Abstract
This study demonstrates the ability of magnolol, a hydroxylated biphenyl compound isolated from Magnolia officinalis, to inhibit LPS-induced expression of iNOS gene and activation of NF-κB/Rel in RAW 264.7 cells. Immunohisto-chemical staining of iNOS and Western blot analysis showed magnolol to inhibit iNOS gene expression. Reporter gene assay and electrophoretic mobility shift assay showed that magnolol inhibited NF-κB/Rel transcriptional activation and DNA binding, respectively. Since p38 is important in the regulation of iNOS gene expression, we investigated the possibility that magnolol to target p38 for its anti-inflammatory effects. A molecular modeling study proposed a binding position for magnolol that targets the ATP binding site of p38 kinase (3GC7). Direct interaction of magnolol and p38 was further confirmed by pull down assay using magnolol conjugated to Sepharose 4B beads. The specific p38 inhibitor SB203580 abrogated the LPS-induced NF-κB/Rel activation, whereas the selective MEK-1 inhibitor PD98059 did not affect the NF-κB/Rel. Collectively, the results of the series of experiments indicate that magnolol inhibits iNOS gene expression by blocking NF-κB/Rel and p38 kinase signaling.
Go to : 
References
1. Wang JP, Hsu MF, Raung SL, Chen CC, Kuo JS, Teng CM. Anti-inflammatory and analgesic effects of magnolol. Naunyn Schmiedebergs Arch Pharmacol. 1992; 346:707–712.

2. Teng CM, Yu SM, Chen CC, Huang YL, Huang TF. EDRF-release and Ca2+-channel blockade by magnolol, an anti-platelet agent isolated from Chinese herb Magnolia officinalis, in rat thoracic aorta. Life Sci. 1990; 47:1153–1161.
3. Fujita S, Taira J. Biphenyl compounds are hydroxy radical scavengers: their effective inhibition for UV-induced mutation in Salmolella typhimurium TA102. Free Radic. Biol Med. 1994; 17:273–277.
4. Wang JP, Lin PL, Hsu MF, Chen CC. Possible involvement of protein kinase C inhibition in the reduction of phorbol ester-induced neutrophil aggregation by magnolol in the rat. J Pharm Pharmacol. 1998; 50:1167–1172.

5. Wang JP, Hsu MF, Raung SL, Chang LC, Tsao LT, Lin PL, Chen CC. Inhibition by agnolol of formylmethionyl-leucyl-phenylalanine-induced respiratory burst in rat neutrophils. J Pharm Pharmacol. 1999; 51:285–294.
6. Chen YH, Lin SJ, Chen JW, Ku HH, Chen YL. Magnolol attenuates VCAM-1 expression in vitro in TNF-a-treated human aortic endothelial cells and in vivo in the aorta of cholesterol-fed rabbits. Br J Pharmacol. 2002; 135:37–47.
7. Matsuda H, Kageura T, Oda M, Morikawa T, Sakamoto Y, Yoshikawa M. Effects of constituents from the bark of Magnolia obovata on nitric oxide production in lipopolysaccharide-activated macrophages. Chem Pharm Bull. 2001; 49:716–720.

8. Lee J, Jung E, Park J, Jung K, Lee S, Hong S, Park J, Park E, Kim J, Park S, Park D. Anti-inflammatory effects of magnolol and honokiol are mediated through inhibition of the downstream pathway of MEKK-1 in NF-kappaB activation signaling. Planta Med. 2005; 71:338–343.
10. Palmer RM, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature. 1988; 333:664–666.

11. Hibbs JB Jr, Taintor RR, Vavrin Z. Macrophage cytotoxicity: role for Larginine deiminase and imino nitrogen oxidation to nitrite. Science. 1987; 235:473–476.

12. Lowenstein CJ, Alley EW, Raval P, Snowman AM, Snyder SH, Russell SW, Murphy WJ. Macrophage nitric oxide synthase gene: two upstream regions mediate induction by interferon gamma and lipopolysaccharide. Proc Natl Acad Sci USA. 1993; 90:9730–9734.

13. Xie QW, Kashiwabara Y, Nathan C. Role of transcription factor NF-kappa B/Rel in induction of nitric oxide synthase. J Biol Chem. 1994; 269:4705–4708.

14. Raingeaud J, Gupta S, Rogers JS, Dickens M, Han J, Ulevitch RJ, Davis RJ. Pro-inflammatory cytokines and environmental stress cause p38 mitogen-activated protein kinase activation by dual phosphorylation on tyrosine and threonine. J Biol Chem. 1995; 270:7420–7426.

15. Lee JC, Young PR. Role of CSB/p38/RK stress response kinase in LPS and cytokine signaling mechanisms. J Leukoc Biol. 1996; 59:152–157.
16. Da Silva J, Pierrat B, Mary JL, Lesslauer W. Blockade of p38 mitogen-activated protein kinase pathway inhibits inducible nitric-oxide synthase expression in mouse astrocytes. J Biol Chem. 1997; 272:28373–28380.

17. Chen CC, Wang JK. p38 but not p44/42 mitogen-activated protein kinase is required for nitric oxide snthase induction mediated by lipopolysaccharide in RAW 264.7 cells. Mol Pharmacol. 1999; 55:481–488.
18. Jeon YJ, Yang KH, Pulaski JT, Kaminski NE. Attenuation of inducible nitric oxide synthase gene expression by delta 9-tetrahydrocannabinol is mediated through the inhibition of nuclear factor-kappa B/Rel activation. Mol Pharmacol. 1996; 50:334–341.
19. Xie H, Chiles TC, Rothstein TL. Induction of CREB activity via the surface Ig receptor of B cells. J Immunol. 1993; 151:880–889.
20. Jeon YJ, Han SH, Lee YW, Yea SS, Yang KH. Inhibition of NF-kappa B/Rel nuclear translocation by dexamethasone: mechanism for the inhibition of iNOS gene expression. Biochem Mol Biol Int. 1998; 45:435–441.
21. Ruppert J, Welch W, Jain AN. Automatic identification and representation of protein binding sites for molecular docking. Protein Sci. 1997; 6:524–533.

22. Dunnett CW. A multiple comparison procedure for comparing several treatments with a control. J Am Stat Assoc. 1955; 50:1096–1121.

23. Cuenda A, Rouse J, Doza YN, Meier R, Cohen P, Gallagher TF, Young PR, Lee JC. SB 203580 is a specific inhibitor of a MAP kinase homologue which is stimulated by cellular stresses and interleukin-1. FEBS Lett. 1995; 364:229–233.
24. Dudley DT, Pang L, Decker SJ, Bridges AJ, Saltiel AR. A synthetic inhibitor of the mitogen-activated protein kinase cascade. Proc Natl Acad Sci USA. 1995; 92:7686–7689.

25. Jeon YJ, Kim YG, Lee M, Park SM, Han SB, Kim HM. Radicicol suppresses expression of inducible nitric oxide synthase by blocking p38 kinase and nuclear factor-kB/Rel in lipopolysaccharide-stimulated macrophages. J Pharmacol Exp Ther. 2000; 294:548–554.
26. Foey AD, Parry S, Williams LM, Feldmann M, Foxwel lBM, Brennan FM. Regulation of monocyte IL-10 synthesis by endogenous IL-1 and TNF-a: Role of the p38 and p442/44 mitogen-activated protein kinases. J Immunol. 1998; 160:920–928.
27. Beyaert F, Cuenda A, Vanden Berghe W, Plaisance S, Lee JC, Haegeman G, Cohen P, Fiers W. The p38/RK mitogen-activated protein kinase pathway regulates interleukin-6 synthesis response to tumor necrosis factor. EMBO J. 1996; 15:1914–1923.

28. Chae HJ, Kim HK, Lee WK, Chae SW. Bolckade of p38 Mitogen-activated protein kinase pathway inhibits interleukin-6 release and expression in primary neonatal cardiomyocytes. Korean J Physiol Pharmacol. 2002; 6:319–325.
Go to : 
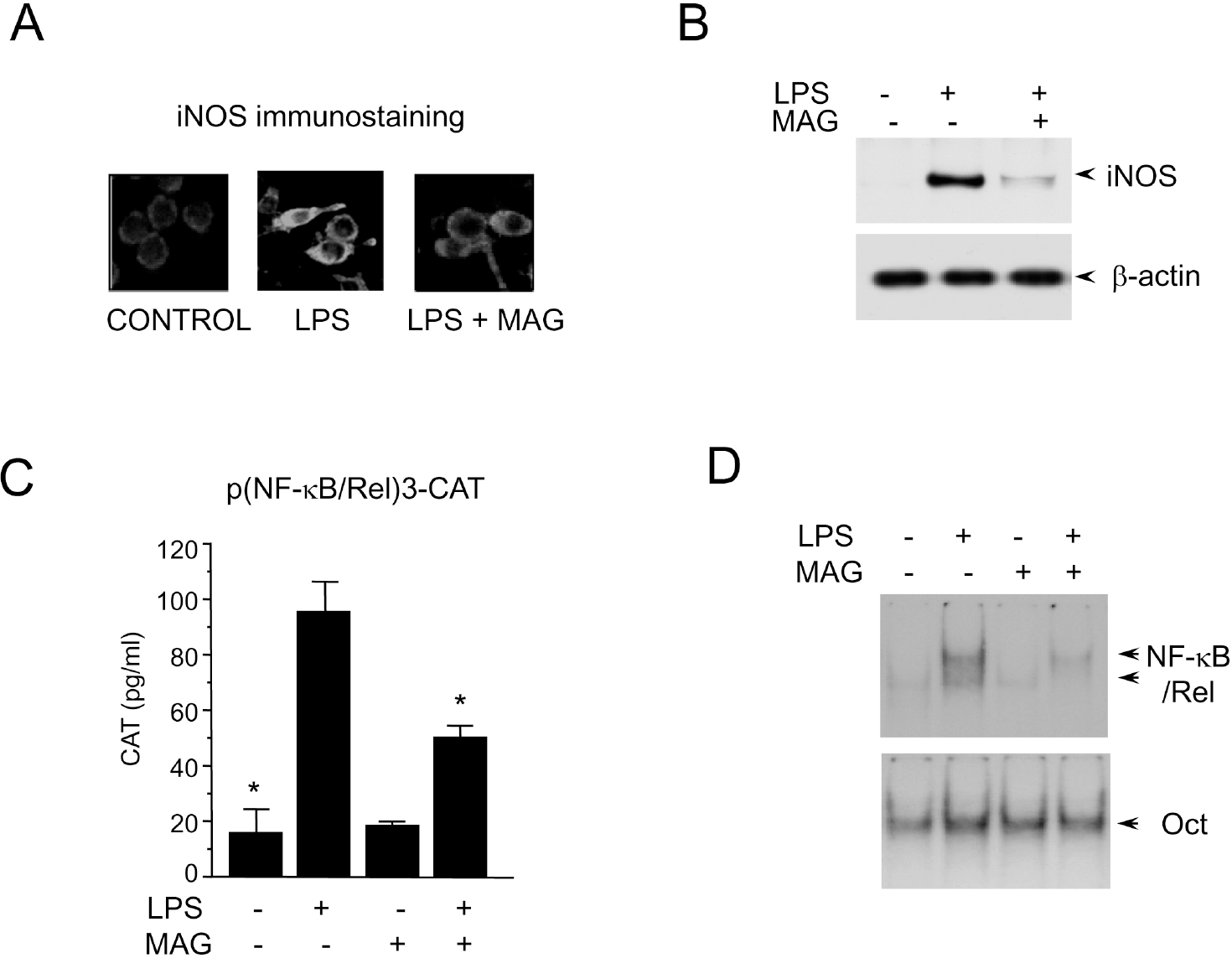 | Fig. 1.Inhibition of macrophage activation by magnolol. (A) RAW 264.7 cells (5×105 cells/ml) incubated with magnolol (50μM) in the presence of LPS (200 ng/ml) for 24 hr on cover slide in 12 well plates. Cells were subjected to immunohistochemical staining using an antibody specific for murine iNOS. Immunoreactivity of iNOS was localized along the margins of the cytoplasm in control group. (B) Cells were treated with magnolol in the presence of LPS (200 ng/ml) for 24 hr. Cell lysates were then prepared and subjected to Western immunoblotting. (C) RAW 264.7 cells were transfected with p(NF-κB/Rel)3-CAT by DEAE dextran method. Twenty-four hours after transfection, cells were treated with the magnolol in the presence or absence of LPS (200 ng/ml) for 18 hr. Cell extracts were then prepared and analyzed for the expression of CAT using CAT ELISA kit. (D) Cells (5×105 cells/ml) were incubated with magnolol (50μM) in the presence or absence of LPS (200 ng/ml) for 2 hr. Nuclear extracts (5μg/ml) were then isolated and analyzed for the activity of NF-κB/Rel and Oct. |
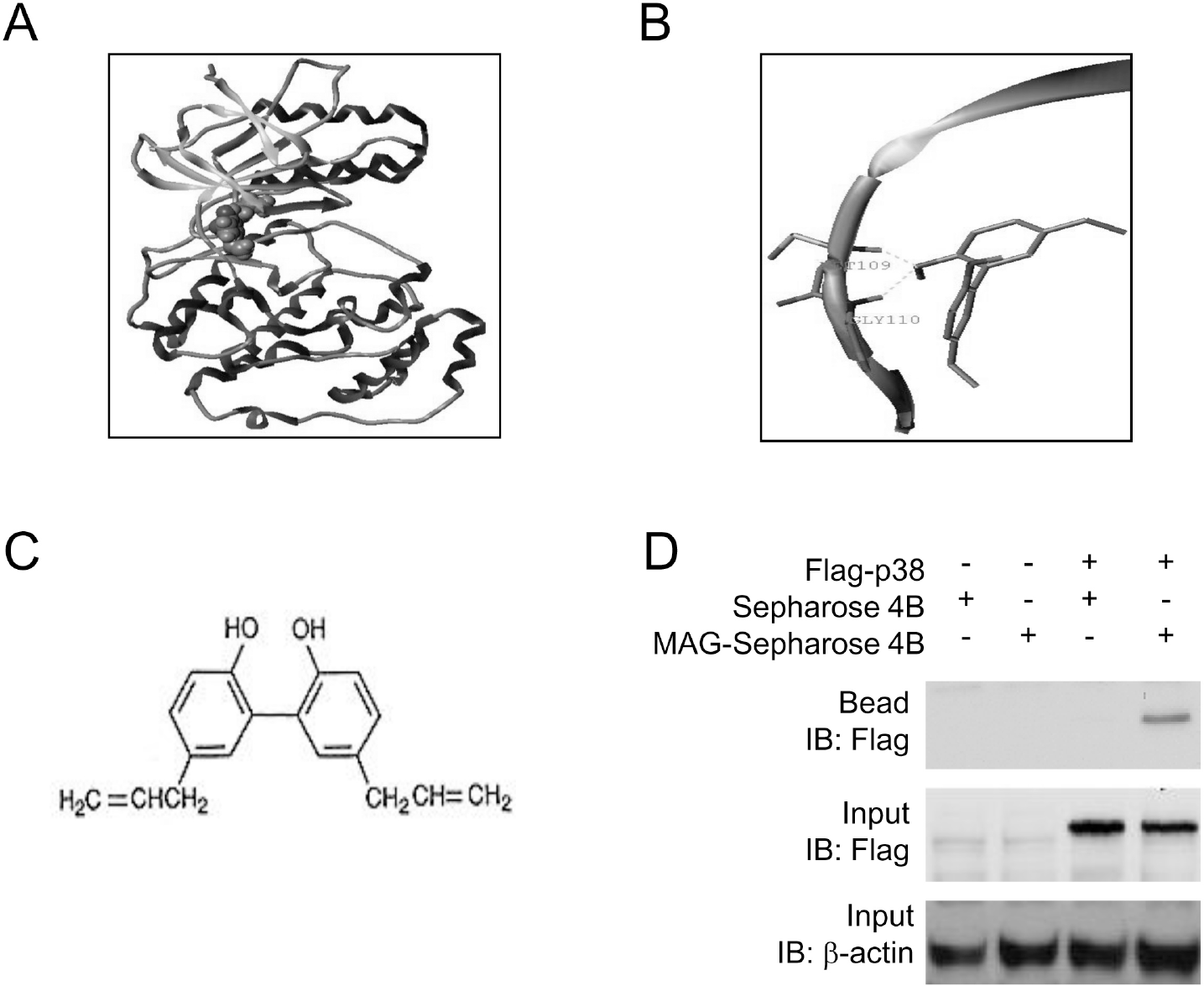 | Fig. 2.Molecular docking and pose generation. (A) A docking study was performed using SYBYL8.1 molecular modeling package as described in Materials and methods. Magnolol was docked with p38 kinase structure (PDB code: 3GC7). The ligand is represented in space fill model and the macromolecule is in ribbon and tube. (B) The proposed binding pose of Magnolol showed the interaction with the backbone of hinge residues M109 and G110. (C) Chemical structure of magnolol is shown. (D) Flag-p38 expression vectror was transiently transfected to 293 cells. Cell lysates were then prepared, used for pull-down assay using magnolol-Sepharose 4B beads and subjected to Western blot analysis. |
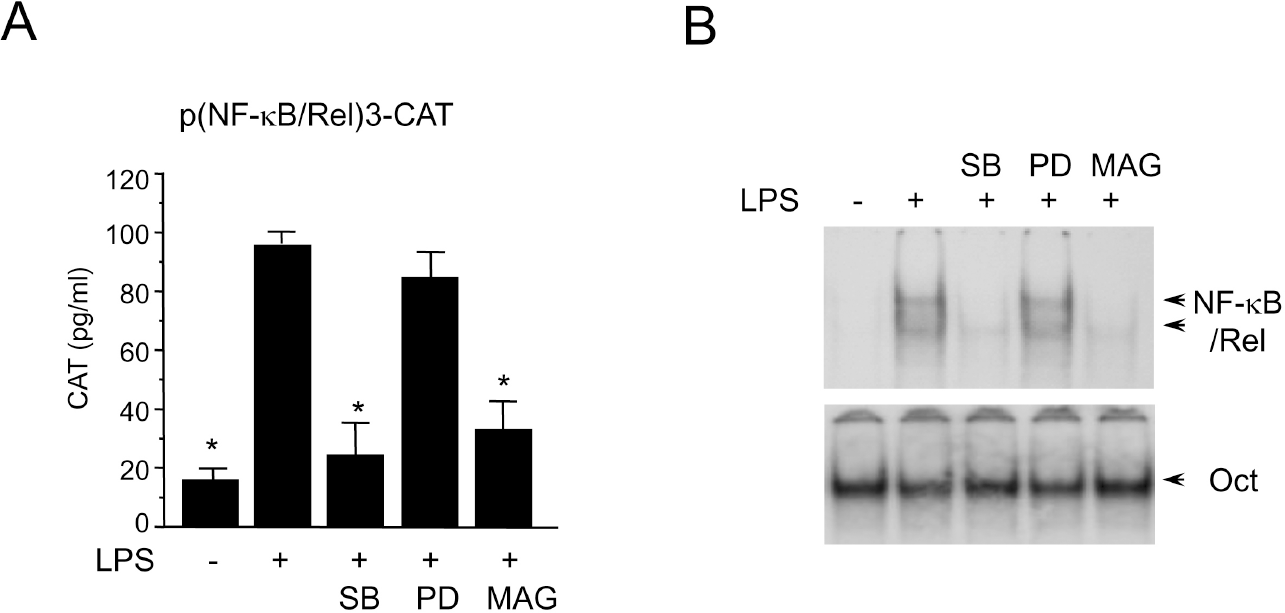 | Fig. 3.Effects of SB203580 and PD98059 on NF-κB/Rel activation in LPS-stimulated RAW 264.7 cells. (A) RAW 264.7 cells were transfected with p(NF-κB/Rel)3-CAT by DEAE dextran method. Twenty-four hours after transfection, cells were treated with SB203580 (30μM), PD98059 (50μM), or magnolol (50μM) in the presence of LPS (200 ng/ml) for 18 hr. Cell extracts were then prepared and analyzed for the expression of CAT using CAT ELISA kit. (B) RAW 264.7 cells were pretreated with SB203580 (30μM), PD98059 (50μM), or magnolol (50μM) for 30 min before incubation with LPS (200 ng/ml) for 2 hr. Nuclear extracts were then isolated and analyzed for the activity of NF-κB/Rel and Oct. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download