Dear Editor:
Linear Immunoglobulin A bullous dermatosis (LABD) is a rare, heterogeneous, acquired, subepidermal and autoimmune blistering disorder. It is characterized by linear deposits of immunoglobulin (Ig) A along the basement membrane zone (BMZ) that are seen by direct immunofluorescence (IF)1. The etiology of LABD is not fully understood, but some cases are reported in association with drugs, infections, malignancies and connective tissue disease2.
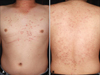
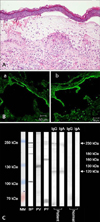
A 33-year-old man presented with a 5-month history of pruritic erythematous papules and vesicles over the trunk and extremities. The skin eruptions were seen as dermatitis herpetiformis-like lesions (Fig. 1). The skin biopsy showed a subepidermal blister containing mainly lymphocytes and neutrophils infiltrated in the upper dermis (Fig. 2A). For diagnosis, we performed additional tests including IF, immunoblotting and the enzyme linked immunosorbent assay (ELISA). The direct IF showed linear deposits of IgA and C3 in the BMZ, and the indirect IF with 1 M NaCl split-skin showed linear deposits of IgA and IgG in the epidermal side of 1 M NaCl-split skin (Fig. 2B). In immunoblotting, there are found 230 KDa and 120 KDa molecular sized polypeptides on IgA and IgG, corresponding to LAD-1 antigens, respectively (Fig. 2C). On ELISA (Medical & Biological Laboratories Co., Ltd., Nagoya, Japan), serum autoantibody level against BP180 NC1 was markedly elevated [BP 180 (38.648 U/ml), BP230 (2.498 U/ml), Dsg 1 (2.253 U/ml), Dsg 3 (9.606 U/ml)]. Most lamina lucida-type LABD sera react with the 97-kDa and 120-kDa LAD-1, truncated extracellular domains of BP180. Also in consideration our results of IF, immunoblotting and ELISA, we made a diagnosis of lamina lucida-type LABD3. Oral dapsone (100~200 mg/day) was started, but not responded favorably, and oral methylprednisolone (12 mg/day), mycophenolate mofetil (MMF) (250 mg/day) was added, but the skin lesions were waxed and waned.
Two months later, the patient's symptoms as malaise, fatigue, photosensitivity and multiple joints arthritis, presented mildly for several years, were aggravated. Laboratory tests revealed anemia, lymphopenia, high erythrocyte sedimentation rate and low C3/C4 levels. Anto-antibodies [(ANA;speckled(1:80), anti-dsDNA(–), anti-Scl-70(1+), antihistone(1+)] were checked. Also, proteinuria and hematuria were found. Renal biopsy revealed IgA lupus nephritis. Finally, systemic lupus erythematosus (SLE) was also diagnosed. Oral methylprednisolone (24 mg/day) and MMF (500 mg/day) were started. The skin lesions and SLE symptoms were improved and the patient has been taking low dose methylprednisolone and dapsone for maintenance since then.
LABD are known as dramatically responded to oral dapsone, inhibiting neutrophils-mediatied reaction as well as IgA adherence4. But our case was not responded. SLE accompanied with LABD may have altered the immune mechanism, which may in turn have resulted in unresponsiveness to dapsone.
Additionally, LABD and IgA lupus nephritis may have similar immune mechanisms related with the deposition of IgA in the BMZ and glomerular mesangium, which suggests the common immunopathogenic mechanisms might be shared in the pathogenesis of LABD associated with SLE5.
In conclusion, our case indicates that the association of LABD and SLE is a rare and interesting event and further studies are needed to illuminate the common immunologic mechanisms between them.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download