Abstract
Background
Psoriasis is characterized by uncontrolled hyperproliferation, aberrant differentiation, and dermal infiltration of immune cells. Recent studies have reported that Wnt5a and Notch1 signaling are altered in psoriatic skin lesions.
Objective
We aimed to investigate the interaction of Wnt5a with Notch 1 with respect to inflammation-mediated epidermal hyperproliferation in psoriasis.
Methods
Expression of Wnt5a and Notch1 signaling-related proteins were examined in psoriatic skin biopsies. Wnt5a was upregulated in human keratinocytes by treating the cells with its recombinant form (rWnt5a).
Results
In psoriatic lesions, expression of Wnt5a increased while that of Notch1 decreased when compared to that in non-lesional and normal skin. Treatment with rWnt5a increased the proliferation of keratinocytes and increased their secretion of interleukin (IL)-23, IL-12, and tumor necrosis factor (TNF)-α. Further, exposure of keratinocytes to IL-1α, TNF-α, transforming growth factor-α, and interferon-γ downregulated Notch1 as well as HES 1, which is downstream to Notch1, but increased the Wnt5a levels. The upregulated Wnt5a in keratinocytes downregulated both Notch1 and HES1.
Psoriasis, a common chronic inflammatory skin disease, is characterized by abnormal proliferation and differentiation of keratinocytes. Further, an abnormality in the Th17 pathway is the immunologic contributor to keratinocyte proliferation1. Recent reports234 have described the upregulation of Wnt5a in psoriasis, but the underlying mechanisms have not been elucidated. In this context, previous studies have reported negative regulation of Wnt canonical signaling, and suggested Notch1 to be a tumor suppressor in mouse skin56.
Wnt5a is the prototypical non-canonical Wnt family ligand. Apart from its complex role in development, Wnt5a, which is expressed in a variety of adult tissues, promotes cell proliferation789 as well as adhesion of fibroblasts1011. Recent evidences support the involvement of Wnt5a in inflammatory responses and innate immunity1213. Meanwhile, the Notch system plays a crucial role in epidermal cell growth and differentiation1415. It consists of four transmembrane receptors (Notch1~4), five transmembrane ligands (Jagged-1 and -2; and Delta-like-1, -3, and -4), and three regulatory proteins (lunatic, radical, and manic fringe) that modulate ligand-receptor-induced signals16. Previous studies have suggested the possible involvement of Notch1 in psoriasis1718.
Wnt and Notch signaling are involved in several pathways depending on the cell line and disease. However, the interaction between the Wnt5a and Notch1 in psoriasis has not yet been studied. Moreover, although aberrant differentiation and uncontrolled proliferation is a key feature of the psoriatic epidermis, the role of these processes and their possible association with immune cells are obscure. Therefore, in this study, we investigated the interaction between Wnt5a and Notch1 signaling in the pathogenesis of psoriasis.
Ten patients with psoriasis were recruited for the study. All donors provided written informed consent, and all procedures were conducted upon approval by the ethics committee of Asan Medical Center (No. 2004-0161).
Two punch biopsies (3~5 mm diameter) were obtained from lesional and non-lesional skin samples of each patient. Total RNA from the biopsy tissues was isolated using an RNeasy isolation kit (Qiagen, Hilden, Germany). cDNA was synthesized using SuperScript II reverse transcriptase (Invitrogen, Carlsbad, CA, USA). Quantitative reverse transcription-polymerase chain reaction (qRT-PCR) was performed using SYBR green PCR reagents (Roche Applied Science, Indianapolis, IN, USA). GAPDH was used as endogenous reference gene, and all experiments were performed thrice. The following specific primers were used: Wnt5a, 5'-TAG CAG CAT CAG TCC ACA AA-3' (sense) and 5'-CAA AAC ACG GCA TCT CTC TT-3' (antisense); Notch1, 5'-TGC GAG GTC AAC ACA GAC GAG-3' (sense) and 5' TGT AAG TGT TGG GTC CGT CCA G-3' (antisense); HES1, 5'-ATG GAG AAA AAT TCC TCG TCC C-3' (sense) and 5'-TTC AGA GCA TCC AAA ATC AGT GT-3' (antisense); and GAPDH, 5'-ATG GGG AAG GTG AAG GTC G-3' (sense) and 5'-GGG GTC ATT GAT GGC AAC AAT A-3' (antisense).
Acetone-fixed cryostat sections and formalin-fixed paraffin-embedded sections were immunostained using an Elite ABC Kit (Vector Laboratories, Burlingame, CA, USA). The primary antibodies used in this study are listed in Table 1. Positively stained cells were scored as follows: 0, no activity; 1, ≤10%; 2, 10% to ≤25%; 3, 25% to ≤50%; 4, 50% to ≤75%; 5, >75%.
Neonatal normal human keratinocytes (NHKs; Invitrogen) were cultured in keratinocyte growth media (EpiLife; Invitrogen) supplemented with Human Keratinocyte Growth Supplement (HKGS; Invitrogen). Human dermal fibroblasts (HDFs) from abdominal skin samples were cultured in Dulbecco's Modified Eagle Medium with 10% fetal bovine serum. All cells were maintained at 37℃ in a humidified atmosphere at 5% CO2.
NHKs (5×104/well) were incubated in a 96-well microtiter plate with culture medium at a final volume of 100 µl/well. Next day, the cells were treated with rWnt5a (100 ng/ml) and transfected with Wnt5a-specific and negative control siRNAs (3 pmol; Bioneer, Daejeon, Korea) using Lipofectamine RNAiMAX transfection reagent (Invitrogen). Cell proliferation was determined after 2 and 3 d using Cell Proliferation enzyme-linked immunosorbent assay (ELISA), Bromodeoxyuridine (BrdU) (Roche Applied Science) following the manufacturer's instructions.
The migration of HDFs and NHKs were assayed using Boyden chambers with 8 µm pore membranes (Transwell; Coster, Boston, MA, USA). Lower surfaces of the transwell membranes were coated with gelatin B (10 µg/ml), following which the membranes were allowed to dry at 25℃ for 1 h. The transwells were assembled in a 24-well plate, and the lower chambers were filled with either control- or Wnt5a-conditioned media containing 100 ng/ml basic-fibroblast growth factor. Cells were cultured in either control- or Wnt5a-conditioned media for 3 d, following which 5×104 cells were added to the upper chambers. The plate was then incubated at 37℃ for 24 h. The cells that migrated to the lower surface of the filters were stained, and enumerated in four randomly selected fields.
NHKs (5×105) were seeded onto 60-mm plates 24 h before the treatments and cultured in media without supplements (except antibiotics) for another 24 h. They were then stimulated with 10 ng/ml of tumor necrosis factor (TNF)-α (Invitrogen), 20 ng/ml of interferon (IFN)-γ (LG Life Sciences, Iksan, Korea), 25 ng/ml of transforming growth factor (TGF)-α (Millipore, Billerica, MA, USA), and 10 ng/ml of interleukin (IL)-1α (R&D Systems, Rochester, MN, USA) and a mixture of four cytokines for 24 h. Total proteins (20 µg) were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS PAGE) and probed with anti-Wnt5a (1:500; Imgenex, San Diego, CA, USA), -Notch1 (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA), and -HES1 (1:500; Millipore) antibodies. β-actin (Sigma-Aldrich, St. Louis, MO, USA) was used as a loading control.
For analyses of phosphorylated proteins, NHKs were stimulated with 100 ng/ml of recombinant mouse Wnt5a (rWnt5a) (Millipore) for 15 min, 30 min, 1 h, and 3 h. Total proteins (30 µg) were resolved by SDS PAGE and probed with anti-phospho-AKT (Ser473) (Cell Signaling, 1:500) and -AKT antibodies (Cell Signaling, 1:500). Densitometric analyses were performed using a VersaDoc Imaging System (Bio-Rad, Hercules, CA, USA).
Cytokines secreted by the Wnt5a-treated NHKs were detected using specific ELISAs. Cells were treated with rWnt5a (100 ng/ml) for 6, 24, 48, and 72 h. The culture media were harvested and ELISAs for TNF-α (Invitrogen) and IL-1α (R&D Systems) were performed according to the manufacturer's instructions. Optical densities were measured using a microplate reader (Molecular Devices, Sunnyvale, CA, USA).
Wnt5a-downregulated genes were investigated in NHKs previously starved in serum-free media for 24 h and treated with 100 ng/ml of rWnt5a for 1, 3, 6, 24, and 72 h. PCR reactions were performed using the following cycling conditions: 94℃ for 5 min followed by 25~30 cycles of 94℃ for 30 s, 60℃ for 30 s, and 72℃ for 30 s.
Statistical analyses were performed by SPSS ver. 12.0 (SPSS Inc., Chicago, IL, USA). All values are expressed as means±standard deviation. Statistical significance between groups was analyzed by the Student's t-test. Paired Student's t-test was performed to analyze the results obtained from paired lesional and non-lesional psoriatic skin samples from each patient. A p-value<0.05 was considered significant.
qRT-PCR analyses showed that Wnt5a mRNA levels increased about 2.4-fold (p<0.001) in the psoriatic lesions compared with that in non-lesional skin samples (Fig. 1A). These results were confirmed by immunohistochemistry (IHC), which showed increased Wnt5a staining in all the layers of lesional skin compared with that in non-lesional skin samples (Fig. 1B, Table 2).
The mRNA levels of several Notch signaling components were evaluated by RT-PCR. There was no change in NOTCH1 mRNA expression in the psoriatic skin biopsies, while that of HES1 was decreased (Fig. 2). IHC analyses confirmed the decrease in Notch1 and HES1 expression at their protein levels in psoriatic lesions compared with that in non-lesional skin samples. Notch1 was distributed from spinous to granular layers of the normal epidermis, but was markedly decreased in psoriatic lesions. IHC staining scores for Notch2, Delta-like 1, and Jagged-1 were also decreased in the psoriatic skin samples, whereas Jagged-2 expression remained unaltered (Table 2).
Treatment with rWnt5a stimulated the proliferation of cultured NHKs, as demonstrated by BrdU-incorporation assays. The percentage of BrdU-positive NHKs increased to 110.1% and 126.0% after treatment with rWnt5a for 2 and 3 d, respectively (Fig. 3A). The number of cells decreased to 82.8% and 43.0%, respectively, 2 and 3 d after transfection with siRNAs against Wnt5a (Fig. 3B).
Treatment of NHKs with Wnt5a did not induce keratinocyte migration, as assessed using Boyden chamber assays. However, treatment of HDFs with conditioned media from Wnt5a-treated NHKs induced a 1.4-fold increase in the migration of HDFs (Fig. 4).
To model the effects of a proinflammatory cytokine environment on Wnt5a expression in keratinocytes, we stimulated NHKs with TNF-α, IFN-γ, TGF-α, and IL-1α for 24 h. This decreased the Notch1 and HES1 protein levels while increasing that of Wnt5a (Fig. 5A). Moreover, ELISAs demonstrated that treatment of NHKs with rWnt5a increased the levels of TNF-α and IL-1α in the supernatant compared with that in controls (Fig. 5B). Notch1 and HES1 were downregulated in rWnt5a-treated NHKs, as demonstrated by RT-PCR (Fig. 5C).
Silver staining and western blotting with anti-phosphorylated-AKT (p-AKT) antibody revealed the presence of a 50 kDa tyrosine-phosphorylated protein (consistent with the molecular weight of AKT) in NHKs infected with Wnt5a adenovirus (data not shown). This suggested that intracellular AKT signaling was substantially activated in the Wnt5a-overexpressing cells. To rule out possible signaling effects due to adenoviral infection alone, we tested the effects of exogenously applied rWnt5a. Similar to the results obtained with adenoviral-mediated Wnt5a infection, western blot analyses showed an increase in p-AKT in the rWnt5a-treated cells (Fig. 6).
Psoriasis is a chronic inflammatory disease accompanied by hyperproliferation of keratinocytes. Although the uncontrolled proliferation of keratinocytes has been much emphasized upon, deregulated differentiation is also an important aspect of psoriatic keratinocytes1. Proteins associated with early differentiation, including involucrin, are highly expressed in the early layers, whereas the expression of late-differentiation markers, including loricrin and filaggrin, are markedly diminished in psoriasis19. Psoriasis, therefore, involves immune-deregulated keratinocytes.
In the skin, the fate of keratinocytes-proliferation versus differentiation- is determined by the action of Notch versus p631520. Notch signaling is a "direct" determinant of keratinocyte growth arrest and entry into terminal differentiation in NHKs and skin21. Thus, the Notch signaling pathway is important for cell-cell communication, and is involved in gene regulatory mechanisms that control a number of cellular processes, including cell fate decisions, stem cell maintenance, proliferation, differentiation, and survival. Notch reporters undergo successive cleavages, leading to the release of their activated form, the Notch intracellular domain (NID). The NID translocates to the nucleus and forms a complex with DNA-binding proteins, thereby activating transcription2223. This interaction results in the expression of various target genes, including HES and HES-related repressor protein (HEY)24. A major downstream effector of Notch1 in human keratinocytes and fibroblasts is HES12526. Notch activation also upregulates early-differentiation markers such as keratin1 and involucrin21.
In normal skin, Notch1 is distributed in all the layers, while Notch2 is present only in the basal layer, though their ligands exhibit similar distribution patterns2527. Here, we found that Notch1 protein levels were decreased in lesional skin samples, together with decrease in both HES1 mRNA and protein. It is notable that we did not separate the epidermis and dermis of the psoriatic lesion. Therefore, dermal components including fibroblasts and vascular structures, might have countervailed the decrease in epidermal NOTCH1 mRNA in lesional skin2829. Immunohistochemical analyses further showed that expression of the Notch1 ligand, Delta-like-1, was also decreased in lesional psoriatic epidermis compared with that in both non-lesional and normal skin samples. Downregulation of Notch1 and HES1 in psoriatic epidermis suggests defective Notch1 signaling in psoriasis, which is likely attributable to deregulation of the normal keratinocyte differentiation process.
Wnt signaling directs growth and morphogenesis during embryonic development, and continues to regulate stem cell proliferation and differentiation throughout adult life. Classically, Wnt5a, a prototypical non-canonical Wnt family ligand, is involved in pathways other than Wnt/β-catenin signaling. It has been shown to promote proliferation of glioblastoma cells, as well as adhesion of fibroblasts and breast cancer cells. Cheng et al.7 demonstrated that Wnt5a-mediated signaling regulates human endothelial cell proliferation and migration. Accumulating evidences also suggest the involvement of Wnt5a in IL-1α- and TNF-α-related innate immunity. Further, Wnt5a is induced by Toll-like receptors, eliciting the secretion of IL-12 and TNF-α in response to mycobacterial infection. Furthermore, Wnt5a is dramatically upregulated in macrophages by lipopolysaccharides and IFN-γ, and induces the production of the pro-inflammatory cytokines IL-1, IL-6, and IL-8. In fact, increased expression of Wnt5a might be a common mechanism underlying chronic inflammation4. Recent studies suggest a link between Wnt5a and psoriasis. Elevated Wnt5a expression has been reported in psoriatic plaques230. Importantly, expression profiling studies by Gudjonsson et al.4 provided evidence for altered Wnt signaling in psoriatic skin, demonstrating differential expression of several genes in the Wnt pathway in lesional psoriatic versus uninvolved skin. However, they could not prove the effect of Wnt5a on either cellular proliferation or differentiation and focused epidermal inflammation. Romanowska et al.3 reported that Wnt5a exhibits layer-specific expression in adult skin and is upregulated in psoriasis. They further demonstrated the synergy between Wnt5a and type 1 interferon as a pathomechanism underlying psoriasis.
Recent evidences suggest that canonical and non-canonical ligands act through cross-reactive pathways. Thus, it is likely that individual Wnt proteins activate multiple pathways, depending on the receptors expressed on the cell surface431. Initial insights into Wnt/Notch interactions were obtained from studies in Drosophila, which showed that Notch is required for wingless signaling in epidermal development32. Furthermore, in Wnt1-transformed mouse mammary epithelium cells, Notch signaling is upregulated and is required for expression of the tumorigenic phenotype33. In human skin, Wnt4, a canonical Wnt pathway ligand, has been shown to counteract Notch1 in NHKs. Conversely, Notch1 suppresses Wnt4 signaling, as evidenced by an increase in Wnt4 signaling in Notch1-knocked out mice15. Meanwhile, previous studies have also reported positive regulation between Wnt5a and Notch1 in different cell lines343536. These results conclude that Wnt ligands interact with Notch signaling through various pathways.
In order to elucidate the Wnt5a/Notch1 signaling pathway in psoriasis, we explored the crosstalk between Notch1 and Wnt5a, and sought to identify molecular targets in psoriasis. Interestingly, in vitro studies showed that rWnt5a treatment enhanced the migration of HDFs and enhanced the proliferation of both NHKs and HDFs. Unfortunately, it was not possible to study the effects of Wnt5a signal directly in these native cells, which were scant in the psoriatic dermis. However, we did find another evidence for the effect of Wnt5a signaling on cellular behavior and interaction. Importantly, treatment of NHKs with TNF-α, IL-1α, IFN-γ, or TGF-α increased Wnt5a and decreased Notch1 and HES1 levels. Treatment with rWnt5a substantially decreased Notch1 and HES1 and increased TNF-α and IL-1 levels. This corroborates the idea that Wnt5a is an important link between immunologic alterations and deregulation of psoriatic keratinocytes in the pathomechansim of the disease. The current data thus suggest that Wnt5a is responsible for abnormal proliferation and differentiation of keratinocytes as well as epidermal inflammation through negative regulation of Notch1 signaling. It is, therefore, an important player in the pathophysiology of psoriasis.
A recent study reported that transgenic mice overexpressing epidermal Wnt5a possessed normal, not psoriatic hyperproliferative epidermis37. Therefore, it is still unclear whether increased Wnt5a in psoriatic lesions is the causal pathomechanism or simply an attempt by the organism to restore normal skin phenotype2. Based on our results, we propose that Wnt5a participates in two pathomechanisms of psoriasis. The first relates to similarities between events associated with psoriatic keratinocytes and wound healing. Wnt5a is mostly involved in fibroblast activation, and we previously reported an association between Wnt5a upregulation and wound healing of laser-treated skin38. Proliferation and aberrant differentiation of psoriatic keratinocytes might involve modification of Wnt5a signaling in the psoriatic epidermis. In our study, the psoriatic epidermis showed increased and decreased expression of Wnt5a and Notch1, respectively. rWnt5a also stimulated the proliferation of NHKs in our study, and increased the levels of activated AKT.
The second possible pathomechanism involves the link between increased Wnt5a in psoriatic epidermis and inflammation. A recent study showed that genetic expression of Wnt5a did not return to the baseline after restoring psoriatic epidermis to normal-appearing skin. Although epidermal inflammation was relieved by >75% in treated psoriatic patients, dermal CD8+ cells were not fully resolved. These findings suggest that Wnt5a is associated with a network of cytokines and inflammatory responses in psoriasis39.
In conclusion, Wnt5a/Notch signaling serves a counterbalancing function in the pathomechanism of psoriasis. This study highlights the potential therapeutic application of Notch-enhancing and/or Wnt5a-inhibitory peptides in psoriasis. Further studies are warranted, including investigation of a wide range of cytokines affected by Wnt5a/Notch signaling and large-scale genetic studies of patient skin biopsy samples.
Figures and Tables
Fig. 1
Wnt5a expression in psoriasis. (A) Quantitative reverse transcription polymerase chain reaction results show that Wnt5a is upregulated approximately 2.4-fold in psoriatic lesional skin compared with that in non-lesional samples (n=10, paired samples). The box plot represents the log ratio of the expression levels in lesional and non-lesional psoriatic skin samples. Upper and lower borders of the boxes indicate 75th and 25th percentiles, respectively, whereas horizontal lines within boxes denote medians. (B) Immunohistochemical analyses show that Wnt5a is increased in all the layers of psoriatic lesions (left) compared with non-lesional skin (right). Samples were stained with DAB (3,3'-diaminobenzidine; brown color) and counterstained with hematoxylin (purple).

Fig. 2
Notch1 and hairy and enhancer of split (HES) 1 expression in psoriasis. (A) Quantitative reverse transcription polymerase chain reaction results did not yield any difference in the mRNA levels of NOTCH1 between psoriatic lesional and non-lesional skin samples, but revealed a decrease in HES1 mRNA expression in the former. (B) Immunohistochemical analyses (DAB [3,3'-diaminobenzidine] staining, ×400) showed that protein levels of Notch1 (upper panel) were decreased in all the layers of psoriatic lesions (left) compared with non-lesional skin samples (right). HES1 (lower panel) was also decreased at the protein level in psoriatic lesional samples.

Fig. 3
The effects of Wnt5a on normal human keratinocyte (NHK) proliferation. NHKs were treated with (A) rWnt5a (100 ng/ml) and (B) siRNAs against Wnt5a for 2 and 3 d, and then examined by Cell Proliferation Bromodeoxyuridine (BrdU) enzyme-linked immunosorbent assay. The graph shows the percentage of BrdU-positive cells with respect to the control. Data represent the mean±standard deviation. Con: control, d: day. *p-value<0.05, determined by the Student's t-test.

Fig. 4
Migration assays in neonatal normal human keratinocytes (NHKs) and human dermal fibroblasts (HDFs). NHKs (A) and HDFs (B) were cultured in either Wnt5a-conditioned or control media for 3 d, following which the cells (A: 1×106, B: 5×104) were incubated on transwell membranes (8-µm pore size) for 24 h. Migrated cells were stained with hematoxylin and eosin, and then enumerated under a microscope (×100).
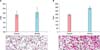
Fig. 5
Association of Wnt5a and Notch1 expression with the cytokine environment. (A) Neonatal normal human keratinocytes (NHKs) were stimulated with tumor necrosis factor (TNF)-α, interferon (IFN)-γ, transforming growth factor (TGF)-α, and interleukin (IL)-1α, either alone or with a cytokine mixture containing TNF-α, IFN-γ, TGF-α, and IL-1α for 24 h. Western blot analyses detected the expression of Notch1, hairy and enhancer of split (HES) 1, and Wnt5a. (1) Untreated NHKs (control), (2) IL-1α (10 ng/ml), (3) TNF-α (10 ng/ml), (4) TGF-α (24 ng/ml), (5) IFN-γ (20 ng/ml), (6) cytokine mixture of IL-1α (10 ng/ml), and TNF-α (10 ng/ml), (7) IL-1α (10 ng/ml), TNF-α (10 ng/ml), TGF-α (24 ng/ml), and IFN-γ (20 ng/ml)-treated NHKs. (B) Analysis of rWnt5a-treated NHK culture media by enzyme-linked immunosorbent assay (ELISA). Cells were treated with rWnt5a (100 ng/ml) for 6, 24, 48, and 72 h, following which the culture media were harvested. TNF-α and IL-1α production (pg/ml) were determined by ELISA. Data represent the mean±standard deviation. *p-value<0.05, **p-value<0.01; determined by the Student's t-test. (C) NHKs were treated with rWnt5a (100 ng/ml) for 1, 3, 6, 24, and 72 h, following which the expression levels of NOTCH1 and HES1 were determined by reverse transcription polymerase chain reaction.
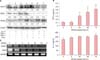
Fig. 6
Phospho (p)-AKT signaling in rWnt5a-treated neonatal normal human keratinocytes (NHKs). NHKs, serum-deprived for 24 h, were either treated with 100 ng/ml of rWnt5a or left untreated, and harvested after 15 min, 30 min, 1 h, and 3 h. Proteins in the cell lysates were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), and analyzed by western blotting using antibodies against p-AKT and total AKT. (A) Representative western blot for total AKT and p-AKT, showing an increase in the phosphorylated (activated) form of AKT in rWnt5a-treated NHKs. (B) Intensities of p-AKT and total AKT signals were determined using the BioRad VersaDoc Imaging System. The p-AKT/total AKT ratio is indicated for each time point.
Table 1
Primary antibodies used for immunohistochemistry
Table 2
Immunohistochemical scoring
ACKNOWLEDGMENT
This work was supported by the Korean Science Ministry NRF 2011-0009121, Asan Life Science Institute 2011-415; and a grant of Korean Health Technology R&D project, Ministry of Health & Welfare, Republic of Korea (A110564).
References
2. Reischl J, Schwenke S, Beekman JM, Mrowietz U, Stürzebecher S, Heubach JF. Increased expression of Wnt5a in psoriatic plaques. J Invest Dermatol. 2007; 127:163–169.

3. Romanowska M, Evans A, Kellock D, Bray SE, McLean K, Donandt S, et al. Wnt5a exhibits layer-specific expression in adult skin, is upregulated in psoriasis, and synergizes with type 1 interferon. PLoS One. 2009; 4:e5354.

4. Gudjonsson JE, Johnston A, Stoll SW, Riblett MB, Xing X, Kochkodan JJ, et al. Evidence for altered Wnt signaling in psoriatic skin. J Invest Dermatol. 2010; 130:1849–1859.

5. Nicolas M, Wolfer A, Raj K, Kummer JA, Mill P, van Noort M, et al. Notch1 functions as a tumor suppressor in mouse skin. Nat Genet. 2003; 33:416–421.

6. Devgan V, Mammucari C, Millar SE, Brisken C, Dotto GP. p21WAF1/Cip1 is a negative transcriptional regulator of Wnt4 expression downstream of Notch1 activation. Genes Dev. 2005; 19:1485–1495.

7. Cheng CW, Yeh JC, Fan TP, Smith SK, Charnock-Jones DS. Wnt5a-mediated non-canonical Wnt signalling regulates human endothelial cell proliferation and migration. Biochem Biophys Res Commun. 2008; 365:285–290.

8. Masckauchán TN, Agalliu D, Vorontchikhina M, Ahn A, Parmalee NL, Li CM, et al. Wnt5a signaling induces proliferation and survival of endothelial cells in vitro and expression of MMP-1 and Tie-2. Mol Biol Cell. 2006; 17:5163–5172.

9. Yu JM, Jun ES, Jung JS, Suh SY, Han JY, Kim JY, et al. Role of Wnt5a in the proliferation of human glioblastoma cells. Cancer Lett. 2007; 257:172–181.

10. Kawasaki A, Torii K, Yamashita Y, Nishizawa K, Kanekura K, Katada M, et al. Wnt5a promotes adhesion of human dermal fibroblasts by triggering a phosphatidylinositol-3 kinase/Akt signal. Cell Signal. 2007; 19:2498–2506.

11. Säfholm A, Leandersson K, Dejmek J, Nielsen CK, Villoutreix BO, Andersson T. A formylated hexapeptide ligand mimics the ability of Wnt-5a to impair migration of human breast epithelial cells. J Biol Chem. 2006; 281:2740–2749.

12. Blumenthal A, Ehlers S, Lauber J, Buer J, Lange C, Goldmann T, et al. The Wingless homolog WNT5A and its receptor Frizzled-5 regulate inflammatory responses of human mononuclear cells induced by microbial stimulation. Blood. 2006; 108:965–973.

13. Pereira C, Schaer DJ, Bachli EB, Kurrer MO, Schoedon G. Wnt5A/CaMKII signaling contributes to the inflammatory response of macrophages and is a target for the antiinflammatory action of activated protein C and interleukin-10. Arterioscler Thromb Vasc Biol. 2008; 28:504–510.

14. Mammucari C, Tommasi di Vignano A, Sharov AA, Neilson J, Havrda MC, Roop DR, et al. Integration of Notch 1 and calcineurin/NFAT signaling pathways in keratinocyte growth and differentiation control. Dev Cell. 2005; 8:665–676.

15. Nguyen BC, Lefort K, Mandinova A, Antonini D, Devgan V, Della Gatta G, et al. Cross-regulation between Notch and p63 in keratinocyte commitment to differentiation. Genes Dev. 2006; 20:1028–1042.

16. Lai EC. Notch signaling: control of cell communication and cell fate. Development. 2004; 131:965–973.

17. Okuyama R, Tagami H, Aiba S. Notch signaling: its role in epidermal homeostasis and in the pathogenesis of skin diseases. J Dermatol Sci. 2008; 49:187–194.

18. Thélu J, Rossio P, Favier B. Notch signalling is linked to epidermal cell differentiation level in basal cell carcinoma, psoriasis and wound healing. BMC Dermatol. 2002; 2:7.

19. Iizuka H, Takahashi H, Honma M, Ishida-Yamamoto A. Unique keratinization process in psoriasis: late differentiation markers are abolished because of the premature cell death. J Dermatol. 2004; 31:271–276.

20. Okuyama R, Ogawa E, Nagoshi H, Yabuki M, Kurihara A, Terui T, et al. p53 homologue, p51/p63, maintains the immaturity of keratinocyte stem cells by inhibiting Notch1 activity. Oncogene. 2007; 26:4478–4488.

21. Rangarajan A, Talora C, Okuyama R, Nicolas M, Mammucari C, Oh H, et al. Notch signaling is a direct determinant of keratinocyte growth arrest and entry into differentiation. EMBO J. 2001; 20:3427–3436.

22. Blaumueller CM, Qi H, Zagouras P, Artavanis-Tsakonas S. Intracellular cleavage of Notch leads to a heterodimeric receptor on the plasma membrane. Cell. 1997; 90:281–291.

23. Kim JE, Lee JH, Jeong KH, Kim GM, Kang H. Notch intracellular domain expression in various skin fibroproliferative diseases. Ann Dermatol. 2014; 26:332–337.

24. Collins BJ, Kleeberger W, Ball DW. Notch in lung development and lung cancer. Semin Cancer Biol. 2004; 14:357–364.

25. Okuyama R, Nguyen BC, Talora C, Ogawa E, Tommasi di Vignano A, Lioumi M, et al. High commitment of embryonic keratinocytes to terminal differentiation through a Notch1-caspase 3 regulatory mechanism. Dev Cell. 2004; 6:551–562.

26. Lefort K, Mandinova A, Ostano P, Kolev V, Calpini V, Kolfschoten I, et al. Notch1 is a p53 target gene involved in human keratinocyte tumor suppression through negative regulation of ROCK1/2 and MRCKalpha kinases. Genes Dev. 2007; 21:562–577.

27. Nickoloff BJ, Qin JZ, Chaturvedi V, Denning MF, Bonish B, Miele L. Jagged-1 mediated activation of notch signaling induces complete maturation of human keratinocytes through NF-kappaB and PPARgamma. Cell Death Differ. 2002; 9:842–855.

28. Ota T, Takekoshi S, Takagi T, Kitatani K, Toriumi K, Kojima T, et al. Notch signaling may be involved in the abnormal differentiation of epidermal keratinocytes in psoriasis. Acta Histochem Cytochem. 2014; 47:175–183.

29. Rooney P, Connolly M, Gao W, McCormick J, Biniecka M, Sullivan O, et al. Notch-1 mediates endothelial cell activation and invasion in psoriasis. Exp Dermatol. 2014; 23:113–118.

30. Zhou X, Krueger JG, Kao MC, Lee E, Du F, Menter A, et al. Novel mechanisms of T-cell and dendritic cell activation revealed by profiling of psoriasis on the 63,100-element oligonucleotide array. Physiol Genomics. 2003; 13:69–78.

31. van Amerongen R, Mikels A, Nusse R. Alternative wnt signaling is initiated by distinct receptors. Sci Signal. 2008; 1:re9.

32. Couso JP, Martinez Arias A. Notch is required for wingless signaling in the epidermis of Drosophila. Cell. 1994; 79:259–272.

33. Ayyanan A, Civenni G, Ciarloni L, Morel C, Mueller N, Lefort K, et al. Increased Wnt signaling triggers oncogenic conversion of human breast epithelial cells by a Notch-dependent mechanism. Proc Natl Acad Sci U S A. 2006; 103:3799–3804.

34. Ann EJ, Kim HY, Seo MS, Mo JS, Kim MY, Yoon JH, et al. Wnt5a controls Notch1 signaling through CaMKII-mediated degradation of the SMRT corepressor protein. J Biol Chem. 2012; 287:36814–36829.

35. Katoh M, Katoh M. Transcriptional mechanisms of WNT5A based on NF-kappaB, Hedgehog, TGFbeta, and Notch signaling cascades. Int J Mol Med. 2009; 23:763–769.
36. Koyanagi M, Bushoven P, Iwasaki M, Urbich C, Zeiher AM, Dimmeler S. Notch signaling contributes to the expression of cardiac markers in human circulating progenitor cells. Circ Res. 2007; 101:1139–1145.

37. Zhu X, Wu Y, Huang S, Chen Y, Tao Y, Wang Y, et al. Overexpression of Wnt5a in mouse epidermis causes no psoriasis phenotype but an impairment of hair follicle anagen development. Exp Dermatol. 2014; 23:926–928.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download