Abstract
Background
Particulate matter may be toxic to human tissue. Ambient air particulate matter ≤10µm in aerodynamic size (PM10), which changes under different environmental conditions, is a complex mixture of organic and inorganic compounds. The Asian dust event caused by meteorological phenomena can also spread unique particulate matter in affected areas. We evaluated production of ROS, TGF-β, fibronectin, and NFκB by exposing normal epithelial cells to Asian dust particulate matter.
Methods
Bronchial epithelial cells were exposed to 0, 50, 100µg/ml of a suspension of PM10 for 24 h. ROS were detected by measurement of DCF release from DCF-DA by FACScan. TGF-β, fibronectin, and NFκB were detected by western blotting.
Figures and Tables
 | Figure 1(A) ROS expression in WI26-VA4 cells after 24 hours. WI 26 cells were exposed to 0 to 100µg/ml of a suspension of ambient particulate matter with a diameter of less than 10µm (PM10). (B) ROS expression between control and NAC in WI26-VA4 cells. The ROS levels were increased after exposed to PM10, compared to those in NAC and control. ROS expression increased after exposure to PM10 in a dose-dependent manner (*p<0.05, †p<0.1). |
 | Figure 2NF-κB expression in WI- 26 VA4 cells after 24 hours incubation with PM10. The NF-κB Levels were increased after exposed to PM10, compared to those in NAC and control. NFκB expression increased after 24 hours of exposure to PM10 in a dose-dependent manner. |
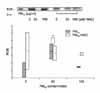 | Figure 3TGF-β expression in WI- 26 VA4 cells after 24 hours incubation with PM10. The TGF-β levels were increased after exposed to PM10, compared to those in NAC and control. TGF-β expression increased after 24 hours of exposure to PM10 in a dose-dependent manner (*p<0.05). |
References
1. Chun YS. The Yellow-Sand phenomenon recorded in the Chosunwangjosilok. J Meteorol Soc Korea. 2000. 36:285–292.
2. Donaldson K, Stone V, Clouter A, Renwick L, MacNee W. Ultrafine particle. Occup Environ Med. 2001. 58:211–219.
3. Buschini A, Cassoni F, Anceschi E, Pasini L, Poli P, Rossi C. Urban airborne particulate: genotoxicity evaluation of different size fractions by mutagenesis tests on microorganisms and comet assay. Chemosphere. 2001. 44:1723–1736.
4. Dihl RR, da Silva CG, do Amaral VS, Reguly ML, de Andrade HH. Mutagenic and recombinagenic activity of airborne particulates, PM10 and TSP, organic extracts in the Drosophila wing-spot test. Environ Pollut. 2008. 151:47–52.
5. Hong YH, Peng HB, La Fata V, Liao JK. Hydrogen peroxide-mediated transcriptional induction of macrophage colony-stimulating factor by TGF-beta1. J Immunol. 1997. 159:2418–2423.
6. Junn E, Lee KN, Ju HR, Han SH, Im JY, Kang HS, et al. Requirement of hydrogen peroxide generation in TGF-beta 1 signal transduction in human lung fibroblast cells: involvement of hydrogen peroxide and Ca2+ in TGF-beta 1-induced IL-6 expression. J Immnol. 2000. 165:2190–2197.
7. Hagiwara SI, Ishii Y, Kitamura S. Aerosolized administration of N-acetylcysteine attenuates lung fibrosis induced by bleomycin in mice. Am J Respir Crit Care Med. 2000. 162:225–231.
8. Kim JH, Jeon HK, Kim MK, Kyung SY, An CH, Lee SP, et al. Particulate matter from Asian dust storms induces the expression of proinflammatory cytokine in A549 epithelial cells. Tuberc Respir Dis. 2006. 60:663–672.
9. Jimenez LA, Thomson J, Brown D. PM10 particles activate NF-κB in alveolar epithelial cells. Am J Respir Crit Care Med. 1999. 159:A27.
10. Hwang HJ, Kang SJ, Kang SE, Park YM, Kim HK, Roh CU. Single particle characterization of aerosol samples collected during Asian dust storm events and non-Asian dust period in Incheon. Proceedings of the 2007 Environmental Societies Joint Conference; 2007 May 2-4; Busan, Korea. 2007. 2007 May 2-4; Busan, Korea. Seoul: Korean Society for Atmospheric Environment;179–182.
11. Baek KW, Chung JD. Study on the yellow sandy dust phenomena in Korean peninsula and chemical compositions in fine particles at background sites of Korea. Korean J Sanitation. 2004. 19:9–18.
12. Gonzalez-Flecha B. Oxidant mechanisms in response to ambient air particles. Mol Aspects Med. 2004. 25:169–182.
13. Ha EH. Effects of Asian dust storm on health. The magazine of the Society of Air-conditioning and Refrigeration Engineering of Korea. 2006. 35:21–25.
14. Donaldson K, Stone V. Current hypotheses on the mechanisms of toxicity of ultrafine particles. Ann Ist Super Sanita. 2003. 39:405–410.
15. Long CM, Suh HH, Kobzik L, Catalano PJ, Ning YY, Koutrakis P. A pilot investigation of the relative toxicity of indoor and outdoor fine particles: in vitro effects of endotoxin and other particulate properties. Environ Health Perspect. 2001. 109:1019–1026.
16. Osornio-Vargas AR, Bonner JC, Alfaro-Moreno E, Martínez L, García-Cuellar C, Ponce-de-León Rosales S, et al. Proinflammatory and cytotoxic effects of Mexico City air pollution particulate matter in vitro are dependent on particle size and composition. Environ Health Perspect. 2003. 111:1289–1293.
17. Brown DM, Wilson MR, MacNee W, Stone V, Donaldson K. Size-dependent proinflammatory effects of ultrafine polystyrene particles: a role for surface area and oxidative stress in the enhanced activity of ultrafines. Toxicol Appl Pharmacol. 2001. 175:191–199.
18. Jiang Z, Seo JY, Ha H, Lee EA, Kim YS, Han DC, et al. Reactive oxygen species mediate TGF-beta1-induced plasminogen activator inhibitor-1 upregulation in mesangial cells. Biochem Biophys Res Commun. 2003. 309:961–966.
19. Lee HB, Yu MR, Yang Y, Jiang Z, Ha H. Reactive oxygen species-regulated signaling pathways in diabetic nephropathy. J Am Soc Nephrol. 2003. 14:S241–S245.
20. Vallyathan V, Shi X. The role of oxygen free radicals in occupational and environmental lung diseases. Environ Health Perspect. 1997. 105 Suppl 1:165–177.
21. Ha H, Yu MR, Uh ST, Park CS, Lee HB. Role of reactive oxygen species in transforming growth factor - beta1 - inuduced fibronectin secretion and alpha - smooth muscle actin expression in human lung fibroblasts. Tuberc Respir Dis. 2005. 58:267–275.




 PDF
PDF ePub
ePub Citation
Citation Print
Print








 XML Download
XML Download