Abstract
Purpose
Using transmission electron microscopy (TEM), we studied the ultrastructures of rapidly proliferating preretinal membranes of young patients with very extensive ischemic proliferative diabetic retinopathy and diabetes with uncontrollable blood sugar level.
Methods
Nine cases of preretinal membranes were obtained from six eyes of five patients with rapidly progressed proliferative diabetic retinopathy (mean age, 35 years) during vitrectomy. We obtained each preretinal membrane bimanually as one single sheet membrane using intraocular scissors and forceps. Each tissue was fixed in 3% glutaraldehyde in the operating room. All specimens were prepared and studied using TEM.
Results
The preretinal membranes were composed of blood vessels and some interstitial cells. The blood vessels within the preretinal membranes varied in developmental stages, from the immature stage to the mature stage. The blood vessels were highly active, in that primitive cells showed a large nucleus and prominent chromatin clumping with abundant cytoplasm. Highly active fibroblast-like cells were also noted.
References
1. Cardillo Piccolino F, Zingirian M, Mosci C. Classification of proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol. 1987; 225:245–50.
2. Clarkson JG, Green WR, Massof D. A histopathologic review of 168 cases of preretinal membrane. Am J Ophthalmol. 1977; 84:1–17.

3. Hamilton CW, Chandler D, Klintworth GK, Machemer R. A transmission and scanning electron microscopic study of surgically excised preretinal membrane proliferations in diabetes mellitus. Am J Ophthalmol. 1982; 94:473–88.

4. Taniguchi Y. Ultrastucture of newly formed blood vessels in diabetic retinopathy. Jpn J Ophthalmol. 1976; 20:19.
5. Taniguchi Y, Sameshima M, Fukiyama MM. Fine structure of retinal blood vessels in diabetic retiopathy. Nippon Ganka Gakkai Zasshi. 1972; 76:751–9.
6. Wallow IH, Geldner PS. Endothelial fenestrae in proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci. 1980; 19:1176–83.
7. Wallow IH, Greaser ML, Stevens TS. Actin filaments in diabetic fibrovascular preretinal membrane. Arch Ophthalmol. 1981; 99:2175–81.

8. Tripathi RC. Fine structure of mesodermal tissues of the human eye. Trans Ophthalmol Soc U K. 1974; 94:663–95.
9. Constable IJ, Tolentino FI, Donovan RH, et al. Clinicopathologic correlation of vitreous membranes. Pruett FC, Regan CD, editors. Retina Congress. New York: Appleton-Century-Crofts;1982. p. 245–57.
10. Tolentino MJ, Miller JW, Gragoudas ES, et al. Intravitreous injections of vascular endothelial growth factor produce retinal ischemia and microangiopathy in an adult primate. Ophthalmology. 1996; 103:1820–8.

11. Hofman P, van Blijswijk BC, Gaillard PJ, et al. Endothelial cell hy-pertrophy induced by vascular endothelial growth factor in the retina. Arch Ophthalmol. 2001; 119:861–6.

12. Merin S, Ber I, Ivry M. Retinal ischemia (capillary nonperfusion) in diabetic retinopathy of patients with and without systemic hypertension. Ophthalmologica. 1978; 177:76–81.

13. Avery RL, Pearlman J, Pieramici DJ, et al. Intravitreal bevacizumab (Avastin) in the treatment of proliferative diabetic retinopathy. Ophthalmology. 2006; 113:1695.e1–15.

14. Jorge R, Costa RA, Calucci D, et al. Intravitreal bevacizumab (Avastin) for persistent new vessels in diabetic retinopathy (IBEPE study). Retina. 2006; 26:1006–13.

15. Mirshahi A, Roohipoor R, Lashay A, et al. Bevacizumab-aug-mented retinal laser photocoagulation in proliferative diabetic retinopathy: A randomized double-masked clinical trial. Eur J Ophthalmol. 2008; 18:263–9.

16. Tonello M, Costa RA, Almeida FP, et al. Panretinal photocoagulation versus PRP plus intravitreal bevacizumab for high-risk proliferative diabetic retinopathy (IBeHi study). Acta Ophthalmol. 2008; 86:385–9.

17. Rizzo S, Genovesi-Ebert F, Di Bartolo E, et al. Injection of intravitreal bevacizumab (Avastin) as a preoperative adjunct before vitrectomy surgery in the treatment of severe PDR. Graefes Arch Clin Exp Ophthalmol. 2008; 246:837–42.
Figure 1.
A 39-year-old male had diabetes mellitus for 15 years. Very extensive capillary nonperfusion is observed on the fundus fluorescein angiography.
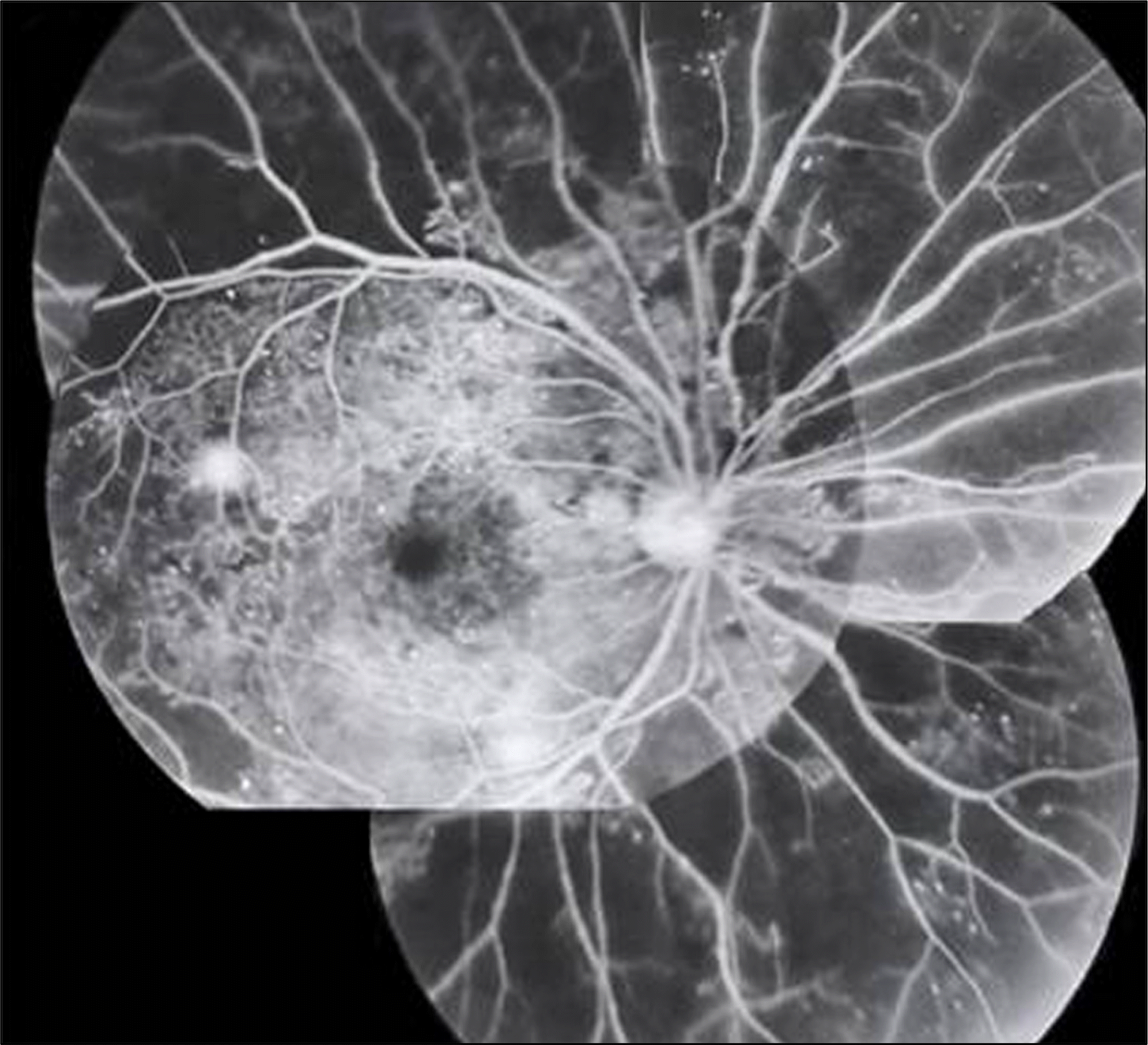
Figure 2.
A 38-year-old male had had diabetes mellitus for 8 years. (A, B) In the left eye, very extensive capillary non-perfusion was observed in fundus fluorescein angiography. (C) There was mild vitreous hemorrhage and fibrovascular membrane in superotemporal macular area in left eye. Instant panretinal argon laser photocoagulation was performed. (D) After 4 months, despite of successful laser application, the more dense fibrovascular membrane and severe vitreous hemorrhage were observed on the entire macula. A pars plana vitrectomy and membrane peeling was performed.

Figure 3.
A 50-year-old male had diabetes mellitus for 10 years. (A, B) Extensive capillary nonperfusion with neovascularization was observed in the right eye in the initial fundus fluorescein angiography (A, B, C). Instant panretinal laser photocoagulation was performed. (D) After 2 months, despite of complete and favorable laser application, extensive preretinal hemorrhage has occurred over the entire macula in the right eye. A pars plana vitrectomy was performed.

Figure 4.
A 46-year-old patient had had diabetes mellitus for 14 years. (A, B, C) In right eye, there was wide capillary nonperfusion in fluorescein angiography. (D) There were mild vitreous and preretinal hemorrhage and pre-retinal membrane in superior retina from optic nerve in right eye. Panretinal argon laser photocoagulation was performed. After 3 months, the preretinal membrane rapidly proliferated to entire macula. A pars plana vitrectomy was performed.
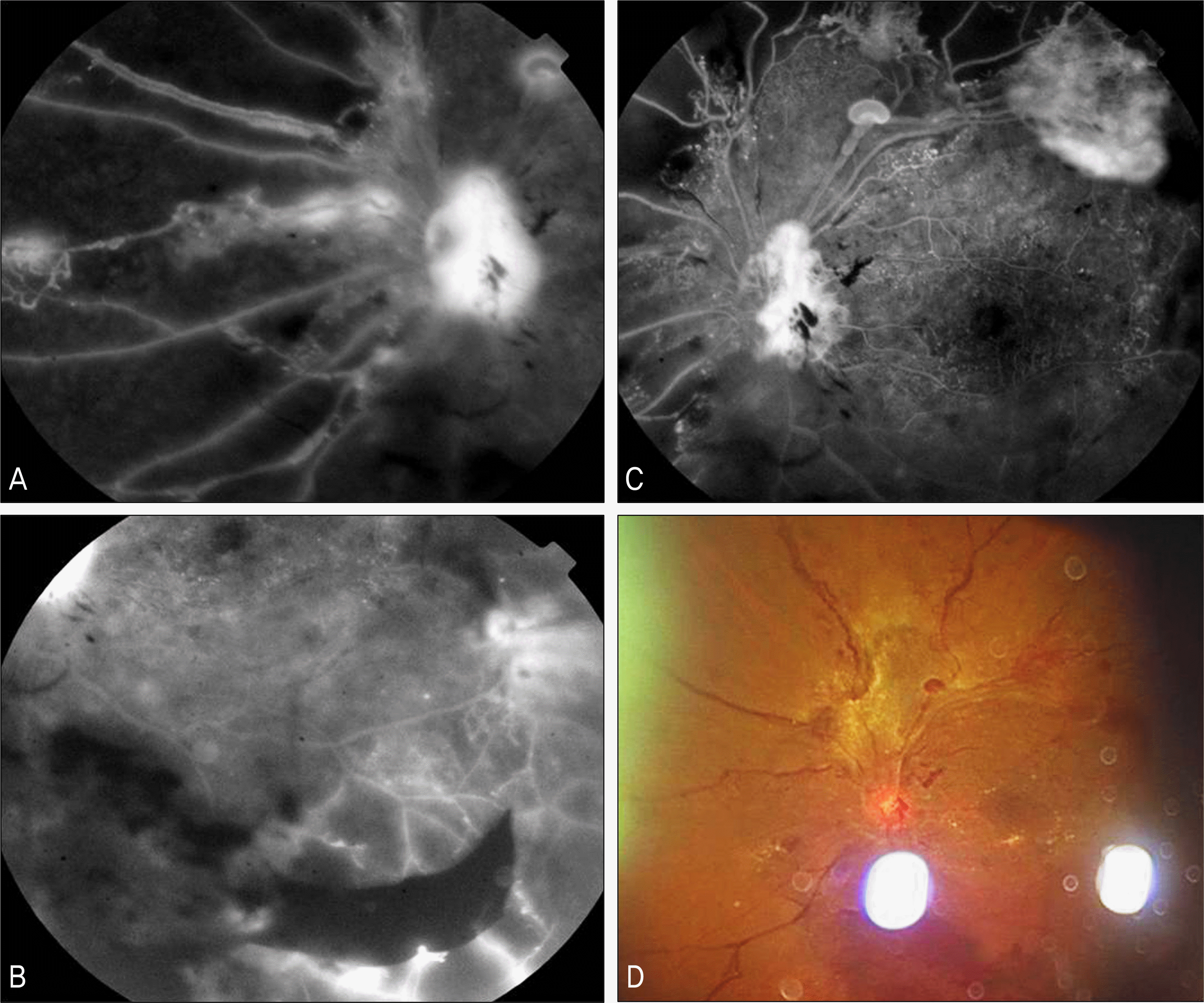
Figure 5.
A 38-year-old patient had had diabetes mellitus for 10 years. (A, B) There was wide capillary non-perfusion in fluorescein angiography. (C) There were severe vitreous hemorrhage and preretinal membrane in superotemporal retina from optic nerve in right eye. Panretinal argon laser photocoagulation was performed. (D) After 4 months, the preretinal membrane rapidly proliferated to entire macula. Pars plana vitrectomy was performed.

Figure 6.
Transmission electron micrograph of primitive cell. Presence of primitive cell (asterisk) within collagen fiber. The primitive cell has large nucleus and prominent chromatin clumping with scanty cytoplasm. Prominent villous processes are also observed (arrowhead). Bar indicates 2 μ m.
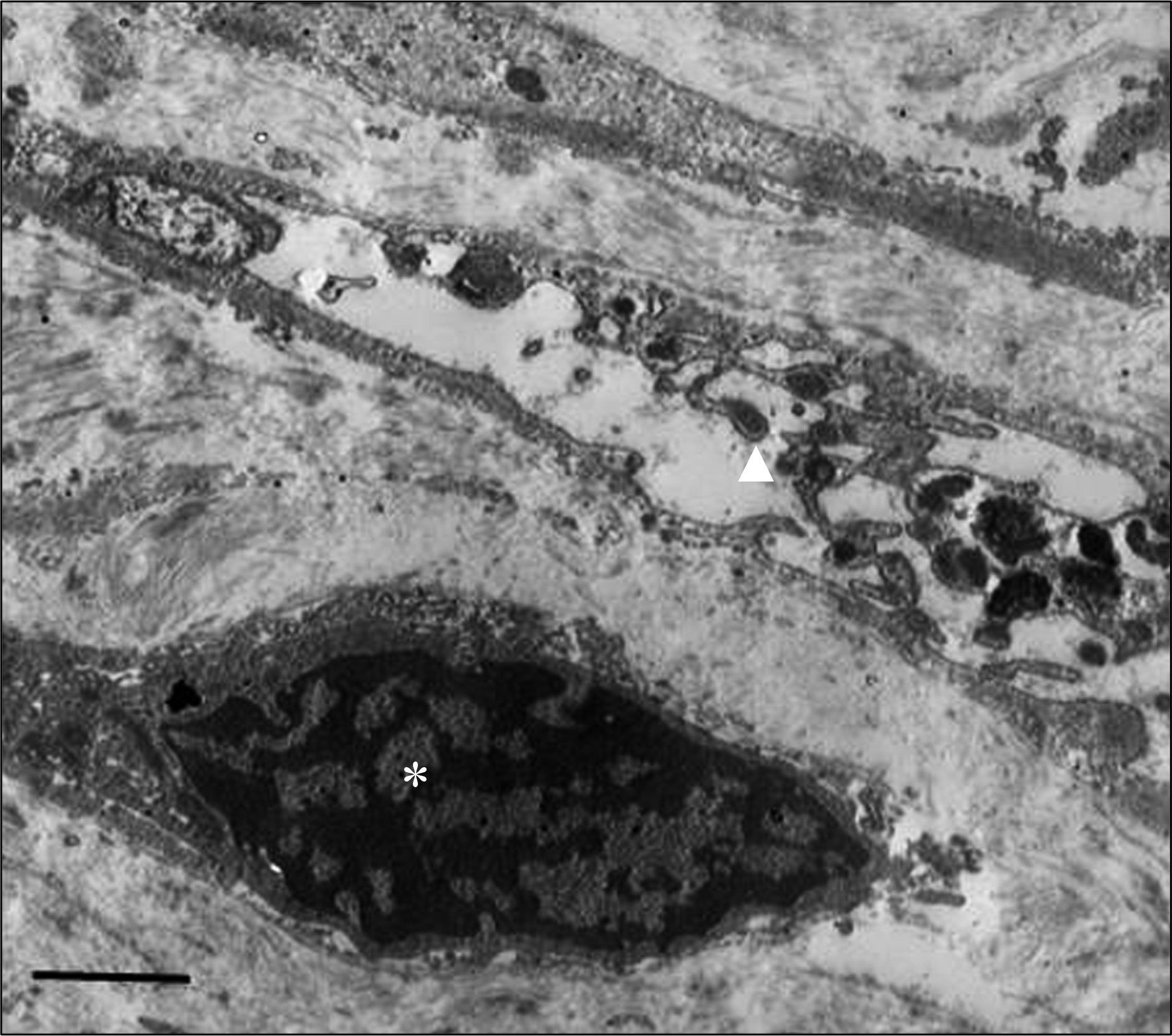
Figure 7.
Transmission electron micrograph showing primitive vascular cell. The primitive cells have large nucleus and chromatin clumping with scanty cytoplasm. The primitive cells form vascular sheet like structure (A). A lumen (L)-like space is also noted (B). The primitive vascular cells have varying stage of electron density. Bar indicates A: 4 μ m; B: 5 μ m.

Figure 8.
Transmission electron micrograph of more developed primitive vascular cells. The vascular cells composed of vascular pericyte (P), vascular endothelial cell (E) and basement membrane (arrowheads). Macrophage (M) is also noted. Bar indicates 5 μ m.
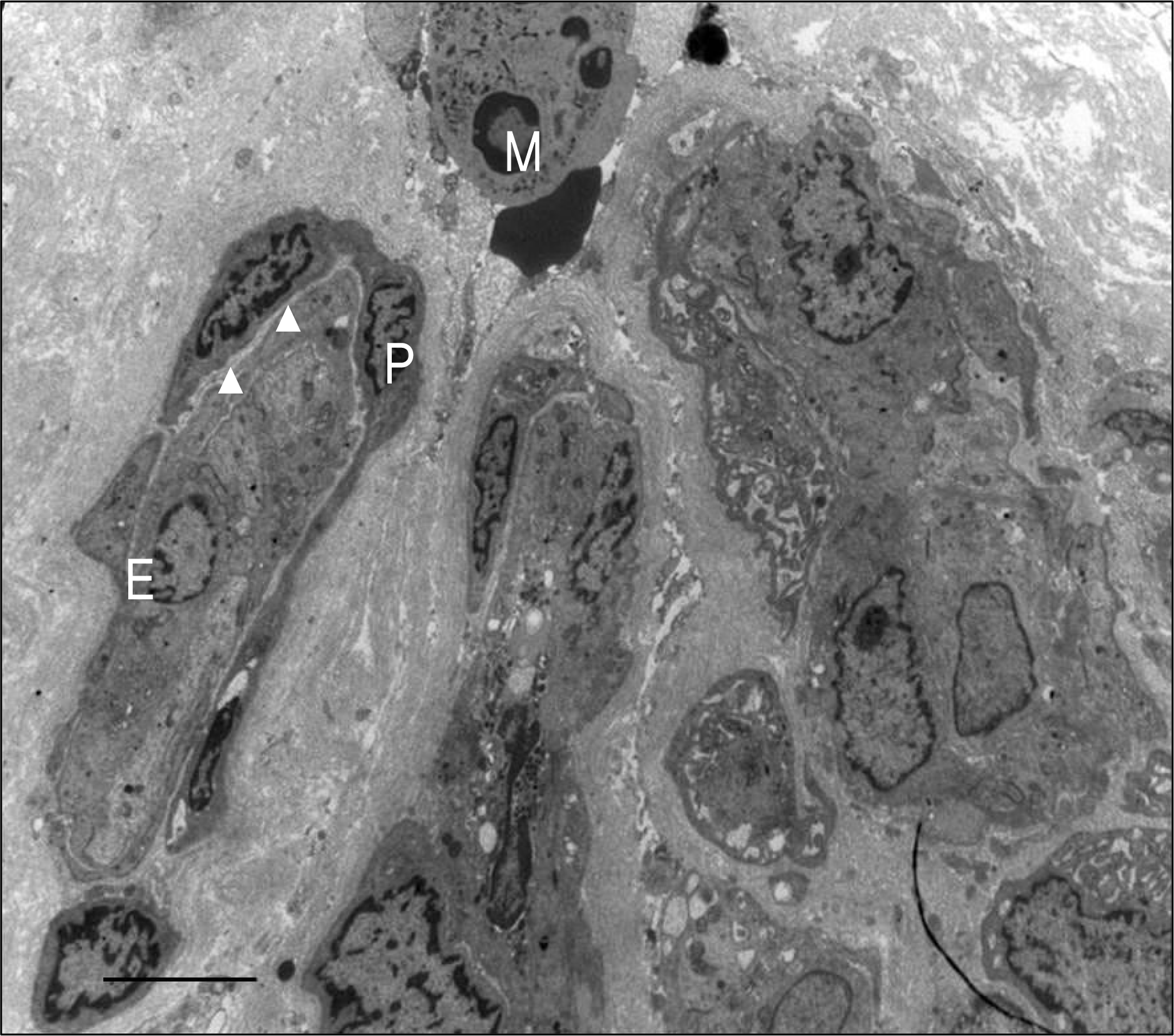
Figure 9.
Transmission electron micrograph of matured vessel. The vascular endothelial cell (E) and pericyte (P) are shown. The nucleus of vascular endothelial cell has prominent nucleoli with chromatin clumping. The vascular endothelial cell has prominent villous processes. Bar indicates 3 μ m.
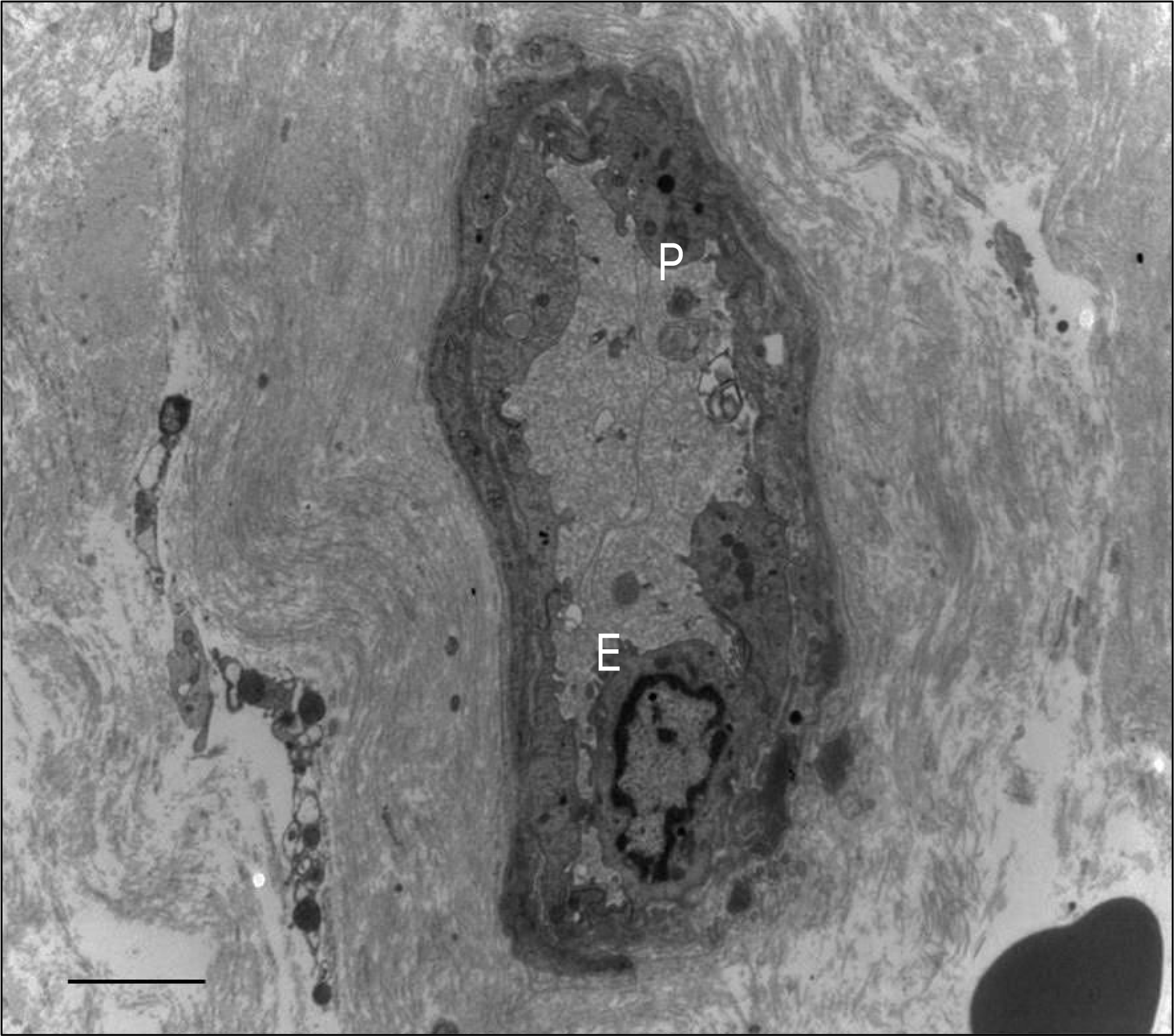
Table 1.
Clinical characteristics of the patients involved in current study
| Case | R/L* | Sex/ Age | Family History | HTN | Non-perfusion area | NVD/ NVE | Full PRP | Vitrectomy‡ | Preop. VA | Postop VA | Anatomical success | Later NVG | Later DN |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | L | M/38 | + | + | 4Q† | +/+ | + | 4 month | HM | 0.1 | + | 2 months | + |
| 2 | L | M/46 | − | + | 4Q | +/+ | + | 3 month | HM | 0.1 | + | 3 months | + |
| 3 | R | M/38 | − | + | 4Q | +/+ | + | 4 month | HM | 0.2 | + | 4 months | + |
| 3 | L | M/38 | + | + | 4Q | +/+ | + | 4 month | HM | 0.1 | + | − | + |
| 4 | R | M/50 | + | + | 4Q | +/+ | + | 2 month | HM | 0.15 | + | − | + |
| 5 | L | M/39 | + | + | 4Q | +/+ | + | 3 month | HM | 0.3 | + | − | + |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download