compared to non-exposed controls and the induction of TGF-β1 in the bronchial epithelial cells with AD-PM10 was inhibited by the antioxidant, NAC, suggesting that intracellular ROS mediates AD-PM10-induced TGF-β1 production. The respiratory system, including the airway and lungs, is exposed to higher environmental oxidants than other tissues26. Exogenous oxidants and pollutants, such as cigarette smoke, asbestos fibers, and other particles, increase oxidant production27. Several studies in cellular and animal models suggest a variety of possible mechanisms, including effects of particle components on the intracellular sources of ROS, indirect effects due to pro-inflammatory mediators released from PM-stimulated macrophages, and neural stimulation after particle deposition in the lungs28. There are several pieces of evidence that oxidative stress may be involved in the mechanism of disease initiation and progression following exposure to particular particles, such as silica, asbestos, and mineral dusts27,28. Oxidative stress also was proposed as an important mechanism of COPD and pulmonary fibrosis26,27. Airway diseases, such as bronchial asthma and COPD, have evidence of increased oxidative stress27. Recently, there are studies that patients with IPF have higher levels of oxidant stress than control patients, and antioxidant NAC have a protective effect on the progression of IPF26,29. There are several potential interactions between TGF-β and oxidants/antioxidants in the lung. TGF-β differentiated myofibroblasts can themselves serve as a source of oxidant production30. Furthermore, in vitro studies have shown that ROS increase the release of TGF-β from pulmonary epithelial cells31. The peak reaction on the induction of ROS, TGF-β1 revealed moderate concentration of AD-PM10 such as 50 or 100 µg/mL. We measured cell viability via MTT assay, high concentration of AD-PM10, 250 and 500 µg/mL, showed significant decrease of viability (data not shown). As these results, high concentration of AD-PM10 decreases cell viability, and then may attenuates ability to produce inctracellular ROS. However, the peak production of fibronectin by AD-PM10 was different with the peak ROS production. It suggests that other mechanism may be associated with AD-PM10 induced fibronectin production.
We conclude that AD-PM10 might have fibrotic potential which is mediated by ROS. Further studies are needed that which components of AD-PM10 induce fibrotic potential.
Figures and Tables
Figure 1
The proportion of chemical components in AD-PM10 used in this study. This was analyzed at the Korea Institute of Ceramic Engineering and Technology.
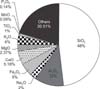
Figure 2
Time-dependent and dose-dependent effects of AD-PM10 on transforming growth factor β1 (TGF-β1) and fibronectin production. WI-26VA4 cells were incubated for the indicated period of time with 100 µg/mL of AD-PM10 (A). Alternatively, these cells were incubated for 48 hours with the indicated concentrations of AD-PM10 (B). The levels of TGF-β1 production with 50 and 100 µg/mL of AD-PM10 were significantly higher compared with control values (black squares). The levels of fibronectin production with AD-PM10 were significantly higher compared with the control values (white circle). In the dose-response experiments of AD-PM10, the means±SD of five separate experiments are shown. *Significant increase over the non-stimulated control cells.

Figure 3
Dicholorodihydrofluorescin (DCF)-sensitive reactive oxygen species (ROS) production in bronchial epithelial cells incubated with 0~500 µg/mL of a AD-PM10 suspension. The confocal microscopic examination shows a high expression of intracellular ROS in AD-PM10-exposed bronchial epithelial cells (A). On measuring the levels of DCF-sensitive ROS using FACScan, ROS in AD-PM10-exposed bronchial epithelial cells showed significantly higher expression compared with controls (black squares) and N-acetyl-L-cysteine (NAC) attenuated the increase of ROS generation (white squares) (B). The experiments were repeated over five times. *Significant increase over unstimulated control cells. †Significant differences between NAC-treated cells and cells not treated with NAC.
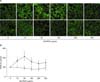
Figure 4
Effect of NAC on AD-PM10-induced transforming growth factor β1 (TGF-β1). On Western blotting, TGF-β1 with exposure to AD-PM10 increased more than the unexposed control, and N-acetyl-L-cysteine (NAC) attenuated TGF-β1 induction with AD-PM10 (A). The levels of TGF-β1 production with 50 and 100 µg/mL of AD-PM10 were significantly higher compared with control values (white bar). NAC effectively blocked the expression of TGF-β1 (black bar) (B). The experiments were repeated over five times. *A significant increase over unstimulated control cells. †,‡Significant difference between cells treated with NAC and cells not treated with NAC (†p<0.05, ‡0.05<p<0.1).
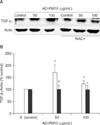
Figure 5
Effect of N-acetyl-L-cysteine (NAC) on AD-PM10 induced fibronectin. On Western blotting, fibronectin with exposure to AD-PM10 increased greater than the unexposed control, and NAC attenuated fibronctin production with AD-PM10 (A). The levels of fibronectin production with 50 and 100 µg/mL of AD-PM10 were significantly higher compared with control values (white bar). And NAC effectively blocked the expression of fibronectin (black bar) (B). The experiments were repeated over five times.
References
1. Wright JL, Postma DS, Kerstjens HA, Timens W, Whittaker P, Churg A. Airway remodeling in the smoke exposed guinea pig model. Inhal Toxicol. 2007. 19:915–923.
2. Raghu G, Collard HR, Egan JJ, Martinez FJ, Behr J, Brown KK, et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011. 183:788–824.
3. Nemery B, Bast A, Behr J, Borm PJ, Bourke SJ, Camus PH, et al. Interstitial lung disease induced by exogenous agents: factors governing susceptibility. Eur Respir J Suppl. 2001. 32:30s–42s.
4. Hubbard R, Cooper M, Antoniak M, Venn A, Khan S, Johnston I, et al. Risk of cryptogenic fibrosing alveolitis in metal workers. Lancet. 2000. 355:466–467.
5. Taskar VS, Coultas DB. Is idiopathic pulmonary fibrosis an environmental disease? Proc Am Thorac Soc. 2006. 3:293–298.
6. Dockery DW, Pope CA 3rd, Xu X, Spengler JD, Ware JH, Fay ME, et al. An association between air pollution and mortality in six U.S. cities. N Engl J Med. 1993. 329:1753–1759.
7. Schwartz J. Short term fluctuations in air pollution and hospital admissions of the elderly for respiratory disease. Thorax. 1995. 50:531–538.
8. Foster WM, Costa DL. Air pollutants and the respiratory tract. 2005. 2nd ed. New York: Taylor & Francis.
9. Bonner JC, Rice AB, Moomaw CR, Morgan DL. Airway fibrosis in rats induced by vanadium pentoxide. Am J Physiol Lung Cell Mol Physiol. 2000. 278:L209–L216.
10. Park JW, Lim YH, Kyung SY, An CH, Lee SP, Jeong SH, et al. Effects of ambient particulate matter on peak expiratory flow rates and respiratory symptoms of asthmatics during Asian dust periods in Korea. Respirology. 2005. 10:470–476.
11. Kwon HJ, Cho SH, Chun Y, Lagarde F, Pershagen G. Effects of the Asian dust events on daily mortality in Seoul, Korea. Environ Res. 2002. 90:1–5.
12. Chen YS, Sheen PC, Chen ER, Liu YK, Wu TN, Yang CY. Effects of Asian dust storm events on daily mortality in Taipei, Taiwan. Environ Res. 2004. 95:151–155.
13. Lei YC, Chan CC, Wang PY, Lee CT, Cheng TJ. Effects of Asian dust event particles on inflammation markers in peripheral blood and bronchoalveolar lavage in pulmonary hypertensive rats. Environ Res. 2004. 95:71–76.
14. Hiyoshi K, Ichinose T, Sadakane K, Takano H, Nishikawa M, Mori I, et al. Asian sand dust enhances ovalbumin-induced eosinophil recruitment in the alveoli and airway of mice. Environ Res. 2005. 99:361–368.
15. Ichinose T, Sadakane K, Takano H, Yanagisawa R, Nishikawa M, Mori I, et al. Enhancement of mite allergen-induced eosinophil infiltration in the murine airway and local cytokine/chemokine expression by Asian sand dust. J Toxicol Environ Health A. 2006. 69:1571–1585.
16. Ichinose T, Yoshida S, Sadakane K, Takano H, Yanagisawa R, Inoue K, et al. Effects of asian sand dust, Arizona sand dust, amorphous silica and aluminum oxide on allergic inflammation in the murine lung. Inhal Toxicol. 2008. 20:685–694.
17. He M, Ichinose T, Yoshida S, Yamamoto S, Inoue K, Takano H, et al. Asian sand dust enhances murine lung inflammation caused by Klebsiella pneumoniae. Toxicol Appl Pharmacol. 2012. 258:237–247.
18. Yeo NK, Hwang YJ, Kim ST, Kwon HJ, Jang YJ. Asian sand dust enhances rhinovirus-induced cytokine secretion and viral replication in human nasal epithelial cells. Inhal Toxicol. 2010. 22:1038–1045.
19. Fujii T, Hayashi S, Hogg JC, Vincent R, Van Eeden SF. Particulate matter induces cytokine expression in human bronchial epithelial cells. Am J Respir Cell Mol Biol. 2001. 25:265–271.
20. Gursinsky T, Ruhs S, Friess U, Diabaté S, Krug HF, Silber RE, et al. Air pollution-associated fly ash particles induce fibrotic mechanisms in primary fibroblasts. Biol Chem. 2006. 387:1411–1420.
21. Dai J, Gilks B, Price K, Churg A. Mineral dusts directly induce epithelial and interstitial fibrogenic mediators and matrix components in the airway wall. Am J Respir Crit Care Med. 1998. 158:1907–1913.
22. Hashimoto S, Gon Y, Takeshita I, Matsumoto K, Jibiki I, Takizawa H, et al. Diesel exhaust particles activate p38 MAP kinase to produce interleukin 8 and RANTES by human bronchial epithelial cells and N-acetylcysteine attenuates p38 MAP kinase activation. Am J Respir Crit Care Med. 2000. 161:280–285.
23. Saldiva PH, Clarke RW, Coull BA, Stearns RC, Lawrence J, Murthy GG, et al. Lung inflammation induced by concentrated ambient air particles is related to particle composition. Am J Respir Crit Care Med. 2002. 165:1610–1617.
24. Thannickal VJ, Lee DY, White ES, Cui Z, Larios JM, Chacon R, et al. Myofibroblast differentiation by transforming growth factor-beta1 is dependent on cell adhesion and integrin signaling via focal adhesion kinase. J Biol Chem. 2003. 278:12384–12389.
25. Bonner JC, Rice AB, Lindroos PM, O'Brien PO, Dreher KL, Rosas I, et al. Induction of the lung myofibroblast PDGF receptor system by urban ambient particles from Mexico City. Am J Respir Cell Mol Biol. 1998. 19:672–680.
26. Kinnula VL, Fattman CL, Tan RJ, Oury TD. Oxidative stress in pulmonary fibrosis: a possible role for redox modulatory therapy. Am J Respir Crit Care Med. 2005. 172:417–422.
27. Bowler RP, Crapo JD. Oxidative stress in airways: is there a role for extracellular superoxide dismutase? Am J Respir Crit Care Med. 2002. 166(12 Pt 2):S38–S43.
28. Gonzalez-Flecha B. Oxidant mechanisms in response to ambient air particles. Mol Aspects Med. 2004. 25:169–182.
29. Demedts M, Behr J, Buhl R, Costabel U, Dekhuijzen R, Jansen HM, et al. High-dose acetylcysteine in idiopathic pulmonary fibrosis. N Engl J Med. 2005. 353:2229–2242.
30. Thannickal VJ, Fanburg BL. Activation of an H2O2-generating NADH oxidase in human lung fibroblasts by transforming growth factor beta 1. J Biol Chem. 1995. 270:30334–30338.
31. Bellocq A, Azoulay E, Marullo S, Flahault A, Fouqueray B, Philippe C, et al. Reactive oxygen and nitrogen intermediates increase transforming growth factor-beta1 release from human epithelial alveolar cells through two different mechanisms. Am J Respir Cell Mol Biol. 1999. 21:128–136.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download