Abstract
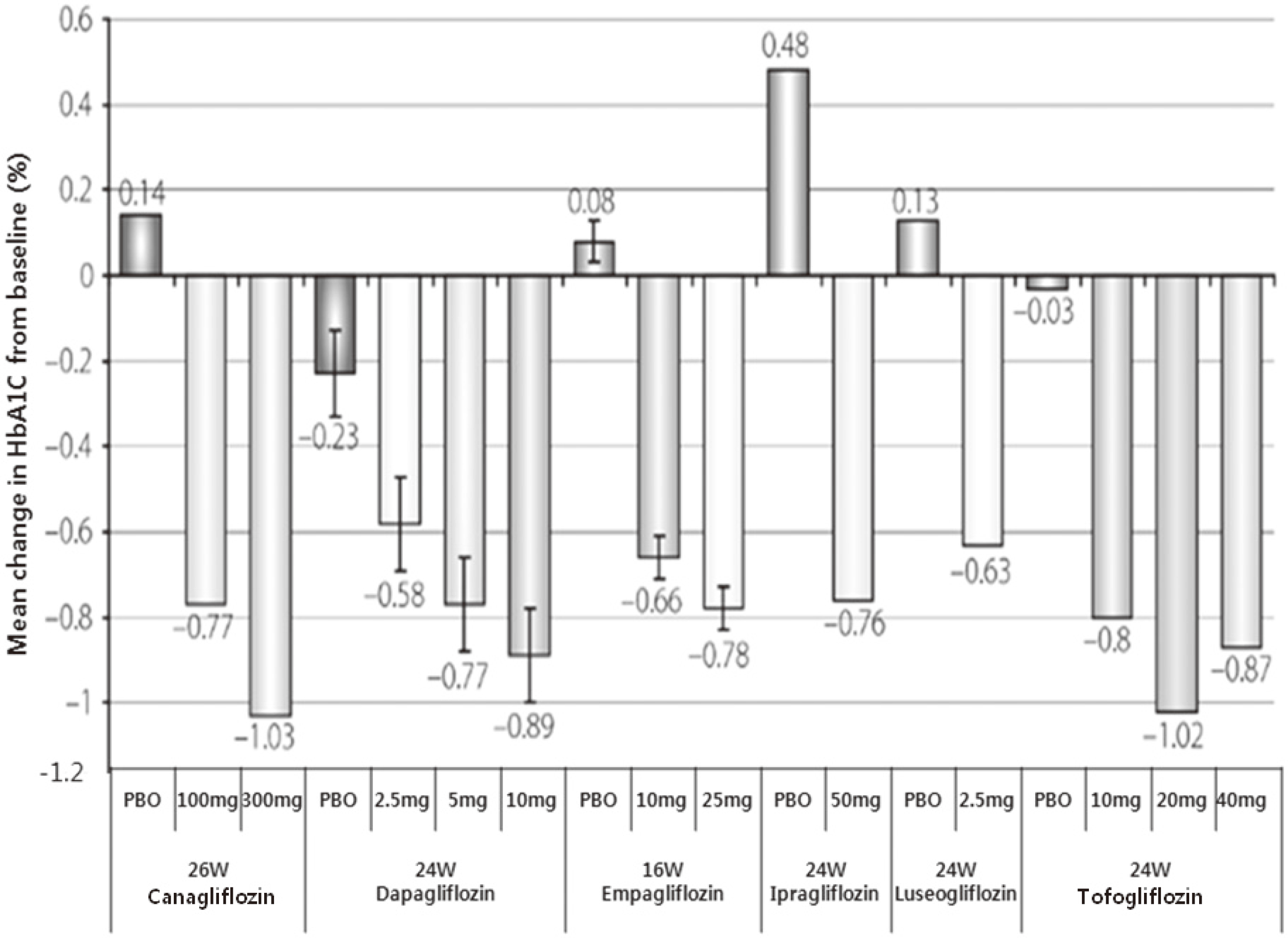
There are numerous glucose lowering agents in clinical use for type 2 diabetic patients, yet most do not achieve glycemic targets. Recently, inhibitors of the sodium glucose cotransporter 2 (SGLT2) have been developed, which show great potential of being a novel therapeutic option to treat type 2 diabetes. Many researches have turned their attention to the kidney as a target because renal glucose reabsorption is increased in type 2 DM. SGLT2 is a protein transporter that mediates glucose reabsorption from glomerular filtrates. In an insulin-independent manner, inhibition of SGLT2 increases urinary glucose excretion and decreases plasma glucose levels. To date, there are a few SGLT2 inhibiting agents being clinically studied both as a monotherapy and in combination with other antidiabetic agents. This review will focus on the efficacy of SGLT2 inhibitors based on clinical studies.
REFERENCES
1. Gerich JE. Role of the kidney in normal glucose homeostasis and in the hyperglycaemia of diabetes mellitus: therapeutic implications. Diabet Med. 2010; 27:136–42.

2. Hasan FM, Alsahli M, Gerich JE. SGLT2 inhibitors in the treatment of type 2 diabetes. Diabetes Res Clin Pract. 2014; 104:297–322.

3. Fujita Y, Inagaki N. Renal sodium glucose cotransporter 2 inhibitors as a novel therapeutic approach to treatment of type 2 diabetes: Clinical data and mechanism of action. J Diabetes Investig. 2014; 5:265–75.

4. Kaku K, Inoue S, Matsuoka O, Kiyosue A, Azuma H, Hayashi N, Tokudome T, Langkilde AM, Parikh S. Efficacy and safety of dapagliflozin as a monotherapy for type 2 diabetes mellitus in Japanese patients with inadequate glycaemic control: a phase II multicentre, randomized, double-blind, placebo-controlled trial. Diabetes Obes Metab. 2013; 15:432–40.

5. List JF, Woo V, Morales E, Tang W, Fiedorek FT. Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes. Diabetes Care. 2009; 32:650–7.

6. Ferrannini E, Ramos SJ, Salsali A, Tang W, List JF. Dapagliflozin monotherapy in type 2 diabetic patients with inadequate glycemic control by diet and exercise: a randomized, double-blind, placebo-controlled, phase 3 trial. Diabetes Care. 2010; 33:2217–24.
7. Ji L, Ma J, Li H, Mansfield TA, T'joen CL, Iqbal N, Ptaszynska A, List JF. Dapagliflozin as monotherapy in drug-naive Asian patients with type 2 diabetes mellitus: a randomized, blinded, prospective phase III study. Clin Ther. 2014; 36:84–100.e9.

8. Henry RR, Murray AV, Marmolejo MH, Hennicken D, Ptaszynska A, List JF. Dapagliflozin, metformin XR, or both: initial pharmacotherapy for type 2 diabetes, a randomised controlled trial. Int J Clin Pract. 2012; 66:446–56.

9. Bailey CJ, Gross JL, Pieters A, Bastien A, List JF. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with metformin: a randomised, double-blind, placebo-controlled trial. Lancet. 2010; 375:2223–33.

10. Bolinder J, Ljunggren Ö, Kullberg J, Johansson L, Wilding J, Langkilde AM, Sugg J, Parikh S. Effects of dapagliflozin on body weight, total fat mass, and regional adipose tissue distribution in patients with type 2 diabetes mellitus with inadequate glycemic control on metformin. J Clin Endocrinol Metab. 2012; 97:1020–31.

11. Nauck MA, Del Prato S, Meier JJ, Durán-García S, Rohwedder K, Elze M, Parikh SJ. Dapagliflozin versus glipizide as add-on therapy in patients with type 2 diabetes who have inadequate glycemic control with metformin: a randomized, 52-week, double-blind, active-controlled noninferiority trial. Diabetes Care. 2011; 34:2015–22.
12. del Prato S, Nauck MA, Durán-garcia S, Rohwedder K, Theuerkauf A, Langkilde AM, Parikh SJ. Durability of dapagliflozin vs. glipizide as add-on therapies in T2DM inadequately controlled on metformin: 4-year data. In: American Diabetes Association. 73rd Scientific Session. 2013 Jun. Chicago.
13. Strojek K, Yoon KH, Hruba V, Elze M, Langkilde AM, Parikh S. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with glimepiride: a randomized, 24-week, double-blind, placebo-controlled trial. Diabetes Obes Metab. 2011; 13:928–38.

14. Rosenstock J, Vico M, Wei L, Salsali A, List JF. Effects of dapagliflozin, an SGLT2 inhibitor, on HbA(1c), body weight, and hypoglycemia risk in patients with type 2 diabetes inadequately controlled on pioglitazone monotherapy. Diabetes Care. 2012; 35:1473–8.

15. Jabbour SA, Hardy E, Sugg J, Parikh S. Study 10 Group. Dapagliflozin is effective as add-on therapy to sitagliptin with or without metformin: a 24-week, multicenter, randomized, double-blind, placebo-controlled study. Diabetes Care. 2014; 37:740–50.

16. Wilding JP, Norwood P, T'joen C, Bastien A, List JF, Fiedorek FT. A study of dapagliflozin in patients with type 2 diabetes receiving high doses of insulin plus insulin sensitizers: applicability of a novel insulin-independent treatment. Diabetes Care. 2009; 32:1656–62.
17. Kohan DE, Fioretto P, Tang W, List JF. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int. 2014; 85:962–71.

18. Stenlöf K, Cefalu WT, Kim KA, Alba M, Usiskin K, Tong C, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes Metab. 2013; 15:372–82.

19. Inagaki N, Kondo K, Yoshinari T, Maruyama N, Susuta Y, Kuki H. Efficacy and safety of canagliflozin in Japanese patients with type 2 diabetes: a randomized, double-blind, placebo-controlled, 12-week study. Diabetes Obes Metab. 2013; 15:1136–45.

20. Lavalle-González FJ, Januszewicz A, Davidson J, Tong C, Qiu R, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin compared with placebo and sitagliptin in patients with type 2 diabetes on background metformin monotherapy: a randomised trial. Diabetologia. 2013; 56:2582–92.

21. Matthews D, Fulcher G, Perkovic V, de Zeeuw D, Mahaffey KW, Rosenstock J, Davies M, Capuano G, Desai M, Shaw W, Vercruysse F, Meininger G, Neal B. Efficacy and safety of canagliflozin, an inhibitor of sodium glucose cotransporter 2, added on to insulin therapy with or without oral agents in type 2 diabetes. Diabetologie und Stoffwechsel. 2013; 8(Suppl 1):S251.

22. Schernthaner G, Gross JL, Rosenstock J, Guarisco M, Fu M, Yee J, Kawaguchi M, Canovatchel W, Meininger G. Canagliflozin compared with sitagliptin for patients with type 2 diabetes who do not have adequate glycemic control with metformin plus sulfonylurea: a 52-week randomized trial. Diabetes Care. 2013; 36:2508–15.
23. Forst T, Guthrie R, Goldenberg R, Yee J, Vijapurkar U, Meininger G, Stein P. Efficacy and safety of canagliflozin over 52 weeks in patients with type 2 diabetes on background metformin and pioglitazone. Diabetes Obes Metab. 2014; 16:467–77.

24. Ferrannini E, Seman L, Seewaldt-Becker E, Hantel S, Pinnetti S, Woerle HJ. A Phase IIb, randomized, placebo-controlled study of the SGLT2 inhibitor empagliflozin in patients with type 2 diabetes. Diabetes Obes Metab. 2013; 15:721–8.

25. Rosenstock J, Seman LJ, Jelaska A, Hantel S, Pinnetti S, Hach T, Woerle HJ. Efficacy and safety of empagliflozin, a sodium glucose cotransporter 2 (SGLT2) inhibitor, as add-on to metformin in type 2 diabetes with mild hyperglycaemia. Diabetes Obes Metab. 2013; 15:1154–60.

26. Ferrannini E, Berk A, Hantel S, Pinnetti S, Hach T, Woerle HJ, Broedl UC. Long-term safety and efficacy of empagliflozin, sitagliptin, and metformin: an active-controlled, parallel-group, randomized, 78-week open-label extension study in patients with type 2 diabetes. Diabetes Care. 2013; 36:4015–21.
27. Häring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Woerle HJ, Broedl UC. EMPA-REG METSU Trial Investigators. Empagliflozin as add-on to metformin plus sulfonylurea in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial. Diabetes Care. 2013; 36:3396–404.
28. Rosenstock J, Jelaska A, Frappin G, Salsali A, Kim G, Woerle HJ, Broedl UC. EMPA-REG MDI Trial Investigators. Improved glucose control with weight loss, lower insulin doses, and no increased hypoglycemia with empagliflozin added to titrated multiple daily injections of insulin in obese inadequately controlled type 2 diabetes. Diabetes Care. 2014; 37:1815–23.

29. Kovacs CS, Seshiah V, Swallow R, Jones R, Rattunde H, Woerle HJ, Broedl UC. EMPA-REG PIO™ trial investigators. Empagliflozin improves glycaemic and weight control as add-on therapy to pioglitazone or pioglitazone plus metformin in patients with type 2 diabetes: a 24-week, randomized, placebo-controlled trial. Diabetes Obes Metab. 2014; 16:147–58.

30. Fonseca VA, Ferrannini E, Wilding JP, Wilpshaar W, Dhanjal P, Ball G, Klasen S. Active- and placebo-controlled dose-finding study to assess the efficacy, safety, and tolerability of multiple doses of ipragliflozin in patients with type 2 diabetes mellitus. J Diabetes Complications. 2013; 27:268–73.

31. Kashiwagi A, Kazuta K, Goto K, Yoshida S, Ueyama E, Utsuno A. Ipragliflozin in combination with metformin for the treatment of Japanese patients with type 2 diabetes: ILLUMINATE, a randomized, double-blind, placebo-controlled study. Diabetes Obes Metab 2014 Jun 12 [Epub].http://dx.doi.org/10.1111/dom.12331.
32. Seino Y, Sasaki T, Fukatsu A, Ubukata M, Sasaki T, Kasai Y, Sakai S, Samukawa Y. Luseogliflozin, a SGLT2 inhibitor, improves glycaemic control and reduces body weight as monotherapy up to 52 weeks in Japanese patients with type 2 diabetes mellitus. Diabetologia. 2013; 56(Suppl 1):S384.
33. Inagaki N, Seino Y, Sasaki T, Fukatsu A, Ubukata M, Umeuchi H, Sakai S, Samukawa Y. Luseogliflozin, a selective SGLT2 inhibitor, added on to glimepiride for 52 weeks improves glycaemic control with no major hypoglycaemia in Japanese type 2 diabetes patients. Diabetologia. 2013; 56(Suppl 1):S82.
34. Haneda M, Seino Y, Sasaki T, Fukatsu A, Ubukata M, Sato Y, Sakai S, Samukawa Y. Luseogliflozin, a SGLT2 inhibitor, as add-on therapy to 5 types of oral antidiabetic drugs improves glycaemic control and reduces body weight in Japanese patients with type 2 diabetes mellitus. Diabetologia. 2013; 56(Suppl 1):S384.
35. Tanizawa Y, Araki E, Tobe K, Terauchi Y, Utsunomiya K, Iwamoto Y, Watada H, Kaku K. the Tofogliflozin 004 and 005 Study group. Efficacy and safety of tofogliflozin administered for 52 weeks as monotherapy or combined with other oral hypoglycaemic agents in Japanese patients with type 2 diabetes. Diabetologia. 2013; 56(Suppl 1):S82–S83.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download