Abstract
Anti-Helicobacter pylori activity guided fractionation led to the isolation of five anthraquinones, two stilbenes and one naphthoquinone from the EtOAc fraction of Polygonum cuspidatum, using silica gel column chromatography, Sephadex-LH20, MPLC and recrystallization. The chemical structures were identified to be physcion (1), emodin (2), anthraglycoside B (3), trans-resveratrol (4), anthraglycoside A (5), polydatin (6), 2-methoxy-6-acetyl-7-methyljuglone (7) and citreorosein (8) by UV,1 H-NMR,13 C-NMR and mass spectrometry. Anti-Helicobacter pylori activity including MIC values of each compound was evaluated. All of the isolates exhibited anti-H. pylori activity of which MIC values were lower than that of a positive control, quercetin. Compounds 2 and 7 showed potent growth inhibitory activity. Especially, a naphthoquinone, compound 7 displayed most potent antibacterial activity with MIC50 value of 0.30 µM and MIC90 value of 0.39 µM. Although anti-H. pylori activity of this plant was previously reported, this is the first report on that of compounds isolated from this species. From these findings, P. cuspidatum roots or its isolates may be useful for H. pylori infection and further study is needed to elucidate mechanism of action.
Go to : 
REFERENCES
(1). Goodwin C. S., Armstrong J. A., Chilvers T., Peters M., Collins M. D., Sly L., McConnell W., Harper W. E. S.Int. J. Syst. Bacteriol. 1989; 39:397–405.
(2). Uemura N., Okamoto S., Yamamoto S., Matsumura N., Yamaguchi S., Yamakido M., Taniyama K., Sasaki N., Schlemper R. J. N. Engl. J.Med. 2001; 345:784–789.
(3). Boquet P., Ricci V., Galmiche A., Gauthier N. C.Trends Microbiol. 2003; 11:410–413.
(4). Warren J. R., Marshall B.Lancet. 1983; 321:1273–1275.
(6). Dore M. P., Lu H., Graham D. Y.Gut. 2016; Epub ahead of print.
(7). Wang J., Li W. T., Zheng Y. X., Zhao S. S., Li N., Huang Y., Zhou R. R., Huang Z. B., Fan X. G.Gastroenterol. Res. Pract. 2016; Epub ahead of print.
(8). Malfertheiner P., Selgrad M.Curr. Opin. Gastroenterol. 2014; 30:589–595.
(9). Dos Santos A. A., Carvalho A. A.World J. Gastroenterol. 2015; 21:139–154.
(10). Austin A., Jegadeesan M., Gowrishankar R.Nat. Prod. Sci. 2003; 9:1–3.
(11). Arichi H., Kimura Y., Okuda H., Baba K., Kozawa M., Arichi S.Chem. Pharm. Bull. 1980; 30:1766–1770.
(12). Su P. W., Yang C. -H., Yang J. -F., Su P. Y., Chuang L. Y.Molecules. 2015; 20:11119–11130.
(13). Zhang W. T., Jia Y., Huang Q. W., Li Q., Bi K. S.Chromatographia. 2007; 66:685–689.
(14). Jiangsu New Medical College. Dictionary of Chinese Materia Medica; Science and Technology Press: China. 1977; 1329–1331.
(15). Zhang H., Li C., Kwok S. T., Zhang Q. W., Chan S. W.Evid. Based Complement. Alternat. Med. 2013; 208349.
(16). Peng W., Qin R., Li X., Zhou H. J.Ethnopharmacol. 2013; 148:729–745.
(17). Lin C. J., Lin H. J., Chen T. H., Hsu Y. A., Liu C. S., Hwang G. Y., Wan L.PLoS One. 2015; 10:e0117602.
(18). Lee C. C., Chen Y. T., Chiu C. C., Liao W. T., Liu Y. C., David Wang H. M. J.Biosci. Bioeng. 2015; 119:464–469.
(19). Su P. W., Yang C. -H., Yang J. -F., Su P. Y., Chuang L. Y.Molecules. 2015; 20:11119–11130.
(20). Park W. S., Bae J. -Y., Kim H. J., Kim M. K., Lee W. K., Kang H. -L., Baik S. C., Lim K. M., Lee M. K., Ahn M.-J. Nat. Prod. Sci. 2015; 21:49–53.
(21). Amin M., Anwer M., Naz F., Mehmood T., Saari N.Molecules. 2013; 18:2135–2149.
(22). Yang Lu. In Introduction to Natural Product Chemistry: Anthraquinones. Renheng X. U., Yang Y., Zhao W., editorsCRC press;USA: 2012; 10:189–203.
(23). Chu X., Sun A., Liu R. J.Chromatogr. A. 2005; 1097:33–39.
(24). Sivakumar B., Murugan R., Baskaran A., Khadangale B. P., Murugan S., Senthilkumar U. P.Sci. Pharm. 2013; 81:683–695.
(25). Zhang W., Ye M., Zhan J., Chen Y., Guo D.Biotechnol. Lett. 2004; 26:127–131.
(26). Kimura Y., Kozawa M., Baba K., Hata K.Planta Med. 1983; 48:164–168.
(27). Brown J. C., Wang J., Kasman L., Jiang X., Haley-Zitlin V. J.Appl. Microbiol. 2011; 110:139–146.
(28). Park B. S., Lee H. K., Lee S. E., Piao X. L., Takeoka G. R., Wong R. Y., Ahn Y. J., Kim J. H. J.Ethnopharmacol. 2006; 105:255–262.
(29). Skouloubris S., Djaout K., Lamarre I., Lambry J. C., Anger K., Briffotaux J., Liebl U., de Reuse, H. and Myllykallio H.Open Biol. 2015; 5:150015.
(30). Lee I. -S., Im H. G., Lee S.Korean J. Food Sci. Technol. 2003; 35:1182–1187.
Go to : 
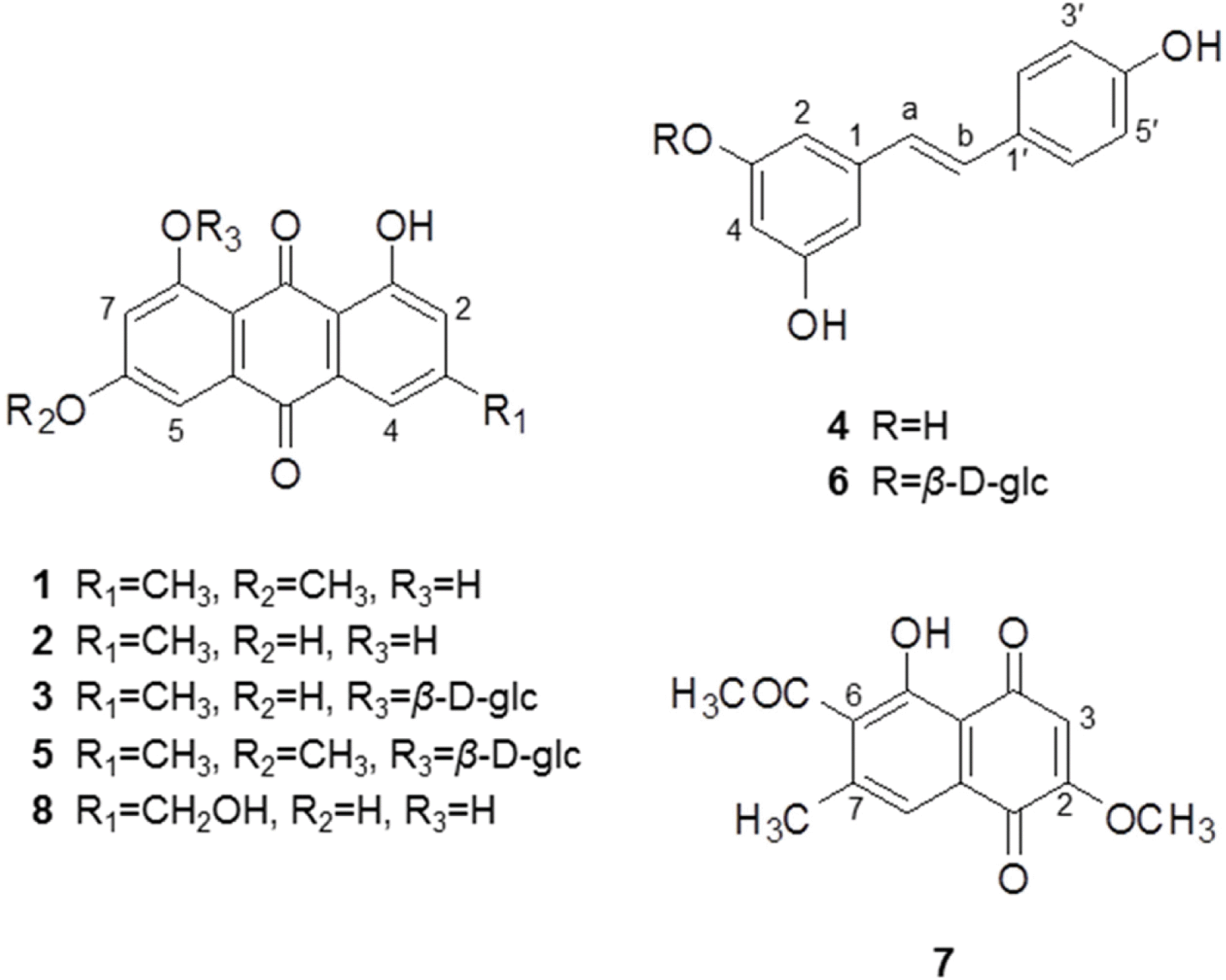 | Fig. 1.Chemical structures of compounds 1–8 isolated from the roots of Polygonum cuspidatum. |
Table 1.
Anti-Helicobacter pylori activity of total extract and the fractions from P. cuspidatum
| Sample | DMSO | Quercetin | Total Ex. | Hexane Fr. | EtOAc Fr. | BuOH Fr. | Water Fr. |
|---|---|---|---|---|---|---|---|
| Clear zone (mm) | – | 11 | 16 | 19 | 21 | 12 | 10 |
Table 2.
13 C-NMR chemical shifts of compounds 1–8




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download