Abstract
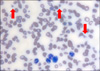
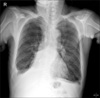
Hemolytic uremic syndrome (HUS) is a rare disorder characterized by the triad of microangiopathic hemolytic anemia, thrombocytopenia, and acute renal failure. HUS arises from a wide spectrum of conditions, and chemotherapeutic agents have been reported to be associated with HUS, including Mitomycin, Cisplatin, Bleomycin, and Gemcitabine. A 76-year-old man treated with Gemcitabine due to non-small cell lung cancer developed clinical and laboratory findings compatible with HUS. Gemcitabine was ceased and hemodialysis and plasma exchange were utilized and he recovered. A high level of suspicion for HUS is necessary when cancer patients are treated with Gemcitabine, and prompt recognition and treatment are also essential.
Figures and Tables
References
1. Moake JL. Thrombotic microangiopathies. N Engl J Med. 2002. 347:589–600.
2. Blackall DP, Marques MB. Hemolytic uremic syndrome revisited: Shiga toxin, factor H, and fibrin generation. Am J Clin Pathol. 2004. 121:Suppl. S81–S88.
3. Loirat C, Frémeaux-Bacchi V. Atypical hemolytic uremic syndrome. Orphanet J Rare Dis. 2011. 6:60.
4. Gore EM, Jones BS, Marques MB. Is therapeutic plasma exchange indicated for patients with gemcitabine-induced hemolytic uremic syndrome? J Clin Apher. 2009. 24:209–214.
5. Fung MC, Storniolo AM, Nguyen B, Arning M, Brookfield W, Vigil J. A review of hemolytic uremic syndrome in patients treated with gemcitabine therapy. Cancer. 1999. 85:2023–2032.
6. Saif MW, Xyla V, Makrilia N, Bliziotis I, Syrigos K. Thrombotic microangiopathy associated with gemcitabine: rare but real. Expert Opin Drug Saf. 2009. 8:257–260.
7. Zupancic M, Shah PC, Shah-Khan F. Gemcitabine-associated thrombotic thrombocytopenic purpura. Lancet Oncol. 2007. 8:634–641.
8. Ruggenenti P, Noris M, Remuzzi G. Thrombotic microangiopathy, hemolytic uremic syndrome, and thrombotic thrombocytopenic purpura. Kidney Int. 2001. 60:831–846.
9. Flombaum CD, Mouradian JA, Casper ES, Erlandson RA, Benedetti F. Thrombotic microangiopathy as a complication of long-term therapy with gemcitabine. Am J Kidney Dis. 1999. 33:555–562.
10. Saif MW, McGee PJ. Hemolytic-uremic syndrome associated with gemcitabine: a case report and review of literature. JOP. 2005. 6:369–374.
11. Duperray A, Tranqui L, Alix JL, Cordonnier D. Effect of mitomycin C on prostacyclin synthesis by human endothelial cells. Biochem Pharmacol. 1988. 37:4753–4757.
12. Licciardello JT, Moake JL, Rudy CK, Karp DD, Hong WK. Elevated plasma von Willebrand factor levels and arterial occlusive complications associated with cisplatin-based chemotherapy. Oncology. 1985. 42:296–300.
13. Porta C, Danova M, Riccardi A, Bobbio-Pallavicini E, Ascari E. Cancer chemotherapy-related thrombotic thrombocytopenic purpura: biological evidence of increased nitric oxide production. Mayo Clin Proc. 1999. 74:570–574.
14. Caprioli J, Noris M, Brioschi S, Pianetti G, Castelletti F, Bettinaglio P, et al. Genetics of HUS: the impact of MCP, CFH, and IF mutations on clinical presentation, response to treatment, and outcome. Blood. 2006. 108:1267–1279.
15. Walker RW, Rosenblum MK, Kempin SJ, Christian MC. Carboplatin-associated thrombotic microangiopathic hemolytic anemia. Cancer. 1989. 64:1017–1020.
16. Werner TL, Agarwal N, Carney HM, Rodgers GM. Management of cancer-associated thrombotic microangiopathy: what is the right approach? Am J Hematol. 2007. 82:295–298.
17. Oberic L, Buffet M, Schwarzinger M, Veyradier A, Clabault K, Malot S, et al. Cancer awareness in atypical thrombotic microangiopathies. Oncologist. 2009. 14:769–779.
18. Tsai HM. Advances in the pathogenesis, diagnosis, and treatment of thrombotic thrombocytopenic purpura. J Am Soc Nephrol. 2003. 14:1072–1081.
19. Fakhouri F, Frémeaux-Bacchi V. Does hemolytic uremic syndrome differ from thrombotic thrombocytopenic purpura? Nat Clin Pract Nephrol. 2007. 3:679–687.
20. Bharthuar A, Egloff L, Becker J, George M, Lohr JW, Deeb G, et al. Rituximab-based therapy for gemcitabine-induced hemolytic uremic syndrome in a patient with metastatic pancreatic adenocarcinoma: a case report. Cancer Chemother Pharmacol. 2009. 64:177–181.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download