Abstract
The radioprotective activity of extracts from the red seaweed Callophyllis (C.) japonica was investigated in mice that underwent whole-body exposure to gamma radiation. A methanol extract of C. japonica and its fractions [hexane, ethyl acetate (EtOAc), butanol and the remaining H2O] were used. Each fraction (100 mg/kg body weight) was administered intraperitoneally (i.p.) 2 times into the BALB/c mice, once at 1 and once at 24 h before exposure to 9 Gray (Gy) of gamma radiation. Pre-irradiation administration of the hexane and EtOAc fractions saved the mice, with their survival rates being greater than 80% at 30 days post-irradiation; the mice that were pretreated with the other fractions showed survival rates lower than 20% over the same time period. To examine the effect of each C. japonica fraction on the survival of intestinal and bone marrow stem cells, the number of intestinal crypts and bone marrow cells in the gamma-irradiated mice were examined. Pre-treatment of mice (i.p., 100 mg/kg body weight at 1 and 24 h before irradiation) with the hexane or EtOAc fraction prior to 6-Gy irradiation significantly protected the number of jejunal crypts and bone marrow cells at 9 days after irradiation. These findings suggest that certain extracts from C. japonica, when they are administered prior to irradiation, play an important role in the survival of irradiated mice, and this is possibly due to the extracts protecting the hematopoietic cells and intestinal stem cells against gamma irradiation.
Radiation causes various pathophysiological alterations in living animals, and it causes death at high doses by multiple mechanisms, including direct DNA damage and indirect oxidative stress [4,7]. The search for useful radioprotectors has been an important issue in the field of radiation biology [9]. Ideal radioprotectors should have low toxicity and an extended window of protection [2,4]. As many synthetic compounds have toxic side effects, the natural products have attracted scientific attention as radioprotectors. Natural products that have been recently shown to be effective radioprotectors were found to have anti-oxidant and immunostimulant activities [2,3,8,12]. Thus, antioxidant and immunostimulant activities may play roles in protection against irradiation damage.
The red seaweed Callophyllis (C.) japonica is abundant in the coastal regions of Jeju Island in South Korea. A previous in vitro study showed that C. japonica extracts exhibit intracellular reactive oxygen species, 1,1-diphenyl-2-picrylhydrazyl radical-scavenging activity and lipid peroxidation inhibitory activity [3]. Furthermore, C. japonica has been demonstrated to be cytoprotective in CCl4-induced liver injury [10], and an ethanol extract of C. japonica has been shown to have protective effects on radiation-induced intestinal injury [6]. However, there's not much known about the in vivo radioprotective effects of C. japonica. This study investigated the effects of C. japonica on mice that were exposed to a sub-lethal dose of gamma radiation.
The C. japonica was collected during August 2006 from Jeju Island in Korea and the collected seaweed was identified by a taxonomist, Dr. Y.P. Lee, Cheju National University, Korea. The air-dried leaves of C. japonica (1,100 g) were powdered and extracted with 80% methanol (MeOH; Merck, Germany) at 95℃. The extract was filtered, evaporated to dryness under reduced pressure and then concentrated in vacuo. The lyophilized crude MeOH extract (65 g) was successively extracted with n-hexane (hexane; Junsei Chemical, Japan), ethyl acetate (EtOAc; Junsei Chemical, Japan), and n-butanol (BuOH; Junsei Chemical, Japan), to obtain the hexane (0.62 g), EtOAc (1.13 g), BuOH (1.5 g), and remaining water (H2O, 48.06 g) fractions; the extraction yields were 1%, 1.7%, 2.3% and 73.9% (w/w), respectively (Fig. 1).
Female BALB/c mice (6-8 weeks old; Orient Bio, Korea) were used in these experiments. Each extract from C. japonica was dissolved in phosphate-buffered saline and administered intraperitoneally at 24 and 1 h before irradiation (100 mg/kg body weight). After treatment, the mice were placed in a specially designed, well-ventilated acrylic container and they were subjected to whole-body irradiation at 6 or 9 Gray (Gy) in a single session with using a 60Co Y-ray source (10,000 Ci; C-188, Canada MDS Nordion; Co-60 Irradiation Facility, Applied Radiological Science Research Institute, Cheju National University, Korea), as was described in our previous reports [1,5,6]. All the experiments were conducted in accordance with the Guideline for the Care and Use of Laboratory Animals at Cheju National University, Korea.
Survival was monitored daily and this was reported as the percentage of animals that were still alive at 30 days after 9-Gy irradiation. The mice used for this study were divided into five groups: irradiation plus vehicle (control) and four treatment groups, one for each C. japonica extract.
To test the effect of C. japonica, the number of bone marrow cells was counted 9 days after the mice were irradiation with 6 Gy. Each treatment group consisted of five mice. Bone marrow cells were obtained from the anesthetized mice by aseptic isolation of the femurs, from which the marrow was flushed with Hank's balanced salt solution (HBSS; Invitrogen, USA) and using a 25-gauge needle. The cells were suspended in HBSS and then they were counted using a hemocytometer. The results are expressed as the number of live bone marrow cells (×106)/femur.
The jejunal crypt stem cell survival was determined with using the microcolony technique described by Withers and Elkind [14]. Each treatment group consisted of five mice. The mice were sacrificed 9 days after their irradiation (6 Gy). Two sections of four different parts of the jejunum from each animal were prepared for histological examination. The regenerating crypts in the jejunal cross-sections were then counted.
For the control mice that were given 9 Gy irradiation, 80% died by day 12, and all of them died before 15 days post-irradiation (Fig. 2). The mortality rate of the irradiated mice that were pre-treated with the remaining water-soluble extract was 60% at day 12 and 100% by day 15.
For the mice pre-treated with the BuOH, EtOAc, and hexane fractions prior to irradiation, 20, 80 and 100%, respectively, of the animals were alive at day 30. The mortality rates for the irradiated mice that were treated with the hexane and EtOAc extracts were significantly reduced compared with the mortality rate for the control group. These results suggest that the hexane and EtOAc fractions of C. japonica effectively decreased the radiation-induced mortality.
The number of bone marrow cells (Fig. 3) was significantly lower in the irradiation-only group (2.22 ± 0.31 × 106 cells/femur) than that in the normal control group (13.28 ± 1 ×106 cells/femur, p < 0.05). The numbers of bone marrow cells in the irradiation groups that had been pre-treated with the hexane and EtOAc extracts (6.06 ± 0.77 × 106 and 4.11± 0.74 × 106 cells/femur, respectively) were significantly higher than the number of bone marrow cells in the irradiation-only group (p < 0.05 for each extract). There was no significant protective effect on the numbers of bone marrow cells in the groups that were treated with the BuOH and remaining water-soluble fractions (2.93 ± 1.89 × 106 and 1.23 ± 0.27 × 106 cells/femur, respectively) compared with that of the irradiation-only group.
Fig. 4 shows the results of the jejunal crypt survival assay. The number of jejunal crypts was significantly lower in the irradiation-only group (81.25 ± 2.53) compared with the normal control group (104.57 ± 5.32, p < 0.05). The number of jejunal crypts in the hexane extract-treated mice with irradiation (88.36 ± 2.48) and in the EtOAc extract-treated group (101.13 ± 1.6) was significantly increased compared with the number of jejunal crypts in the irradiation-only controls (p < 0.05 for each extract). The number of jejunal crypts in mice that were pre-treated with the BuOH extract (78.55± 5.78) or the remaining water-soluble fraction (77.26 ±2.96) was not significantly different from the number of jejunal crypts in the irradiation-only control group.
Our study indicates that certain extracted fractions of C. japonica (the hexane and EtOAc fractions) provided protection against radiation-induced mortality. Moreover, our data shows that administration of the hexane or EtOAc fractions of C. japonica prior to irradiation reduced the decrease of bone marrow nucleated cells that was induced by radiation. The death of the irradiated animals was largely attributable to hematopoietic syndrome, which is characterized by a impaired bone marrow hematopoietic function, and this leads to leukopenia, erythropenia and thrombocytopenia [15]. Thus, administration of C. japonica reduced the mice's radiation-induced mortality, and it apparently did so by protecting the blood progenitor cells from the effects of irradiation.
The number of intestinal crypts is generally accepted as a good indicator of the protection of intestinal stem cells and/or the proliferation of surviving cells in animals that are recovering from exposure to radiation [8]. Stem cells in crypts of the small intestine are particularly vulnerable to radiation because of their high rate of proliferation [11,13]. The enhanced number of intestinal crypts in the C. japonica-treated/irradiated mice indicates that the C. japonica extracts protected the stem cells or the extracts stimulated the proliferation of the surviving cells. Thus, we suggest that treatment with C. japonica extract prior to irradiation protected the intestinal stem cells, and so this resulted in an increased survival rate.
The effects of C. japonica extracts in whole-body irradiated animals are not fully understood, but one possible mechanism involves their antioxidant properties. An extract of C. japonica exhibited scavenging activity toward intracellular reactive oxygen species and 1,1-diphenyl-2-picrylhydrazyl radicals, it promoted cell viability, inhibited H2O2 production, inhibited apoptosis and enhanced the effects of antioxidant enzymes [3]. The molecular and cellular mechanisms for the radioprotective effects of C. japonica extracts remain to be determined.
In conclusion, our results suggest that the administration of C. japonica extract to mice prior to irradiation increased their survival rate and this increased survival rate was associated with the protection of hematopoietic and intestinal stem cells.
Figures and Tables
 | Fig. 2The effect of each extract of Callophyllis japonica (CJ) on the survival of mice exposed to irradiation (9 Gy). Each extract was administered intraperitoneally twice, once at 24 and once at 1 h before irradiation (IR). The data is expressed as percentage of surviving mice. |
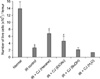 | Fig. 3The effect of each extract of Callophyllis japonica (CJ) on the bone marrow cellularity in the radiation-treated mice. Pre-treatment with the hexane and EtOAc extracts increased the bone marrow cellularity as compared with that in the irradiation (IR)-only group. Each C. japonica extract was administered intraperitoneally twice, once at 24 and once 1 h before irradiation. Hematopoietic stem cell assays were performed 9 days after gamma-irradiation of 6 Gy. The data is expressed as the mean ± SE. *p < 0.05 compared with the normal controls; #p < 0.05 compared with the irradiation-only group. |
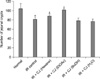 | Fig. 4The effect of each extract of Callophyllis japonica (CJ) on intestinal crypt survival in the radiation-treated mice. Pre-treatment with the hexane and EtOAc extracts increased the number of jejunal crypts as compared with the number of jejunal crypts in the irradiation (IR)-only group. Each C. japonica extract was administered intraperitoneally twice, once at 24 and once 1 h before irradiation. The jejunal crypt assays were performed 9 days after 6-Gy irradiation. The data is expressed as the mean ± SE. *p < 0.05 compared with the normal controls; #p < 0.05 compared with the irradiation-only group. |
Acknowledgments
This work was supported by a program of the Basic Atomic Energy Research Institute, which is a part of the Nuclear R&D Programs funded by the Ministry of Science & Technology, Korea.
References
1. Ahn M, Kim H, Kim JT, Lee J, Hyun JW, Park JW, Shin T. Gamma-ray irradiation stimulates the expression of caveolin-1 and GFAP in rat spinal cord: a study of immunoblot and immunohistochemistry. J Vet Sci. 2006. 7:309–314.

2. Hosseinimehr SJ. Trends in the development of radioprotective agents. Drug Discov Today. 2007. 12:794–805.

3. Kang KA, Bu HD, Park DS, Go GM, Jee Y, Shin T, Hyun JW. Antioxidant activity of ethanol extract of Callophyllis japonica. Phytother Res. 2005. 19:506–510.
4. Karbownik M, Reiter RJ, Qi W, Garcia JJ, Tan DX, Manchester LC, Vijayalaxmi . Protective effects of melatonin against oxidation of guanine bases in DNA and decreased microsomal membrane fluidity in rat liver induced by whole body ionizing radiation. Mol Cell Biochem. 2000. 211:137–144.
5. Kim H, Moon C, Kim J, Ahn M, Hyun JW, Park JW, Kim SH, Kim S, Shin T. Increased phosphorylation of caveolin-1 in the spinal cord of irradiated rats. J Vet Sci. 2007. 8:323–327.

6. Kim JT, Kim H, Shin T. Effect of ethanol extract of Callophyllis japonica in rats with irradiation. J Appl Radiol Sci Res Inst Cheju Natl Univ. 2006. 20:31–35.
7. Miura Y. Oxidative stress, radiation-adaptive responses, and aging. J Radiat Res. 2004. 45:357–372.

8. Moon C, Kim SH, Kim JC, Hyun JW, Lee NH, Park JW, Shin T. Protective effect of phlorotannin components phloroglucinol and eckol on radiation-induced intestinal injury in mice. Phytother Res. 2008. 22:238–242.

10. Park DS, Lee KH, Kim HC, Ahn MJ, Moon C, Ko MS, Lee KK, Go GM, Shin T. Effects of Callophyllis japonica powder on carbon tetrachloride-induced liver injury in rats. Orient Pharm Exp Med. 2005. 5:231–235.

11. Potten CS, Grant HK. The relationship between ionizing radiation-induced apoptosis and stem cells in the small and large intestine. Br J Cancer. 1998. 78:993–1003.

12. Schuchter LM. Guidelines for the administration of amifostine. Semin Oncol. 1996. 23:40–43.
13. Weil MM, Stephens LC, Amos CI, Ruifrok AC, Mason KA. Strain difference in jejunal crypt cell susceptibility to radiation-induced apoptosis. Int J Radiat Biol. 1996. 70:579–585.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download