Abstract
Bilateral interstitial infiltration in chest radiography, which may be fine granular, reticular or of ground glass opacity, is the typical radiographic findings of Pneumocystis jiroveci pneumonia. Recently, atypical radiographic features, including cystic lung disease, spontaneous pneumothorax or nodular opacity, have been reported intermittently in patients with P. jiroveci pneumonia. We report the case of a 29-year-old woman with a transplanted kidney whose simple chest radiography and HRCT scan showed numerous miliary nodules in both lungs, mimicking miliary tuberculosis (TB). Under the presumptive diagnosis of miliary TB, empirical anti-TB medication was started. However, Grocott methenamine silver nitrate staining of a transbronchial lung biopsy tissue revealed P. jiroveci infection without evidence of TB. These findings suggest that even in TB-endemic area other etiology such as P. jiroveci as well as M. tuberculosis should be considered as an etiology of miliary lung nodules in immunocompromised patients.
Although Pneumocystis carinii (or P. jiroveci) most often affects patients with acquired immune deficiency syndrome (AIDS), other immunocompromised hosts are at risk for infection by this opportunistic pathogen1. P. jiroveci pneumonia is usually suspected from the radiographic and compatible clinical findings. Radiographically, P. jiroveci pneumonia usually presents as bilateral interstitial infiltration, which may be finely granular, or reticular or of ground glass opacity2, but it may also present with atypical radiographic findings. We describe here a case of P. jiroveci pneumonia that mimicked milariy tuberculosis (TB) in a kidney transplant recipient.
A 29-year-old woman with end-stage renal failure of an unknown etiology underwent kidney transplantation from a living-related donor in April 2005. She received the immunosuppressive drugs cyclosporine (250 mg/day), mycophenolate mofetil (1,080 mg/day), and methylprednisolone (4 mg/day) continuously as well as prophylactic trimethoprim/sulfamethoxazole for the first month after transplantation. There were no complications. Yet seven months later, she was admitted to the Asan Medical Center (Seoul, South Korea) for intermittent fever without respiratory symptoms.
Her previous medical history was remarkable only for successful treatment of chronic hepatitis C infection with interferon alpha-2b. She denied any previous contact with TB patients. She had received the BCG vaccination as a child and had a BCG scar on her left shoulder area. She also denied any history of smoking or drinking alcohol. Prior to kidney transplantation, her simple chest radiography showed normal lungs except for mild cardiomegaly.
The physical examination showed that her blood pressure was 126/83 mmHg, the respiratory rate 20/min, the heart rate 101/min and the body temperature ranged up to 39.4℃. Chest examination revealed no abnormal breathing sounds. Examination of the heart, abdomen and the rest of her body was unremarkable. The other laboratory tests were within their normal ranges, except that she had mild leukocytosis (white blood cell count: 10,300/mm3), mild hypoalbuminemia (3.3 g/dL), and an increased C-reactive protein (CRP, 3.52 mg/dL) concentration. Serologic test for human immunodeficiency virus (HIV) was negative. Her blood cyclosporine concentration was 137 ng/mL, which was within the normal range.
Chest radiography showed diffuse ground glass opacity with fine nodules in both lungs, suggesting miliary TB with interstitial edema or the early stage of acute respiratory distress syndrome (ARDS) pattern (Figure 1). An HRCT scan showed numerous diffuse miliary nodules less than 3 mm in diameter and scattered ground glass opacities in both lungs, and these findings strongly suggested miliary TB (Figure 2). P. jiroveci pneumonia was not included in the differential diagnosis at this time.
Because of high fever and the radiographic finding typical of miliary TB, she was immediately started on anti-TB medication; this was followed by bronchofiberscopy with bronchoalveolar lavage (BAL) and transbronchial lung biopsy (TBLB) to confirm the diagnosis and obtain the causative organism for drug sensitivity test. Her fever subsided immediately after the start of anti-TB treatment. The staining for acid-fast-bacilli and P. jiroveci, and the PCR for TB were all negative. The culture reports were all negative for ordinary bacteria, P. jiroveci and fungi in the BAL fluid. Yet unexpectedly, TBLB showed a pathological feature indicative of P. jiroveci pneumonia. The H&E staining showed that the interstitial infiltration was composed of lymphocytes and eosinophils, and Grocott methenamine silver nitrate staining showed many P. jiroveci organisms in the intraalveolar exudates (Figure 3). There were no findings suggestive of mycobacterial infection such as granuloma formation and/or caseous necrosis.
The anti-TB medication was discontinued and trimethoprim (280 mg/day) and sulfamethoxazole (1,400 mg/day) were prescribed for 2 weeks. After 2 weeks of treatment with trimethoprim and sulfamethoxazole, the patient was free of symptoms; her chest radiography showed an absence of diffuse military nodules and ground glass opacities.
Despite a declining prevalence, P. jiroveci remains an important opportunistic pathogen in immunocompromised hosts and especially in AIDS patients1. Its radiographic features, that is, the bilateral interstitial pattern that may be characterized as finely granular or reticular, or of ground glass opacity, are often used to diagnose P. jiroveci pneumonia2. However, various atypical radiographic features have been reported in patient both with and without AIDS. These atypical manifestations have ranged from normal chest radiographs to unilateral infiltrates, nodules, cavities, pneumatoceles, lymphadenopathy and pleural effusion2.
The miliary pattern of P. jiroveci pneumonia is very unusual compared with miliary TB. Only a few cases of miliary-type P. jiroveci pneumonia have been reported in immunosuppressed hosts, including both those cases with and without AIDS3-5. Although nodular granulomatous P. jiroveci pneumonia with multiple pulmonary nodules has been reported6,7, the description of the radiographic result was imprecise. However, a miliary nodular pattern of P. jiroveci pneumonia that's suggestive of miliary TB had been reported in a patient with Letterer-Swie's disease8. To the best of our knowledge, this case is the first report of miliary type P. jiroveci pneumonia in Korea.
In Korea, the notification rate of new TB cases in 2004 was 65.4 presons/100,000 general population, making Korea a country with an intermediate TB-burden. In contrast, the incidence of HIV infection in Korea (cumulative incidence: 3,153 persons in the total general population from 1985 to 2004) is much lower than that of western countries. Hence, the first presumptive diagnosis for miliary lung nodules in a compatible clinical setting is usually TB. At times, and especially for severe cases, anti-TB drugs are prescribed prior to the confirmatory diagnosis. In our case, anti-TB medication was started before we confirmed the diagnosis because the patient was in an immunosuppressed state, and the HRCT findings were quite supportive of miliary TB.
In this patient, the ground glass opacities with accompanying miliary nodules may be indicative of P. jiroveci pneumonia rather than of miliary TB. However, miliary TB also often shows ground glass opacity on CT images with or without ARDS9.
To confirm P. jiroveci pneumonia, the diagnostic procedures should include sputum induction with hypertonic saline, bronchofiberscopy (BAL and TBLB) or open lung biopsy. Bronchofiberscopy with BAL and/or TBLB is the most commonly used invasive procedure for diagnosing pneumocystosis10.
In our patient, the first presumptive diagnosis was miliary TB and her fever subsided immediately after the start of anti-TB treatment. Nevertheless, bronchofiberscopy was performed and this led to the correct diagnosis. This finding suggests that even if the clinical and/or radiological findings suggest a specific etiology, a confirmatory diagnostic work-up should be performed, especially for immunocompromised patients who display miliary lung lesion. In other similar cases, performing TBLB or open lung biopsy was also necessary to confirm pneumocystosis3,4.
Among the questions that linger in this case was why did the patient's fever subside immediately after the start of anti-TB treatment? No drugs were changed nor were there newly prescribed drug given concomitant with the anti-TB drugs, and there is no evidence that the first-line anti-TB drugs are effective for the treatment of P. jiroveci pneumonia. Although rifampicin may increase the metabolic degradation of cyclosporin and therefore upregulate immune functioning, this seems unlikely because the defervescence was too rapid. Although dual infection with M. tuberculosis and P. jiroveci was doubtful in our case, it was unlikely that this patient was coinfected with P. jiroveci and M. tuberculosis since her fever did not recur after withdrawal of anti-TB drugs and there was no evidence suggestive of TB on the AFB smears and cultures, the TB-PCR and the histologic findings.
In conclusion, P. jiroveci pneumonia can manifest as miliary lung nodules, and this is a result that's usually indicative of miliary TB. This finding suggests that in immunocompromised patients, and even in endemic areas of TB, a confirmatory diagnostic work-up is required for patients who have the clinical and/or radiographic findings suggestive of miliary TB.
Figures and Tables
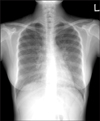 | Figure 1Chest radiography shows diffuse ground glass opacity with suspicious fine granularity in both lungs; this is suggestive of atypical pneumonia or edema with an interstitial pattern. |
References
1. Bartlett JG. Pneumonia in the patient with HIV infection. Infect Dis Clin North Am. 1998. 12:807–820.
2. McGuinness G, Gruden JF. Viral and Pneumocystis carinii infections of the lung in the immunocompromised host. J Thorac Imaging. 1999. 14:25–36.
3. Flannery MT, Quiroz E, Grundy LS, Brantley S. Pneumocystis carinii pneumonia with an atypical granulomatous response. South Med J. 1996. 89:409–410.
4. Ong EL, Murray H, Ellis ME. Miliary Pneumocystis carinii pneumonia in acquired immunodeficiency syndrome. Int J STD AIDS. 1992. 3:54–55.
5. Kennedy CA, Goetz MB. Atypical roentgenographic manifestations of Pneumocystis carinii pneumonia. Arch Intern Med. 1992. 152:1390–1398.
6. Hazzan M, Copin MC, Pruvot FR, Codaccioni MX, Dracon M, Lelievre G, et al. Lung granulomatous pneumocystosis after kidney transplantation: an uncommon complication. Transplant Proc. 1997. 29:2409.
7. Leroy X, Copin MC, Ramon P, Jouet JP, Gosselin B. Nodular granulomatous Pneumocystis carinii pneumonia in a bone marrow transplant recipient: case report. APMIS. 2000. 108:363–366.
8. Cohen WN, McAlister WH. Pneumocystis carinii pneumonia: report of four cases. Am J Roentgenol Radium Ther Nucl Med. 1963. 89:1032–1037.
9. Lee JY, Lee KS, Jung KJ, Han J, Kwon OJ, Kim J, et al. Pulmonary tuberculosis: CT and pathologic correlation. J Comput Assist Tomogr. 2000. 24:691–698.
10. Kroe DM, Kirsch CM, Jensen WA. Diagnostic strategies for Pneumocystis carinii pneumonia. Semin Respir Infect. 1997. 12:70–78.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download