Abstract
Treatment of AR requires a stepwise approach depending on the severity and duration of symptoms. Treatment options for AR consist of allergen avoidance, pharmacotherapy, immunotherapy and surgery. For the mechanisms of AR, anti-IgE antibody and specific antibody to cytokines such as IL-4 or IL-5 that correlate with allergic inflammation have recently emerged. SLIT is currently widely used due to its efficacy, safety and convenience, which replaces subcutaneous immunotherapy. Although allergen avoidance and immunotherapy are theoretically ideal, antihistamines and intranasal corticosteroids will play the main role in the management of AR until an innovative treatment develops. However, patients' main symptom, the duration and severity of AR, patients' compliance, safety of medication and cost-effectiveness should be considered when treatment options are chosen. In conclusion, physicians should be aware of etiology, pathophysiology, symptoms, signs and diseases related to AR in order to make a correct diagnosis and choose a proper treatment option for each patient.
Allergic rhinitis (AR) is a symptomatic disorder of the nose induced after exposure to allergens via IgE-mediated hypersensitivity reactions, which are characterized by 4 cardinal symptoms of watery rhinorrhea, nasal obstruction, nasal itching and sneezing.1 The prevalence of AR is increasing all over the world. In the United States, AR is estimated to affect approximately 60 million peoples, and the prevalence is about 10-30% in adults and nearly 40% in children.2-4 In Korea, the prevalence of perennial AR was 3.39% according to the survey of 71,120 patients who visited the otolaryngology clinics of 23 tertiary referral centers between November 1999 and April 2000.5 According to the surveillance of 42,886 Koreans using the International Study of Asthma and Allergies in Childhood (ISSAC) questionnaire, 12-month prevalences of AR in elementary and middle school children (6-12 and 12-15 years) were 28.8% and 29.1%, respectively.6 AR is associated with an enormous economic burden causing problems in quality of life such as work/school performance and sleep. In the United States, direct cost for AR was 1.9 billion dollars in 1996.7 In another study, the direct cost for AR exceeded 3 billion dollars in 1996, and additional cost of 4 billion dollars resulted from comorbidities.8 Furthermore, as many studies on the relationship between AR and asthma have been reported, there has been a growing interest in the treatment of AR. Therefore, Allergic Rhinitis and Its Impact on Asthma (ARIA) published the guidelines for AR and revised them in 2008.9 The points of the ARIA guidelines are as follows: AR is subdivided by symptom duration and the severity of AR, a stepwise therapeutic approach is needed depending on the ARIA classification, and patients with persistent AR should be evaluated for asthma. There were some changes in the 2008 ARIA guidelines as compared to the 2001 guidelines: (1) intranasal corticosteroid became a first-line drug which was second-line drug in the 2001 guidelines, (2) second-generation antihistamines were preferred to first-generation one's, (3) leukotriene antagonists were entered into suggested drugs and (4) the role of immunotherapy was re-evaluated.
We review herein the pathophysiology, ARIA classification, diagnosis and treatment of AR, including recent update of AR.
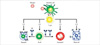
Antigen presenting cells (APCs), such as dendritic cells in the mucosal surface, process allergens and present some peptides from allergens on the major histocompatibility complex (MHC) class II molecule.10 This MHC class II molecule and antigen complex take a role as the ligand of T-cell receptors on Naive CD4+ T cells, which result in differentiation of Naive CD4+ T cells to allergen-specific Th2 cell. Activated Th2 cells secret several cytokines, which induce isotype switching of B cells to produce specific IgE and proliferation of eosinophils, mast cells and neutrophils (Fig. 1).11 Produced antigen-specific IgE binds to high-affinity IgE receptors on mast cells or basophils.
When AR patients are exposed to allergens, allergic reactions develop in 2 different patterns according to time sequence. One is the early reaction, in which sneezing and rhinorrhea develops in 30 minutes and disappears. The other is the late reaction, which shows nasal obstruction approximately 6 hours after exposure to allergens and subsides slowly. The early reaction is the response of mast cells to offending allergens (type I hypersensitivity). Stimulated mast cells induce nasal symptoms by secreting chemical mediators such as histamine, prostaglandins and leukotrienes.12 In contrast to the early reaction, eosinophil chemotaxis is the main mechanism in the late reaction, which is caused by chemical mediators produced in the early reaction. Several inflammatory cells, eosinophils, mast cells and T cells migrate to nasal mucosa, break up and remodel normal nasal tissue,13 and these processes result in nasal obstruction which is the main symptom of AR patients.
When respiratory epithelium is destroyed and nerve endings are exposed by cytotoxic proteins from eosinophils, sensory nerve fibers are excited by nonspecific stimuli and stimulate both sensory afferent and surrounding efferent fibers, the socalled retrograde axonal reflex. This makes the sensory nerve fibers secrete neuropeptides such as substance P and neurokinin A, which induce contraction of smooth muscles, mucous secretion of goblet cells and plasma exudation from capillaries. This process is called neurogenic inflammation.14
Non-specific hyperresponsiveness is one of the clinical characteristics of allergic inflammation. Due to eosinophilic infiltration and destruction of nasal mucosa, the mucosa becomes hyperactive to normal stimuli and causes nasal symptoms such as sneezing, rhinorrhea, nasal itching and obstruction.15 This is a non-immune reaction that is not related to IgE. Hypersensitivity to non-specific stimuli such as tobacco or cold and dry air as well as specific allergens increases in AR patients.
Epidemiologic studies have reported that almost all of the asthma patients have AR symptoms16,17 eosinophilic inflammation of nasal mucosa regardless of nasal symptoms.18 This characteristic finding of asthma is not found in patients with other pulmonary diseases and is the evidence that asthma is a systemic disease. The prevalence of asthma in AR patients has been reported to be from 10% up to 40%.5,16,19-21 Several clinical studies have revealed that although AR patients do not have asthma, they can have eosinophilic infiltration in bronchial mucosa.22-25 Madonini et al.26 have indicated that AR patients with positivity to pollens have bronchial hyperresponsiveness during the pollen season. Corren et al.27 have demonstrated that allergen stimulation to nasal mucosa in AR patients induces bronchial hyperresponsiveness. With cumulative of evidence for structural analog28,29 and the similarity of allergic inflammatory cells, inflammatory mediators and cytokines between upper and lower airways,13,30 the "one airway, one disease" concept has been introduced. This means that since AR and asthma are not separate disease entity, concurrent asthma in AR patients and concurrent AR in asthmatic patients should be indentified and both upper and lower airway allergy should be treated simultaneously. The ARIA (2008) recommends that asthma should be evaluated in moderate-severe persistent AR patients.
In the past, AR was subdivided by the kind of allergens into seasonal or perennial AR. The later is caused by indoor allergens such as house dust mites, cockroaches, animal dander or fungi, and the former by outdoor allergens, pollens. However, in some areas, pollens can induce perennial AR when patients are exposed to the pollens adhering to indoor carpet, furniture or bedclothes after the pollen season. In addition, perennial AR symptoms do not persist all around the year, and seasonal AR patients sensitized to multiple allergens may have rhinitis symptoms in all seasons. Furthermore, there are seasonal exacerbations in perennial AR patients when they are exposed to pollens. For these reasons, in 2001, ARIA suggested "intermittent" and "persistent" instead of "seasonal" and "perennial".1 Also, disease severity was classified as "mild" and "moderate-severe" considering its influence on work/school performance, daily activities and sleep (Fig. 2).
The diagnosis of AR is based on a typical history of allergic symptoms and diagnostic tests.9 When 2 or more symptoms out of watery rhinorrhea, sneezing, nasal obstruction and nasal pruritus persist for ≥1 hour on most days, AR is strongly suspected. In this situation, disease severity should be classified according to the ARIA guidelines and a confirmative diagnosis should be established by the skin prick test or the serum-specific IgE level. Unilateral nasal stuffiness, mucopurulent rhinorrhea, mucoid postnasal drip, pain, recurrent epistaxis or anosmia is usually not associated with AR.
Skin testing is the most important to find offending allergens. There are various testing methods including the scratch, prick/puncture, intradermal and patch tests. Among them, the skin prick test is usually recommended in clinical practice. False-positive or false-negative reactions are frequently evoked in skin tests, which means that positive reactions to specific allergens in skin tests does not always have a direct correlation with actual allergic reactions in the nasal cavity. There is controversy regarding the interpretation of the test results, and criteria for positivity are different among allergy clinics. Furthermore, skin tests have some problems. This test can be influenced by some drugs, particularly antihistamines, patients' age and test sites. If a patient has dermatologic disease, skin tests are difficult to perform. Despite these weak points, skin testing is regarded as the most important diagnostic method. A previous study on skin prick test results of 1,564 Korean AR patients reported that house dust mites was the most common allergen with a positive reactivity of 70%-80% (Table 1).31
Although the radioallergosorbent test (RAST) was the first method to detect serum-specific IgE, this test has not been widely used because it requires a radioactive isotope and expensive equipment and also because this test cannot detect multiple antibodies simultaneously. The next method is the multiple allergen simultaneous test (MAST). Since the MAST has some advantages over the RAST, it has been widely used. The MAST uses a photo reagent instead of a radioactive isotope, does not require expensive equipment and can detect multiple allergens simultaneously. This test is not affected by drugs such as antihistamines, is less invasive and can be adopted in patients with dermographism. One problem with the MAST is a low sensitivity as compared to the skin prick test. However, Finnerty et al.32 reported that the MAST shows 66.5% and 78.5% concordance rates when the criteria for positivity are ≥3 mm and ≥5 mm, respectively, and they recommended the MAST rather than skin tests. The capsulated hydrophilic carrier polymer (CAP) system is a more accurate in vitro test. Its procedure is similar to that of the MAST, but it uses a solid phase that has a high affinity to antigens. The CAP system can detect allergens more quantitatively than the MAST using antigens bound to a fine thread because antigens bind to the inner surface of sponge-like cellulose polymer bubbles.
Guerra et al.33 have reported that the severity of AR has a positive link with asthma and the risk of asthma incidence is 5 times higher in AR patients with elevated serum IgE. Silvestri et al.34 have pointed out that the eosinophil count of the nasal cavity is related to bronchial hyperresponsiveness and that the changes in the number and distribution of eosinophils after nasal mucosal challenge are also associated with bronchial hyperresponsiveness. They suggest that the local inflammation of AR can predict bronchial hyperresponsiveness. A domestic study of 83 pediatric AR patients and 32 normal children reported that prevalence of bronchial hyperresponsivenss was higher in AR patients than in control subjects (32.5% vs. 9.4%) and that persistent AR and parental asthma are closely related to bronchial hyperresponsiveness.35 In addition, it is known that bronchial hyperresponsiveness in AR patients is a predictor of asthma.36 It has generally recognized that the development and severity of asthma increase when a patient is sensitized to indoor allergens such as house dust mites or cat dander. The prevalence of asthma further increases in moderate to severe AR patients sensitized to both indoor and outdoor allergens.
Avoidance of indoor allergens including house dust mites is sometimes difficult. Therefore, few studies on avoidance of offending allergens have been conducted. For this reason, the 2001 ARIA guidelines classified the avoidance as evidence D.1 The 2008 ARIA guidelines have reported that there is the lack of evidence for effectiveness of avoidance of house dust mites or pet animal dander.9 However, a previous study has shown that cleaning with 60℃ hot water removes house dust mites and other allergens effectively as compared to 30℃ water (26.8% vs. 0.6%).37 Avoidance is mandatory for occupational AR. The European Academy of Allergy and Clinical Immunology (EAACI) stated that the safest and most effective treatment of occupational AR is the strict avoidance of offending allergens.38
The principle of pharmacological treatment is a stepwise approach according to the severity and duration (Fig. 3). The 2008 ARIA guidelines are different from the 2001 ARIA guidelines as follows: (1) leukotriene receptor antagonists can be used in all AR, (2) second-generation antihistamines are preferred to first-generation antihistamines and (2) topical steroids are regarded as the most effective drug for adult and pediatric AR patients.
First-generation antihistamines, which have been used since the early 1940s, have some side effects such as sedation, memory impairment and psychomotor dysfunction, which cause many problems in clinical practice. In contrast, second-generation antihistamines penetrate the blood-brain barrier much less than first-generation antihistamines, and thus they have few side effects on the central nervous system.39 Therefore, the 2008 ARIA guidelines recommended second-generation antihistamines rather than first-generation antihistamines. Oral antihistamines are effective in the treatment of rhinorrhea, sneezing, nasal itching and eye symptoms but less effective in nasal obstruction.40 Oral antihistamines have been reported to be safe and effective in children.41 Terfenadine and astemizole were initially used second-generation antihistamines. These drugs have severe cardiac toxicity inducing QT prolongation and torsade de pointes. When these antihistamines are administered along with macrolide antibiotics or azole antifungal agents, the risk of cardiac side effects is elevated because these drugs affect cytochrome p450 isoenzyme CYP3A4 activity. Therefore, terfenadine and astemizole have not been prescribed in many countries. Since ebastine is metabolized by CYP3A4, it can also induce such drug interactions theoretically. When high-dose ebastine (50 mg/kg/day) is given to guinea pigs, QT prolongation is observed in electrocardiography. When ebastine 20 mg/kg/day is administered with ketoconazole 400 mg/kg/day or erythromycin 2,400 mg/kg/day, QT intervals are prolonged up to 10 m sec with no clinical significance. Care should be taken in prescribing ebastine along with other drugs inhibiting CYP3A4 in patients with previous QT prolongation, liver failure or renal dysfunction.
Topical antihistamines have been reported to reduce itching, sneezing and rhiorrhea.42 However, they are less effective than intranasal corticosteroids and ineffective in eye symptoms.43 Intranasal azelastine twice a day can reduce the symptoms of seasonal AR patients who do not respond to oral antihistamines. They have some side effects such as mild sedation and metallic taste.44
Since intranasal corticosteroids are not absorbed systemically, they induce few systemic side effects. Steroid particles penetrate the cellular membrane and bind to cytoplasmic steroid receptors. The steroid-receptor complex is transferred to the nucleus and binds to the specific DNA site. The anti-inflammatory effect is induced by alteration in protein synthesis after binding of the steroid-receptor complex to DNA or by affecting other transcription factors. Intranasal corticosteroids inhibit both early and late reactions and reduce IgE production and eosinophilia by inhibiting the secretion of cytokines including IL-4, IL-5 and IL-13. When intranasal corticosteroids are administered, eosinophils and basophils decrease in 1 week.45 Intranasal corticosteroids are effective in all AR symptoms, especially nasal obstruction and eye symptoms.46 The therapeutic effect of intranasal corticosteroids is encountered 7 hours after administration47 and reaches the maximal level after 2 weeks.
Recently, budesonide, triamcinolone acetonide, fluticasone propionate, mometasone furoate and fluticasone furoate have been widely used. For a better choice of topical steroids, their pharmacological characteristics should be considered. Although these drugs have similar clinical effects, their systemic absorption rates are different. The systemic absorption rates of flunisolide, triamcinolone acetonide and beclomethasone dipropionate are 20-50%, whereas those of mometasone furoate and fluticasone propionate are very low (≤0.1% and ≤2%, respectively). In addition, most of the intranasal corticosteroids are eliminated by first-pass hepatic metabolism.
Currently prescribed intranasal corticosteroids are thought to be safe. Even when children aged 7-12 years use mometasone 200 µg or budesonide 400 µg for 2 weeks, their growth rates of the lower extremities are not significantly affected. Twelve-month use of beclomethasone dipropionate may cause growth retardation in children.48 However, 1-year mometasone or fluticasone therapy in children do not cause growth retardation.49,50
Intranasal corticosteroids usually improve the symptoms of patients with asthma. Watson et al.51 have documented that intranasal beclomethasone therapy reduces bronchial hyperresponsiveness and asthmatic symptoms in patient with AR and asthma. Foresi et al.52 have demonstrated that fluticasone propionate suppresses bronchial hyperresponsiveness in patients with seasonal AR.
The role of leukotrienes in allergic reactions is well known. The efficacy of LTRA has been demonstrated in asthma. Recently, some studies on the efficacy of LTRAs in AR patients have been reported. As previously mentioned, the 2008 ARIA guidelines re-evaluated the role of LTRAs. Interest in LTRAs has been increasing with the concept of "one airway, one disease", and therefore many studies on LTRAs are being conducted. Pranlukast (Onon®), montelukast (Singulair®) are commercially available. Care should be taken in the clinical use of Pranlukast which is metabolized by hepatic CYP3A4 enzymes because its serum concentration can be elevated when administered with terfenadine, astemizole, ketoconazole or erythromycin.
Montelukast is effective in reducing nasal and eye symptoms in patients with seasonal AR and improves nasal obstruction comparable to loratadine.9 The additive or synergic effect of montelukast and loratadine is controversial. Some previous studies have advocated that a combination of montelukast and loratadine has faster and a better efficacy than montelukast or loratadine alone,53 whereas others have not.54 Kurowski et al.55 reported that montelukast plus cetirizine which was administered 6 weeks before the pollen season effectively prevented the exacerbation of seasonal AR symptoms. The additive effect of LTRAs and antihistamines requires more investigations. Up to now, the pharmacological effects of LTRAs are estimated to be similar to those of antihistamines but less than those of intranasal corticosteroids in patients with seasonal AR.9
Omalizumab, an anti-IgE recombinant humanized monoclonal antibody, interferes with the interactions between mast cells/eosinophils and IgE by binding to free IgE and hence lowers serum free IgE.56 It also suppresses inflammatory reactions in blood or nasal mucosa57 and expression of FcεRI located on the surface of mast cells or eosinophils.58 Casale et al.59 have demonstrated that omalizumab pretreatment (300 mg) just before and during the pollen season for 12 weeks with 3-4 weeks intervals reduces AR symptoms significantly in patients with severe seasonal AR. Although there were some adverse reactions, they stated that the incidence of adverse effects of omalizumab such as headache, upper respiratory infection and sinusitis in the patient group is not significantly different from that of the placebo group. Urticaria may occur at the injection site, but it subsides spontaneously or with the administration of antihistamines. While anti-IgE antibody therapy appears to be helpful in severe asthma, it is controversial whether anti-IgE therapy is suitable as a treatment option for AR due to anaphylactic risk60 and high costs.
Immunotherapy is the only therapeutic option that modifies the basic allergic mechanism by inducing desensitization and producing an anergy state for offending allergens. Immunotherapy was initially introduced for seasonal AR due to pollens. At present, its indications have been extended to other allergic diseases due to hymenoptera, house dust mite, animal dander or fungi.61 Extracts of offending allergens are injected subcutaneously with increasing doses until a maintenance dose is reached. The maintenance dose is administered for ≥3 years. Although subcutaneous immunotherapy is a well-established treatment option, the risk of anaphylaxis has led to the development of other administration routes such as the oral, sublingual or nasal route. Sublingual immunotherapy (SLIT) has been used for 20 years in European countries because of its non-invasiveness, low incidence of adverse effects and convenience of self-administration. Recently, it has replaced subcutaneous immunotherapy. In Korea, sublingual immunotherapy for house dust mites was initiated in 2007.62
Taken together, immunotherapy is effective in house dust mite and pollen AR of adult and children, prevents asthma in AR patients and reduces new atopic sensitization. Its long-term effect after discontinuation of immunotherapy has been demonstrated. Herein we describe SLIT whose safety and efficacy have been proven.
Recently, many studies on immunologic changes after SLIT have been conducted. Regulation of antigen-specific responses (an increase in the IgG4/IgE ratio), inhibition of recruitment/activation of inflammatory cells, shift of Th2 to Th1 responses and activation of regulatory T cells are the main mechanisms of SLIT.63 Regulatory T cells are known to play a crucial role in immune tolerance, and they are related to the mechanism of SLIT. High-dose allergen extracts for SLIT induce regulatory T cells, which inhibit allergic inflammatory reactions by suppressing Th2 cells and producing IL-10 and TGF-β (Fig. 4).64
During the last 20 years, researches into SLIT have mainly been conducted in European countries. Although most studies have included a small sample size, meta-analyses of these studies have recently been published. A meta-analysis of 22 trials and 979 subjects showed that SLIT reduced the symptom score and medication frequency.65 In this meta-analysis, the symptom score and medication frequency were not improved in children. However, this study has some limitations due to the small sample size.66 Another meta-analysis of pediatric patients aged 4-18 years have suggested that SLIT reduces allergic symptoms and medication score.67 A previous study conducted in Korea reported that subjective symptoms assessed with a questionnaire were improved 6 months after SLIT, and 45% of patients were satisfied with SLIT.62
Like subcutaneous immunotherapy, SLIT reduces the incidence of asthma.68 A long-term follow-up study of pediatric patients with asthma and AR who were treated with SLIT for 4-5 years have pointed out that SLIT decreases the symptoms of asthma, medication frequency, and peak expiratory flow rate, and its efficacy is maintained for 4 to 5 years after discontinuation.69
SLIT has been shown to reduce sensitization to new allergens. In a previous study in 216 patients, it was found that 5.9% of patients in the SLIT group showed positive skin test results to new allergens, whereas 38% of patients in the control group, suggesting that SLIT could prevent sensitization to new allergens.70
SLIT is safer than subcutaneous immunotherapy. Common adverse events are local reactions (oral pruritus or swelling) and gastrointestinal problems (nausea, vomiting, diarrhea or abdominal pain), which subside spontaneously or with conservative management. Fatal adverse events causing death or severe sequelae have not yet been reported. Three cases of anaphylaxis after SLIT have been reported in the English language literatures: 2 of them were due to a mixture of multiple allergens and the remaining occurred during the treatment with latex allergen.71-73 However, anaphylaxis caused by commercially available allergens has not yet been reported. When 66 reports on SLIT were analyzed, there were no serious adverse effects during 1,181,654 administrations for 4,378 patients.74 Of the 66 reports, 41 reported adverse events: 1,047 adverse events occurred during 386,149 doses (2.7/1,000 doses). In 49 reports, 529 (12%) of 4,378 patients showed adverse events. Of 314,959 doses, 169 (0.056%) induced systemic side reactions and exacerbation of asthma occurred in 7 patients. In a previous study with 126 patients aged 3-5 years who underwent SLIT for allergic respiratory disease for ≥2 years, 9 adverse events were identified, which occurred during the dose escalation: 2 cases of oral pruritus, 1 case of mild abdominal pain and 6 cases of gastrointestinal problems, and all cases were successfully treated by decreasing doses.69 Therefore, SLIT is thought to be safe, even for children aged ≤5 years. Table 2 shows the level of evidence of different interventions in allergic rhinitis.
Allergic conjunctivitis is one of the most common accompanying diseases of AR. This is attributable to outdoor allergens such as pollens rather than indoor allergens. About 75% of AR patients complain of the symptoms of allergic conjunctivitis.
Influence of AR on the paranasal sinuses is not well understood. Many studies have suggested that allergic inflammation could affect acute or chronic rhinosinusitis. In AR patients due to ragweed, abnormal findings in computed tomography are found during the ragweed pollen season.75 A previous radiologic study using computed tomography has demonstrated that nasal allergen challenge can induce paranasal sinus inflammation.76 However, epidemiologic studies have suggested that the incidence of rhinosinusitis is not significantly higher in AR patients than in normal subjects. Therefore, it is not clear whether AR play a critical role in the development of rhinosinusitis. Antihistamines improve nasal symptoms such as sneezing or nasal obstruction in AR patients with acute rhinosinusitis.77
It is noteworthy that long-term use of nasal decongestants in AR patients may cause ciliary dysfunction with subsequent irreversible chronic hypertrophic rhinitis or sinusitis. In an animal study using rabbits, long-term administration of decongestants induced histologic and functional changes of cilia and resulted in ciliary dysfunction and rhinosinusitis.78
The relationship between nasal polyposis and allergy is not clear. It has been suggested that atopy itself may not contribute to the pathophysiology of nasal polyposis because the expression of IL-4, IL-5 and IFN-γ is not significantly different between nasal polyps of patients with seasonal AR and those with infectious rhinitis.79 Mechanisms involving edema and protrusion of nasal mucosa in nasal polyposis are similar to the pathophysiology of AR.80
Sensitization to inhalant allergens can alter immunological parameters of the adenoid. The numbers of CD1a+ Langerhans cells, eosinophils and IL-4 or IL-5-producing inflammatory cells increase in adenoid tissue of AR patients.81,82 The degree of adenoid hypertrophy does not seem to correlate with the presence of atopy.83 Although there have been few reports on the efficacy of antihistamines in AR patients with adenoid hypertrophy, intranasal corticosteroids are known to improve the symptoms of adenoid hypertrophy regardless of the presence of atopy. A recent study reported that the consecutive administration of short-term oral steroid and long-term antihistamines/intranasal corticosteroids reduce the size of adenoid and improve the symptoms of adenoid hypertrophy.84
Since the nasal mucosa is lined with the respiratory epithelium, allergic reactions due to inhalant allergens can occur in the eustachian tube. Therefore, the eustachian tube function of AR patients is impaired and otitis media with effusion occurs frequently, especially in children. It is controversial whether allergic inflammation in the eustachian tube is a reaction to local irritation or part of systemic reaction.
Although cognitive abilities in AR patient have not yet been studied, there is growing interest in this issue. Learning or cognitive problems should be considered as psychological complications of AR. AR patients do not concentrate on their school work due to sneezing, rhinorrhea or nasal itching. Marshall et al.86 reported that during ragweed season, allergic reactions to ragweed pollens cause a decrease in the speed of cognitive processing and difficulties in working memory in patients with ragweed AR. Symptomatic AR is related to poor school performance.87
Figures and Tables
 | Fig. 1Allergen-induced sensitization and inflammation.11
|
 | Fig. 2ARIA classification of severity of allergic rhinitis.1
|
 | Fig. 3Rhinitis management.9
|
 | Fig. 4The possible mechanism of action of sublingual immunotherapy.64
|
References
1. Bousquet J, Van Cauwenberge P, Khaltaev N. Allergic rhinitis and its impact on asthma. J Allergy Clin Immunol. 2001. 108:S147–S334.
2. Nathan RA. The burden of allergic rhinitis. Allergy Asthma Proc. 2007. 28:3–9.
3. Berger WE. Allergic rhinitis in children: diagnosis and management strategies. Paediatr Drugs. 2004. 6:233–250.
4. Settipane RA. Rhinitis: a dose of epidemiological reality. Allergy Asthma Proc. 2003. 24:147–154.
5. Min YG, Choi BY, Kwon SK, Lee SS, Jung YH, Kim JW, Oh SJ. Multicenter study on the prevalence of perennial allergic rhinitis and allergy-associated disorders. J Korean Med Sci. 2001. 16:697–701.
6. Lee SI, Shin MH, Lee HB, Lee JS, Son BK, Koh YY, Kim KE, Ahn YO. Prevalences of symptoms of asthma and other allergic diseases in korean children: a nationwide questionnaire survey. J Korean Med Sci. 2001. 16:155–164.
7. Reed SD, Lee TA, McCrory DC. The economic burden of allergic rhinitis: a critical evaluation of the literature. Pharmacoeconomics. 2004. 22:345–361.
8. Nash DB, Sullivan SD, Mackowiak J. Optimizing quality of care and cost effectiveness in treating allergic rhinitis in a managed care setting. Am J Manag Care. 2000. 6:S3–S15. quiz S19-20.
9. Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, Togias A, Zuberbier T, Baena-Cagnani CE, Canonica GW, van Weel C, Agache I, Ait-Khaled N, Bachert C, Blaiss MS, Bonini S, Boulet LP, Bousquet PJ, Camargos P, Carlsen KH, Chen Y, Custovic A, Dahl R, Demoly P, Douagui H, Durham SR, van Wijk RG, Kalayci O, Kaliner MA, Kim YY, Kowalski ML, Kuna P, Le LT, Lemiere C, Li J, Lockey RF, Mavale-Manuel S, Meltzer EO, Mohammad Y, Mullol J, Naclerio R, O'Hehir RE, Ohta K, Ouedraogo S, Palkonen S, Papadopoulos N, Passalacqua G, Pawankar R, Popov TA, Rabe KF, Rosado-Pinto J, Scadding GK, Simons FE, Toskala E, Valovirta E, van Cauwenberge P, Wang DY, Wickman M, Yawn BP, Yorgancioglu A, Yusuf OM, Zar H, Annesi-Maesano I, Bateman ED, Ben Kheder A, Boakye DA, Bouchard J, Burney P, Busse WW, Chan-Yeung M, Chavannes NH, Chuchalin A, Dolen WK, Emuzyte R, Grouse L, Humbert M, Jackson C, Johnston SL, Keith PK, Kemp JP, Klossek JM, Larenas-Linnemann D, Lipworth B, Malo JL, Marshall GD, Naspitz C, Nekam K, Niggemann B, Nizankowska-Mogilnicka E, Okamoto Y, Orru MP, Potter P, Price D, Stoloff SW, Vandenplas O, Viegi G, Williams D. World Health Organization. GA(2)LEN. AllerGen. Allergic Rhinitis and its Impact on Asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2)LEN and AllerGen). Allerg. 2008. 63:Suppl 86. 8–160.
10. Chaplin DD. 1. Overview of the human immune response. J Allergy Clin Immunol. 2006. 117:S430–S435.
11. Broide DH. The pathophysiology of allergic rhinoconjunctivitis. Allergy Asthma Proc. 2007. 28:398–403.
12. Prussin C, Metcalfe DD. 5. IgE, mast cells, basophils, and eosinophils. J Allergy Clin Immunol. 2006. 117:S450–S456.
13. Kay AB. Allergy and allergic diseases. Second of two parts. N Engl J Med. 2001. 344:109–113.
14. Togias A. Unique mechanistic features of allergic rhinitis. J Allergy Clin Immunol. 2000. 105:S599–S604.
15. Gerth van Wijk RG, de Graaf-in't Veld C, Garrelds IM. Nasal hyperreactivity. Rhinology. 1999. 37:50–55.
16. Linneberg A, Henrik Nielsen N, Frolund L, Madsen F, Dirksen A, Jorgensen T. The link between allergic rhinitis and allergic asthma: a prospective population-based study. The Copenhagen Allergy Study. Allergy. 2002. 57:1048–1052.
17. Montnemery P, Svensson C, Adelroth E, Lofdahl CG, Andersson M, Greiff L, Persson CG. Prevalence of nasal symptoms and their relation to self-reported asthma and chronic bronchitis/emphysema. Eur Respir J. 2001. 17:596–603.
18. Gaga M, Lambrou P, Papageorgiou N, Koulouris NG, Kosmas E, Fragakis S, Sofios C, Rasidakis A, Jordanoglou J. Eosinophils are a feature of upper and lower airway pathology in non-atopic asthma, irrespective of the presence of rhinitis. Clin Exp Allergy. 2000. 30:663–669.
19. Bousquet J, Annesi-Maesano I, Carat F, Leger D, Rugina M, Pribil C, El Hasnaoui A, Chanal I. Characteristics of intermittent and persistent allergic rhinitis: DREAMS study group. Clin Exp Allergy. 2005. 35:728–732.
20. Leynaert B, Neukirch C, Kony S, Guenegou A, Bousquet J, Aubier M, Neukirch F. Association between asthma and rhinitis according to atopic sensitization in a population-based study. J Allergy Clin Immunol. 2004. 113:86–93.
21. Downie SR, Andersson M, Rimmer J, Leuppi JD, Xuan W, Akerlund A, Peat JK, Salome CM. Association between nasal and bronchial symptoms in subjects with persistent allergic rhinitis. Allergy. 2004. 59:320–326.
22. Chakir J, Laviolette M, Boutet M, Laliberte R, Dube J, Boulet LP. Lower airways remodeling in nonasthmatic subjects with allergic rhinitis. Lab Invest. 1996. 75:735–744.
23. Djukanovic R, Lai CK, Wilson JW, Britten KM, Wilson SJ, Roche WR, Howarth PH, Holgate ST. Bronchial mucosal manifestations of atopy: a comparison of markers of inflammation between atopic asthmatics, atopic nonasthmatics and healthy controls. Eur Respir J. 1992. 5:538–544.
24. Brown JL, Behndig AF, Sekerel BE, Pourazar J, Blomberg A, Kelly FJ, Sandstrom T, Frew AJ, Wilson SJ. Lower airways inflammation in allergic rhinitics: a comparison with asthmatics and normal controls. Clin Exp Allergy. 2007. 37:688–695.
25. Marcucci F, Passalacqua G, Canonica GW, Frati F, Salvatori S, Di cara G, Petrini I, Bernini M, Novembre E, Bernardini R, Incorvaia C, Sensi LG. Lower airway inflammation before and after house dust mite nasal challenge: an age and allergen exposure-related phenomenon. Respir Med. 2007. 101:1600–1608.
26. Madonini E, Briatico-Vangosa G, Pappacoda A, Maccagni G, Cardani A, Saporiti F. Seasonal increase of bronchial reactivity in allergic rhinitis. J Allergy Clin Immunol. 1987. 79:358–363.
27. Corren J, Adinoff AD, Irvin CG. Changes in bronchial responsiveness following nasal provocation with allergen. J Allergy Clin Immunol. 1992. 89:611–618.
28. Togias A. Rhinitis and asthma: evidence for respiratory system integration. J Allergy Clin Immunol. 2003. 111:1171–1183. quiz 1184.
29. Igarashi Y, Goldrich MS, Kaliner MA, Irani AM, Schwartz LB, White MV. Quantitation of inflammatory cells in the nasal mucosa of patients with allergic rhinitis and normal subjects. J Allergy Clin Immunol. 1995. 95:716–725.
30. Holgate ST, Polosa R. The mechanisms, diagnosis, and management of severe asthma in adults. Lancet. 2006. 368:780–793.
31. Bang JH, Kim YJ, Shin HS, Lee BJ. Cinical analysis of allergic rhinitis in Seoul. J Rhinol. 1996. 3:130–134.
32. Finnerty JP, Summerell S, Holgate ST. Relationship between skin-prick tests, the multiple allergosorbent test and symptoms of allergic disease. Clin Exp Allergy. 1989. 19:51–56.
33. Guerra S, Sherrill DL, Martinez FD, Barbee RA. Rhinitis as an independent risk factor for adult-onset asthma. J Allergy Clin Immunol. 2002. 109:419–425.
34. Silvestri M, Battistini E, Defilippi AC, Sabatini F, Sale R, Pecora S, Rossi GA. Early decrease in nasal eosinophil proportion after nasal allergen challenge correlates with baseline bronchial reactivity to methacholine in children sensitized to house dust mites. J Investig Allergol Clin Immunol. 2005. 15:266–276.
35. Choi SH, Yoo Y, Yu J, Rhee CS, Min YG, Koh YY. Bronchial hyperresponsiveness in young children with allergic rhinitis and its risk factors. Allergy. 2007. 62:1051–1056.
36. Ferdousi HA, Zetterstrom O, Dreborg S. Bronchial hyper-responsiveness predicts the development of mild clinical asthma within 2 yr in school children with hay-fever. Pediatr Allergy Immunol. 2005. 16:478–486.
37. Choi SY, Lee IY, Shon JH, Lee YW, Shin SH, Lee DH, Kim PH, Yong TS, Hong CS, Park JW. Effect of steam-drum laundry machine for removal of allergens. J Asthma Allergy Clin Immunol. 2006. 26:289–296. Korean.
38. Moscato G, Vandenplas O, Van Wijk RG, Malo JL, Perfetti L, Quirce S, Walusiak J, Castano R, Pala G, Gautrin D, De Groot H, Folletti I, Yacoub MR, Siracusa A. European Academy of Allergology and Clinical Immunolgy. EAACI position paper on occupational rhinitis. Respir Res. 2009. 10:16.
39. Bousquet J, Van Cauwenberge P, Bachert C, Canonica GW, Demoly P, Durham SR, Fokkens W, Lockey R, Meltzer EO, Mullol J, Naclerio RM, Price D, Simons FE, Vignola AM, Warner JO. Requirements for medications commonly used in the treatment of allergic rhinitis. European Academy of Allergy and Clinical Immunology (EAACI), Allergic Rhinitis and its Impact on Asthma (ARIA). Allergy. 2003. 58:192–197.
40. Simons FE. Advances in H1-antihistamines. N Engl J Med. 2004. 351:2203–2217.
41. de Blic J, Wahn U, Billard E, Alt R, Pujazon MC. Levocetirizine in children: evidenced efficacy and safety in a 6-week randomized seasonal allergic rhinitis trial. Pediatr Allergy Immunol. 2005. 16:267–275.
42. McNeely W, Wiseman LR. Intranasal azelastine. A review of its efficacy in the management of allergic rhinitis. Drugs. 1998. 56:91–114.
43. Yanez A, Rodrigo GJ. Intranasal corticosteroids versus topical H1 receptor antagonists for the treatment of allergic rhinitis: a systematic review with meta-analysis. Ann Allergy Asthma Immunol. 2002. 89:479–484.
44. Berger WE, White MV. Efficacy of azelastine nasal spray in patients with an unsatisfactory response to loratadine. Ann Allergy Asthma Immunol. 2003. 91:205–211.
45. Lee BJ, Kim YJ, Kim JH, Shin HS, Chung YS. A comparative study of intranasal budesonide and oral terfenadine in perennial allergic rhinitics: effect on the symptom score and nasal secretion eosinophils. J Asthma Allergy Clin Immunol. 2001. 21:216–222. Korean.
46. Bhatia S, Baroody FM, deTineo M, Naclerio RM. Increased nasal airflow with budesonide compared with desloratadine during the allergy season. Arch Otolaryngol Head Neck Surg. 2005. 131:223–228.
47. Selner JC, Weber RW, Richmond GW, Stricker WE, Norton JD. Onset of action of aqueous beclomethasone dipropionate nasal spray in seasonal allergic rhinitis. Clin Ther. 1995. 17:1099–1109.
48. Skoner DP, Rachelefsky GS, Meltzer EO, Chervinsky P, Morris RM, Seltzer JM, Storms WW, Wood RA. Detection of growth suppression in children during treatment with intranasal beclomethasone dipropionate. Pediatrics. 2000. 105:E23.
49. Schenkel EJ, Skoner DP, Bronsky EA, Miller SD, Pearlman DS, Rooklin A, Rosen JP, Ruff ME, Vandewalker ML, Wanderer A, Damaraju CV, Nolop KB, Mesarina-Wicki B. Absence of growth retardation in children with perennial allergic rhinitis after one year of treatment with mometasone furoate aqueous nasal spray. Pediatrics. 2000. 105:E22.
50. Allen DB, Meltzer EO, Lemanske RF Jr, Philpot EE, Faris MA, Kral KM, Prillaman BA, Rickard KA. No growth suppression in children treated with the maximum recommended dose of fluticasone propionate aqueous nasal spray for one year. Allergy Asthma Proc. 2002. 23:407–413.
51. Watson WT, Becker AB, Simons FE. Treatment of allergic rhinitis with intranasal corticosteroids in patients with mild asthma: effect on lower airway responsiveness. J Allergy Clin Immunol. 1993. 91:97–101.
52. Foresi A, Pelucchi A, Gherson G, Mastropasqua B, Chiapparino A, Testi R. Once daily intranasal fluticasone propionate (200 micrograms) reduces nasal symptoms and inflammation but also attenuates the increase in bronchial responsiveness during the pollen season in allergic rhinitis. J Allergy Clin Immunol. 1996. 98:274–282.
53. Meltzer EO, Malmstrom K, Lu S, Prenner BM, Wei LX, Weinstein SF, Wolfe JD, Reiss TF. Concomitant montelukast and loratadine as treatment for seasonal allergic rhinitis: a randomized, placebo-controlled clinical trial. J Allergy Clin Immunol. 2000. 105:917–922.
54. Nayak AS, Philip G, Lu S, Malice MP, Reiss TF. Efficacy and tolerability of montelukast alone or in combination with loratadine in seasonal allergic rhinitis: a multicenter, randomized, double-blind, placebo-controlled trial performed in the fall. Ann Allergy Asthma Immunol. 2002. 88:592–600.
55. Kurowski M, Kuna P, Gorski P. Montelukast plus cetirizine in the prophylactic treatment of seasonal allergic rhinitis: influence on clinical symptoms and nasal allergic inflammation. Allergy. 2004. 59:280–288.
56. Holgate S, Casale T, Wenzel S, Bousquet J, Deniz Y, Reisner C. The anti-inflammatory effects of omalizumab confirm the central role of IgE in allergic inflammation. J Allergy Clin Immunol. 2005. 115:459–465.
57. Plewako H, Arvidsson M, Petruson K, Oancea I, Holmberg K, Adelroth E, Gustafsson H, Sandstrom T, Rak S. The effect of omalizumab on nasal allergic inflammation. J Allergy Clin Immunol. 2002. 110:68–71.
58. Beck LA, Marcotte GV, MacGlashan D, Togias A, Saini S. Omalizumab-induced reductions in mast cell Fce psilon RI expression and function. J Allergy Clin Immunol. 2004. 114:527–530.
59. Casale TB, Busse WW, Kline JN, Ballas ZK, Moss MH, Townley RG, Mokhtarani M, Seyfert-Margolis V, Asare A, Bateman K, Deniz Y. Omalizumab pretreatment decreases acute reactions after rush immunotherapy for ragweed-induced seasonal allergic rhinitis. J Allergy Clin Immunol. 2006. 117:134–140.
60. Price KS, Hamilton RG. Anaphylactoid reactions in two patients after omalizumab administration after successful long-term therapy. Allergy Asthma Proc. 2007. 28:313–319.
61. Cohen SG, Evans R 3rd. Allergen immunotherapy in historical perspective. Clin Allergy Immunol. 2004. 18:1–36.
62. Chang H, Han DH, Mo JH, Kim JW, Kim DY, Lee CH, Min YG, Rhee CS. Early compliance and efficacy of sublingual immunotherapy in patients with allergic rhinitis for house dust mites. Clin Exp Otorhinolaryngol. 2009. 2:136–140.
63. Potter PC. Update on sublingual immunotherapy. Ann Allergy Asthma Immunol. 2006. 96:S22–S25.
64. Frew AJ. Sublingual immunotherapy. N Engl J Med. 2008. 358:2259–2264.
65. Wilson DR, Lima MT, Durham SR. Sublingual immunotherapy for allergic rhinitis: systematic review and meta-analysis. Allergy. 2005. 60:4–12.
66. Pajno GB, Morabito L, Barberio G, Parmiani S. Clinical and immunologic effects of long-term sublingual immunotherapy in asthmatic children sensitized to mites: a double-blind, placebo-controlled study. Allergy. 2000. 55:842–849.
67. Penagos M, Compalati E, Tarantini F, Baena-Cagnani R, Huerta J, Passalacqua G, Canonica GW. Efficacy of sublingual immunotherapy in the treatment of allergic rhinitis in pediatric patients 3 to 18 years of age: a meta-analysis of randomized, placebo-controlled, double-blind trials. Ann Allergy Asthma Immunol. 2006. 97:141–148.
68. Novembre E, Galli E, Landi F, Caffarelli C, Pifferi M, De Marco E, Burastero SE, Calori G, Benetti L, Bonazza P, Puccinelli P, Parmiani S, Bernardini R, Vierucci A. Coseasonal sublingual immunotherapy reduces the development of asthma in children with allergic rhinoconjunctivitis. J Allergy Clin Immunol. 2004. 114:851–857.
69. Di Rienzo V, Marcucci F, Puccinelli P, Parmiani S, Frati F, Sensi L, Canonica GW, Passalacqua G. Long-lasting effect of sublingual immunotherapy in children with asthma due to house dust mite: a 10-year prospective study. Clin Exp Allergy. 2003. 33:206–210.
70. Marogna M, Tomassetti D, Bernasconi A, Colombo F, Massolo A, Businco AD, Canonica GW, Passalacqua G, Tripodi S. Preventive effects of sublingual immunotherapy in childhood: an open randomized controlled study. Ann Allergy Asthma Immunol. 2008. 101:206–211.
71. Dunsky EH, Goldstein MF, Dvorin DJ, Belecanech GA. Anaphylaxis to sublingual immunotherapy. Allergy. 2006. 61:1235.
72. Antico A, Pagani M, Crema A. Anaphylaxis by latex sublingual immunotherapy. Allergy. 2006. 61:1236–1237.
73. Eifan AO, Keles S, Bahceciler NN, Barlan IB. Anaphylaxis to multiple pollen allergen sublingual immunotherapy. Allergy. 2007. 62:567–568.
74. Cox LS, Larenas Linnemann D, Nolte H, Weldon D, Finegold I, Nelson HS. Sublingual immunotherapy: a comprehensive review. J Allergy Clin Immunol. 2006. 117:1021–1035.
75. Naclerio RM, deTineo ML, Baroody FM. Ragweed allergic rhinitis and the paranasal sinuses. A computed tomographic study. Arch Otolaryngol Head Neck Surg. 1997. 123:193–196.
76. Piette V, Bousquet C, Kvedariene V, Dhivert-Donnadieu H, Crampette L, Senac JP, Bousquet J, Demoly P. Sinus CT scans and mediator release in nasal secretions after nasal challenge with cypress pollens. Allergy. 2004. 59:863–868.
77. Braun JJ, Alabert JP, Michel FB, Quiniou M, Rat C, Cougnard J, Czarlewski W, Bousquet J. Adjunct effect of loratadine in the treatment of acute sinusitis in patients with allergic rhinitis. Allergy. 1997. 52:650–655.
78. Min YG, Kim HS, Suh SH, Jeon SY, Son YI, Yoon S. Paranasal sinusitis after long-term use of topical nasal decongestants. Acta Otolaryngol. 1996. 116:465–471.
79. Rhee CS, Min YG, Lee CH. Expression of IL-4, IL-5 and IFN-gamma mRNAs in patients with nasal polyps. Korean J Otolaryngol-Head Neck Surg. 1996. 39:1243–1248.
80. Alobid I, Benitez P, Valero A, Berenguer J, Bernal-Sprekelsen M, Picado C, Mullol J. The impact of atopy, sinus opacification, and nasal patency on quality of life in patients with severe nasal polyposis. Otolaryngol Head Neck Surg. 2006. 134:609–612.
81. Vinke JG, KleinJan A, Severijnen LW, Hoeve LJ, Fokkens WJ. Differences in nasal cellular infiltrates between allergic children and age-matched controls. Eur Respir J. 1999. 13:797–803.
82. Nguyen LH, Manoukian JJ, Sobol SE, Tewfik TL, Mazer BD, Schloss MD, Taha R, Hamid QA. Similar allergic inflammation in the middle ear and the upper airway: evidence linking otitis media with effusion to the united airways concept. J Allergy Clin Immunol. 2004. 114:1110–1115.
83. Cassano P, Gelardi M, Cassano M, Fiorella ML, Fiorella R. Adenoid tissue rhinopharyngeal obstruction grading based on fiberendoscopic findings: a novel approach to therapeutic management. Int J Pediatr Otorhinolaryngol. 2003. 67:1303–1309.
84. Georgalas C, Thomas K, Owens C, Abramovich S, Lack G. Medical treatment for rhinosinusitis associated with adenoidal hypertrophy in children: an evaluation of clinical response and changes on magnetic resonance imaging. Ann Otol Rhinol Laryngol. 2005. 114:638–644.
85. Tewfik TL, Mazer B. The links between allergy and otitis media with effusion. Curr Opin Otolaryngol Head Neck Surg. 2006. 14:187–190.
86. Marshall PS, O'Hara C, Steinberg P. Effects of seasonal allergic rhinitis on selected cognitive abilities. Ann Allergy Asthma Immunol. 2000. 84:403–410.
87. Walker S, Khan-Wasti S, Fletcher M, Cullinan P, Harris J, Sheikh A. Seasonal allergic rhinitis is associated with a detrimental effect on examination performance in United Kingdom teenagers: case-control study. J Allergy Clin Immunol. 2007. 120:381–387.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download