Abstract
Background and Purpose
Methods
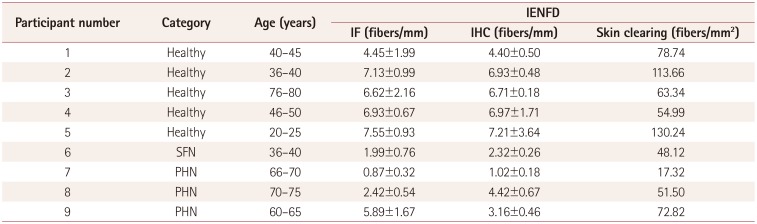
Results
Conclusions
Acknowledgements
Notes
Author Contributions:
Conceptualization: Byung-Jo Kim, Dai Hyun Kim, Im Joo Rhyu, Woong Sun.
Data curation: Dai Hyun Kim, Eunsoo Lee, Se Jeong Lee.
Formal analysis: Dai Hyun Kim.
Funding acquisition: Hyo Hyun Ahn, Im Joo Rhyu, Woong Sun.
Investigation: Byung-Jo Kim, Dai Hyun Kim, Hyo Hyun Ahn, Ji Hyuck Hong, Soo Hong Seo.
Methodology: Byung-Jo Kim, Dai Hyun Kim, Im Joo Rhyu, Woong Sun.
Project administration: Byung-Jo Kim, Hyo Hyun Ahn, Im Joo Rhyu, Soo Hong Seo, Woong Sun.
Resources: Eunsoo Lee, Woong Sun.
Supervision: Byung-Jo Kim, Hyo Hyun Ahn, Im Joo Rhyu, Woong Sun.
Validation: Byung-Jo Kim, Dai Hyun Kim, Im Joo Rhyu.
Visualization: Dai Hyun Kim, Se Jeong Lee.
Writing—original draft: Dai Hyun Kim.
Writing—review & editing: Byung-Jo Kim, Dai Hyun Kim, Hyo Hyun Ahn, Im Joo Rhyu, Soo Hong Seo, Woong Sun.
References
Fig. 1
Summary of the processing steps of the cutaneous ACT-PRESTO protocol, from separating the epidermis from biopsied skin samples to imaging with confocal microscopy. ACT-PRESTO: active clarity technique-pressure related efficient and stable transfer of macromolecules into organs, c-PRESTO: centrifugal-PRESTO, ETC: electrophoretic tissue clearing, RI: reflective index, RIMS: reflective-index matching solution, SSST: salt split skin test.
Fig. 2
Serial changes in transparency and area during the cutaneous ACT-PRESTO process, focusing on ETC and immersion in RIMS. A: Morphological changes in transparency according to the duration of ETC. B: Quantitative alterations of the relative transparency according to the duration of ETC. The improvement in relative transparency was proportional to the duration of ETC, but the differences were standardized after immersion in RIMS. C: Extent of area change related to the duration of ETC. The skin specimens consistently expanded after ETC in proportion to the duration of ETC. The enlarged tissues returned to their original size after washing with PBS. However, ETC durations exceeding 4 h resulted in irreversible shrinkage of the original tissues to an extent proportional to the ETC duration. ACT-PRESTO: active clarity technique-pressure related efficient and stable transfer of macromolecules into organs, ETC: electrophoretic tissue clearing, PBS: phosphate-buffered saline, RIMS: reflective-index matching solution.
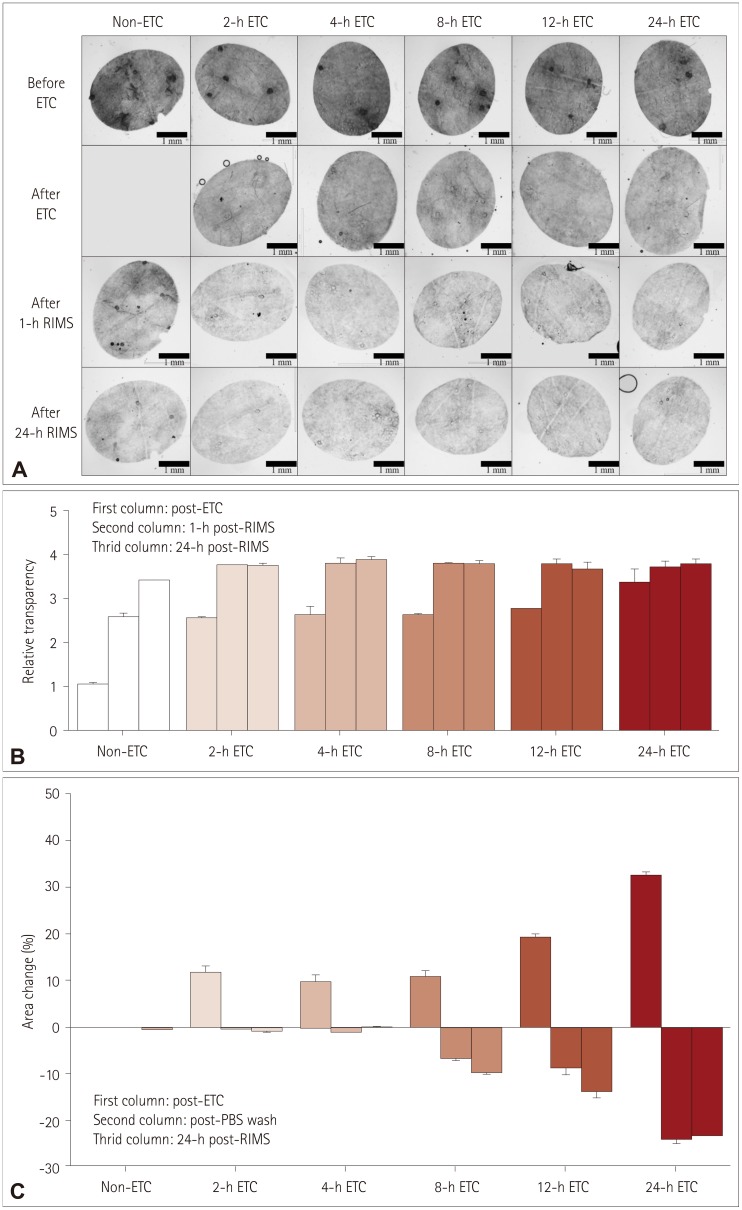
Fig. 3
Schematic diagram of immunohistochemistry for whole-tissue processing after tissue-clearing. The effectiveness of combinations of target antigens and specific antibodies was substantially impeded by the thicker whole tissue compared with conventional sectioned tissue. Tissues for c-PRESTO were centrifuged to obtain sufficient penetration depths of the primary and secondary antibodies, and so the technique could increase the probability of binding between target antigens and specific antibodies. c-PRESTO: centrifugal-pressure related efficient and stable transfer of macromolecules into organs.
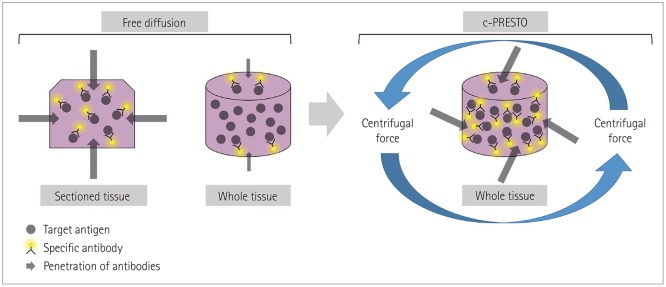
Fig. 4
Characteristics of the IENFs of a healthy participant (A, C, and E) and a PHN patient (B, D, and F). IENFs were quantified using the conventional methods of indirect IF (A and B) and IHC (C and D), as well as the ACT-PRESTO tissue-clearing technique (E and F). Cross-sectional views (A–D) and bird's-eye views (E and F) of IENFs. IF images (A and B) were obtained using confocal microscopy with a Plan-Apochromat 20×/0.8 M27 lens (maximum projection; stack size, 80 µm; stack step, 1 µm). IHC images (C and D) were produced using bright-field microscopy with a UPlanApo 40×/0.85 lens. Cutaneous nerve fibers (arrows) and the basal layer of the epidermis (arrowheads) (A–D). Images associated with skin-clearing (E and F) were also obtained using confocal microscopy with a Plan-Apochromat 20×/0.8 M27 lens (stack size, 200 µm; stack step, 1 µm). ACT-PRESTO: active clarity technique-pressure related efficient and stable transfer of macromolecules into organs, Der: dermis, Ep: epidermis, IENFs: intraepidermal nerve fibers, IF: immunofluorescence, IHC: immunohistochemistry, PGP 9.5: protein gene product 9.5, PHN: postherpetic neuralgia.
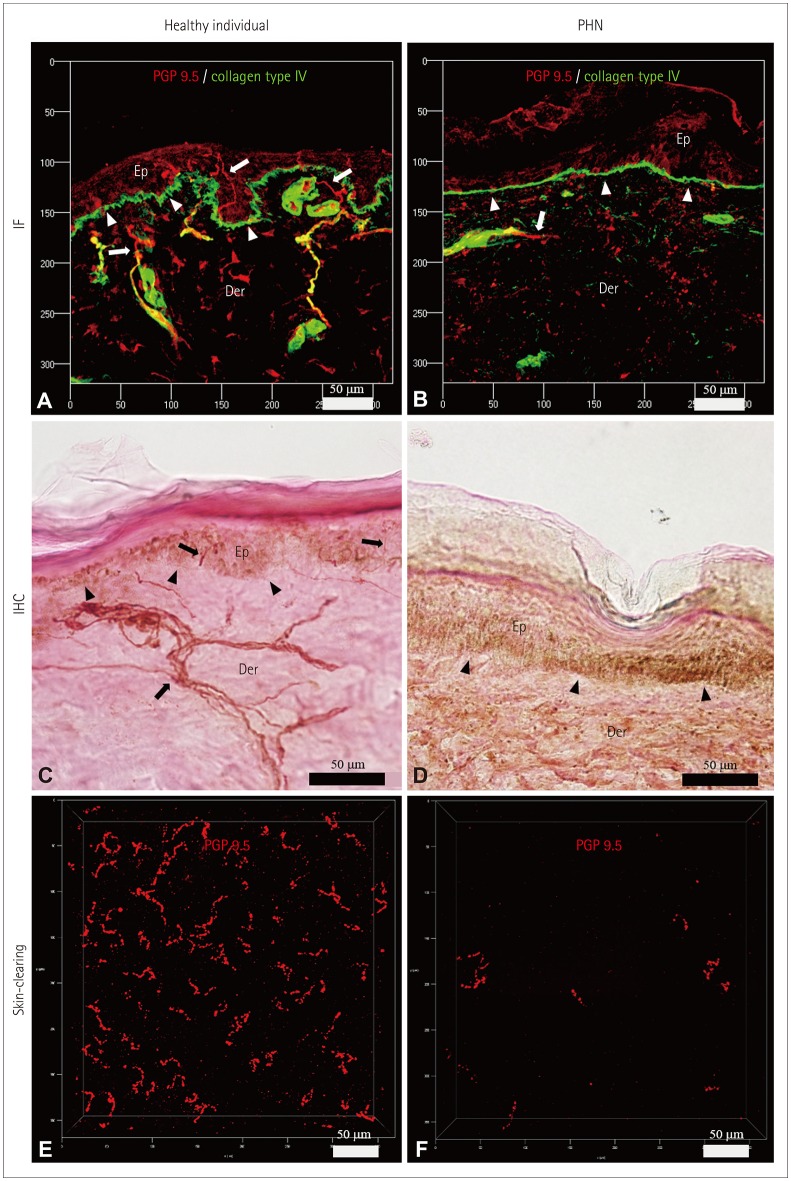
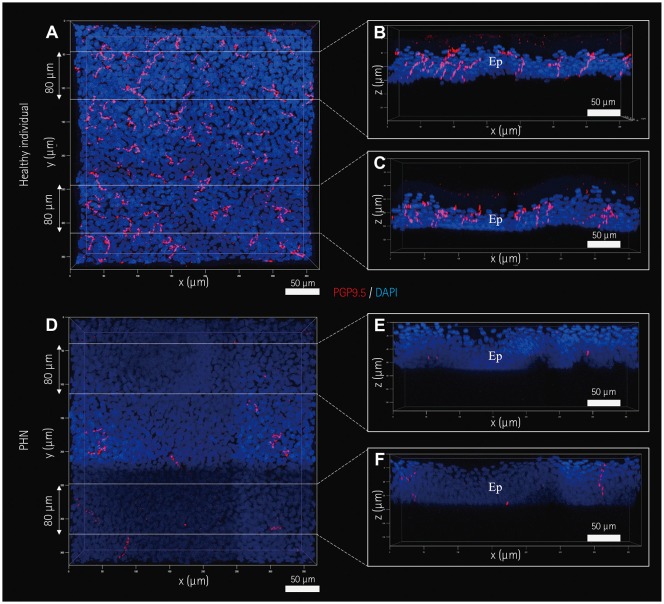
Fig. 5
Images of IENFs produced by the skin-clearing technique. IENFs of a healthy subject (A–C) and a PHN patient (D–F). Selected areas of three-dimensional images (A and D) in Fig. 4E and F, respectively, were optically sectioned at a thickness of 80 µm (B–F). All images were obtained and processed using confocal microscopy with a UPlanApo 40×/0.85 lens and the related software. Ep: epidermis, IENFs: intraepidermal nerve fibers, PGP 9.5: protein gene product 9.5, PHN: postherpetic neuralgia.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download