Abstract
Chemotherapy-induced cognitive impairment is recognized as the most typical symptom in patients with cancer that occurs during and following the chemotherapy treatment. Recently many studies focused on pharmaceutical strategies to control the chemotherapy side effects, however it is far from satisfactory. There may be a need for more effective treatment options. The aim of this study was to investigate the protective effect of exercise on cisplatin-induced neurotoxicity. Eight-week-old C57BL6 mice were separated into three group: normal control (CON, n = 8); cisplatin injection control (Cis-CON, n = 8); cisplatin with aerobic exercise (Cis-EXE, n = 8). Cisplatin was administered intraperitoneally at a dose of 3.5 mg/kg/day. The Cis-EXE group exercise by treadmill running (14–16 m/min for 45 min daily, 3 times/week) for 12 weeks. Compared to the CON group, the cisplatin injection groups showed significant decrease in body weight and food intake, indicating successful induction of cisplatin toxicity. The Cis-CON group showed significantly increased levels of pro-inflammatory cytokines including IL-6, IL-1β, and TNF-α in the hippocampus, while the Cis-EXE group was significantly decreased in the expression of IL-6, IL-1β, and TNF-α. In addition, compared to the CON group, the levels of synapse-related proteins including synapsin-1 and -2 were significantly reduced in the Cis-CON group, and there was a significant difference between the Cis-CON and Cis-EXE groups. Antioxidant and apoptosis factors were significantly improved in the Cis-EXE group compared with the Cis-CON group. This study suggest that exercise could be meaningful approach to prevent or improve cisplatin-induced cognitive impairment.
Cisplatin is a widely used chemotherapeutic agent for treating cancer patients [1-3]. Its bioactivity is based on disruption of calcium homeostasis, lipid peroxidation, and DNA damage to induce apoptosis of cancer cells [4]. Many researchers and studies have generally been accepted that cisplatin is one of the most successful anti-cancer agents, and its therapeutic benefits have been well documented. Nevertheless, there is limiting considerably its prolonged utilization because leads to a range of acute and late toxicities [5,6].
Cisplatin-induced toxicities lead to numerous side effects including cardiovascular and metabolic diseases, hypogonadism, poor mental health-related quality of life, peripheral neuropathy, and cognitive impairment [7]. Particular, cognitive impairment is recognized as the most typical symptom induced by cisplatin neurotoxicity in the central nervous system (CNS). Cisplatin-induced cognitive impairment is known as chemo-brain, and also frequently reported by patients treated for tumor outside the CNS [8]. Actually, it has been reported that approximately 60%–75% of patients suffer from both short- and long-term cognitive impairment during and following cisplatin treatment. Along with, some patients have psychological issues such as depression, anxiety, and lethargy [9]. Importantly, the current problem is to note that these health concerns may lead to future treatment refusal and/or reduce the effectiveness of advanced-stage treatment.
Although the pathogenesis of cisplatin-induced cognitive impairment is not completely understood, accumulating evidence suggests that it is similar to biological progressive brain aging and that cisplatin contributes to both structural and functional changes in the hippocampus causing neuropathologic alterations [10]. The hippocampus plays a pivotal role in cognitive function including attention, memory, processing speed, and learning. However, the hippocampus is very vulnerable and sensitive to anti-cancer drugs such as cisplatin [11], and dysfunction of it strongly is implicated in to cause and/or consequence of various pathological conditions. It has been reported that hippocampal dysfunction is associated with an increased prevalence of mild cognitive impairment and neurodegenerative disorders such as Alzheimer’s disease and dementia [12].
Systemic inflammation is recognized as a critical predictor the onset of the various disease in clinical and animal studies. Among with many pathways, increased inflammation plays a vital role in the etiology of neurodegenerative diseases, and it has been well documented [13,14]. Many studies have reported that cisplatin accelerates releases of pro-inflammatory cytokines such as tumor necrosis factor alpha (TNF-α) and interleukins (ILs), which are involved in oxidative stress, mitochondrial damage, neuronal apoptosis, dysregulation of neurotransmitters, and inhibition of neurogenesis in the hippocampus [15,16]. In addition, synaptic dysfunction such as synapses loss and deficits were thought to be strongly associated with cognitive impairment. Some studies reported that increased inflammatory factors in the CNS contributes to weaken target neuron activity through modification of the translational expression of proteins that control synaptic stability and neuronal excitability [17,18]. In this regard, it has been demonstrated that increased inflammatory response by cisplatin is involved in the regulation of synapsin-1 and -2 which are essential for synaptic plasticity and cognition [19].
Given that increased inflammation is a hallmark of cancer pathophysiology [20] and that inflammation response after cisplatin treatment, modulation of specific cytokines may be a potential strategy to prevent and mitigate cognitive impairment caused by cisplatin. Recently, there has been a growing body of research on pathophysiological mechanisms of cisplatin-induced cognitive impairment, but effective early intervention strategies to delay or ameliorate has not been developed yet.
Exercise is widely accepted as an effective non-pharmacological intervention to prevent and improve various diseases and to maintain health. Many clinical and animal studies indicate that exercise contribute to alteration to brain activation and/or damage. Recent studies reports that well-organized regular exercise and physical activity bring benefits to cisplatin toxicities-mediated side effects in the peripheral regions including skeletal muscle, renal, and heart [21-23] and emphasize the importance of consistent of exercise practice to potential protective effects. In contrast to, there is a limited amount of research on the effects of exercise on common CNS outcomes caused by chemotherapy. Given the potential for cisplatin-induced cognitive impairment, this study aimed to determine the efficacy of exercise in protecting against early indicators of cognitive impairment mediated by hippocampal damage following cisplatin treatment.
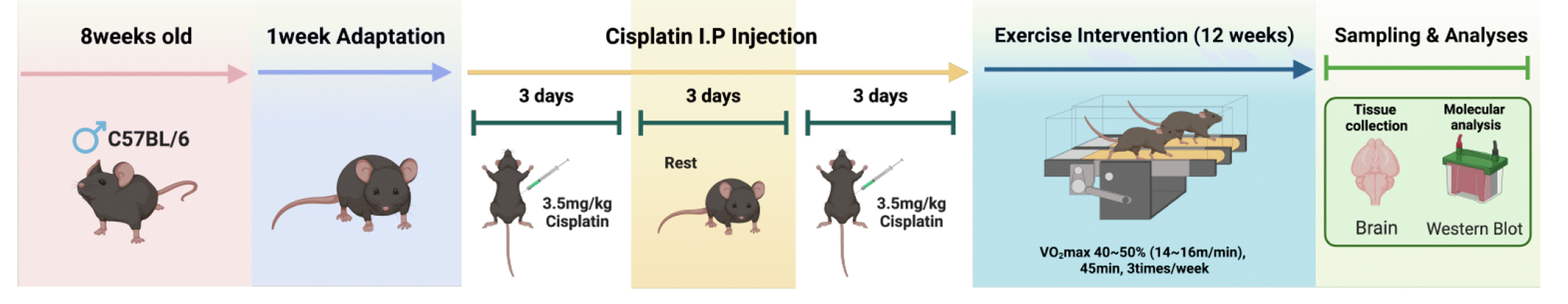
Eight-week-old C57BL6 mice were purchased from the Yeungnam Bio Company. The mice were randomly divided into three groups as follows: control (CON, n = 8); cisplatin injection (Cis-CON, n = 8); exercise with cisplatin injection (Cis-EXE, n = 8). The animals were preserved at a temperature of 20°C ± 2.5°C under a 12-h light-dark cycle with free access to food and water. All experimental procedures were approved by the Institutional Animal USE and Care Committee of Inje University (2023-006).
To induce hippocampus damage by cisplatin administration, we used the platinum-based compound cisplatin (Sigma-Aldrich, Cat# 232120), which has been reported to persist long-term for about 2–5 weeks. Male mouse at the age of 4–5 months were treated with daily cisplatin (3.5 mg/kg/day) through intraperitoneal injection. Notably, previous studies have reported that cisplatin penetrates the blood-brain barrier and persists long-term for about 2–5 weeks after its administration and this dose causes significant neurotoxicity to hippocampal neurons, leading to cognitive impairment in rodents [24]. The overall experimental procedure is shown in Fig. 1.
The Cis-EXE group was familiarized with treadmill running (1050-RM, Exer-3/6 treadmill; Columbus Instruments) three times per week at a pace of 10 m/min for 15 min. During the first week after familiarization, the mice exercised by treadmill running at 12 m/min on a 0% gradient for 30 min/day. The exercise intensity was gradually increased to 14–16 m/min (VO2max 40%–50%) on a 0% gradient for 30–45 min/day over 12 weeks [25].
Tissue samples were collected from the hippocampus, frozen on ice, and stored at –80°C until analysis. Samples were homogenized using RIPA buffer. Samples were spun at 14,000 rpm for 15 min at 4°C, and total protein concentration of the supernatant was determined by a Bradford assay. Tissues were collected and dissolved in a radioimmunoprecipitation analysis buffer using a cocktail of protease inhibitors. Protein lysate was electrophoresed with sodium dodecyl sulfate-polyacrylamide gel and transferred to nitrocellulose membranes. Next, 5% bovine serum albumin was used to block the membranes at 4°C for 2 h. The membranes were subsequently incubated with the following primary antibodies at 4°C overnight: synapsin-1 (1:1,000, Cell Signaling), synapsin-2 (1:1,000, Cell Signaling), TNF-α (1:2,000, Cell Signaling), IL-6 (1:2,000, Cell Signaling), IL-1β (1:2,000, Cell Signaling), IL-10 (1:2,000, Cell Signaling), super oxide dismutase (SOD) (1:1,000, Abcam), Bcl-2 (1:1,000, Abcam), Bax (1:1,000, Abcam) and the reference protein GAPDH (1:100,000, Cell Signaling). After washing with Tris-buffered saline with Tween 20, the membranes were incubated with HRP-conjugated secondary antibody (anti-mouse or anti-rabbit, Cell Signaling) at a dilution of 1:2,000 at 4°C for 4 h. An enhanced chemiluminescence kit (Merck Millipore) and imaging system (Bio Spectrum 600; UVP) were used to visualize the bands. ImageJ (National Institutes of Health) was used to quantify the blots.
Statistical analyses were performed using GraphPad Prism version 8.1.0. software (GraphPad, Inc.). All data are presented as the mean ± standard deviation. Different groups were evaluated using one-way analysis of variance and Tukey’s honestly significant difference test for post-hoc analysis. A p-value of ≤ 0.05 was considered to indicate a statistically significant difference.
To validate the effects of cisplatin toxicity, we measured body weight and food intake among the treatment groups (Fig. 2). Compared to the CON group, body weight and average food intake (g/week) were significantly decreased in the Cis-CON and Cis-EXE groups. Our data pointed to the fact that body weight loss and decreased appetite are the most commonly reported initial adverse effects following chemotherapy and these results are consistent with previous studies indicative of successful induction of cisplatin toxicity.
Since the inflammatory response is involved in the pathological process of cognitive impairment, we used western-blot to observe the alteration of IL-6, IL-1β, TNF-α, and IL-10. The IL-6, IL-1β, and TNF-α levels were elevated in the Cis-CON group when compared with the CON group, as shown in Fig. 3. Compared to the Cis-CON group, there was a significantly decreased levels of IL-6 and TNF-α in the Cis-EXE group. Our data indicated that immediate exercise intervention after cisplatin treatment may contribute to the inhibition of their expression. The level of IL-10 was slightly increased in the Cis-EXE group compared to the Cis-CON group, but the difference between the two group was not significant.
To clarify the differences in the expression of synapse-related protein level among the groups, the protein expression levels of synapsin-1 and -2 in hippocampus tissue were determined (Fig. 4). The protein expression levels of synapsin-1 and synapsin-2 were significantly lower in the Cis-CON group compared with the CON group. Exercise increased the expression levels of synapsin-1 effectively. Synapsin-2 was no significant difference between Cis-CON and Cis-EXE group. These findings suggested that exercise alleviates some synapse-related factors in the hippocampus of mice with cisplatin injection.
To clarify the differences in the expression of antioxidant enzyme and apoptosis-related protein level among the groups, the protein expression levels of SOD, Bax, Bcl-2, and caspase-3 in hippocampus tissue were determined (Fig. 5). Compared to the Cis-CON group, the SOD and Bcl-2 protein levels were significantly increased and the Bax and caspase-3 protein levels were significantly decreased in the Cis-EXE group. These findings indicated that exercise may have antioxidant and anti-apoptosis effects in cisplatin-mediated hippocampal damage.
To the best of our knowledge, this study is the first to elucidate the beneficial effects of exercise as a non-pharmacological strategy to control cisplatin-induced cognitive impairment. Our results suggest that regular exercise intervention could be a viable option to mitigate cisplatin-induced neurotoxicity, which might be attributed to the significant decrease in some inflammatory and increase synapse- related factors.
Recently, many studies have focused on effective pharmaceutical strategies to control the chemotherapy-accompanied various side effects. However, previous results have been far from satisfactory and there may be a need for more effective treatment options or complementary therapies. Over the past decades, it has been demonstrated that exercise plays an important role in alleviating cognitive impairment mediated by certain factors such as aging and metabolic disorders. This indicates that exercise interventions have the potential to improve the cognitive deficits associated with chemotherapy. Regular exercise intervention could be a safe strategy for managing cognitive impairment and mental health problems.
To validate the successful induction of cisplatin toxicity, we observed body weight and food intake. Previous studies have reported that cytotoxic agents, including cisplatin, can cause weight loss despite their anticancer effects, and that cisplatin-mediated toxicity contributes to lipid catabolism and muscle wasting [26,27]. Compared to the CON group, body weight and average food intake (g/week) were significantly decreased in the Cis-CON and Cis-EXE groups. However, the final body weight was no significant difference between the Cis-CON and Cis-EXE groups. These results are consistent with previous studies showing that the most common symptoms of cisplatin toxicity are loss of body weight and appetite [28].
Chemotherapy-induced cognitive impairment results from widespread damage to the CNS caused by neurotoxicity [29]. Increased inflammation is the primary characteristic of cisplatin-mediated neurotoxicity, which contributes to short- or long-term cognitive impairment. Cisplatin administration can cross the blood-brain barrier and directly damage the hippocampus. Accumulating evidence suggests that inflammatory pathways may play a critical role in the underlying mechanisms of cognitive decline and cisplatin amplifies the release of inflammatory factors that trigger a cascade of cellular damage and alter neurochemistry by enhancing the release neurotransmitters in hippocampus [30,31]. In this regard, it has been demonstrated that TNF-α, IL-6, and IL-1β are prominent pro-inflammatory factor observed in the hippocampus of cognitive impairment models, and activation of these may directly contribute to cognitive impairment [32]. Inversely, IL-10 acts as anti-inflammatory and thus suppresses the activity of pro-inflammatory mediators. In the current study, we observed a significant increase in IL-6, IL-1β and TNF-α levels in the Cis-CON group compared to the CON group. Conversely, the exercise group showed a significant decrease in the aforementioned cytokines, while IL-10 showed a significant increase. Persistent these cytokines expression activates astrocytes and microglia for a strong inflammatory response and subsequently may then serve as predictors for the development of cognitive impairment [33,34]. De Senna et al. [35] and Chupel et al. [36] demonstrated that exercise improves the level of inflammation in brain by safeguarding the structural integrity of the blood-brain barrier, which eventually leads to reduction of TNF-α and IL-6 expression. Our results are in line with clinical and animal studies [37,38] that have identified elevated levels of pro-inflammatory factors as a significant predictor of a wide range of neurodegeneration disorders and observed that exercise has anti-inflammatory effects.
In addition, synaptic plasticity is the major cellular mechanism underlying cognitive function including learning and memory and is considered a key function in the process of systems memory consolidation. An increase in cytokines and free radicals causes synaptic dysfunction and synaptic integrity, which are recognized as initial signs of neurodegeneration including cognitive decline [39]. Synapsins are known as representative synaptic plasticity-related proteins and are abundant in most nerve cells. Synapsin-1 and -2 are modulated neurotransmitter release at the pre-synaptic terminal and acts as a link between extracellular stimuli and the intracellular signaling events activated upon neuronal stimulation [40]. Previous studies demonstrated that patterns of synapsins protein expression are closely associated with neurological phenotypes. A significant decrease of the synapsin-1 level in the hippocampus is predominant in the early phase of the development of cognitive deficits and Alzheimer’s disease [41,42]. Moreover, overall synapsin deletion can lead to behavioral alterations similar to neuropsychiatric conditions [43]. These results indicate that interventions focused on maintaining the expression of synapsins in the hippocampus may be beneficial for individuals at a delaying the onset of cognitive impairment. In the current study, we observed that only synapsin-1 was significantly increased in the Cis-EXE group compared with the Cis-CON group. In this regard, it has been reported that synapse-related proteins depended on inflammatory response and the increase of cytokines such as TNF-α and IL-6 inhibits the expression of synapsin-1 and -2 in normal biological aging [44]. On the other hand, many studies indicated that long-term exercise might protect hippocampal by enhancing synaptic connections and either promotes synaptic formation and/or maintain synapses that would have otherwise alteration after brain damage [38,45]. Eventually, reduced inflammation and increased plasticity within hippocampus are thought to be crucial for cognitive function [46,47]. According to previous findings, it have been demonstrated that exercise significantly decreased microgliosis, TNF-α, and IL-1β in the CA1 area of hippocampus and that long-term exercise contributes to protect hippocampal synapses and exercise-mediated FNDC5/irisin improves synaptic plasticity in mice [48,49].
Along with this, our results demonstrated the role of exercise in antioxidant and anti-apoptosis effects. Our data shows that exercise-induced antioxidant activity offsets apoptosis in the hippocampus damage mediated by cisplatin. Exercise is well known as an effective method to maintain and improve the physiological function of overall brain regions. In previous studies, Zare et al. [50] reported neuroprotective effects of exercise mediated by inhibition of apoptosis in the hippocampus and Navazani et al. [51] suggested that exercise decreased the expression of BAX and increased the expression of Bcl-2 in the hippocampus of rats treated with thimerosal, which causes neurotoxicity. Exercise plays a crucial role for controlling activation of antioxidant enzyme system. It was reported that regular exercise-mediated activation of antioxidant enzyme has protective effects on inflammatory and oxidative stress in the hippocampus [52,53].
Although the pathogenesis of cisplatin-induced cognitive impairment is not fully understood, previous studies have shown that the disruption of inflammatory signaling pathways and synaptic plasticity are involved in onset of mild cognitive impairment and neurodegenerative disorders by causing neurotoxicity. Given that, it is believed that significant control in inflammation and synaptic-related proteins through regular exercise intervention from the early phase chemotherapy may contribute to preventing and improving the risk of brain damage-accompanied cognitive impairment. Our study has certain limitations. We have not conducted behavioral tests to assess cognitive dysfunction. The previous studies using cognitive behavioral assessments have documented increased inflammation as a significant factor in cognitive deficits. This study only focused on the effects of exercise as a pre-intervention strategy on the toxicity of cisplatin, regardless of tumor control because the toxicities of it affect not only target tumor cells but also normal host cell.
It is essential to understand the hippocampus damage mediated by cisplatin and to develop therapeutic intervention strategies in order to improve the quality of life of cancer patients. Exercise plays an important role in cognitive functions, regardless of the presence or absence of disease. Our study suggests that engaging in exercise immediately after receiving cisplatin treatment could be a meaningful approach to prevent or improve cisplatin-induced cognitive impairment by regulating inflammation and synapse-related factors.
Notes
FUNDING
This work was supported by the Ministry of Education of the Republic of Korea and the National Research Foundation (NRF) of Korea (grant number NRF-2021R1C1C2013256); Cardiovascular and Metabolic Disease Core Research Support Center (2023R1A6C101B008); and National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number NRF-2022R1I1A1A01053891).
REFERENCES
1. Whitney KA, Lysaker PH, Steiner AR, Hook JN, Estes DD, Hanna NH. 2008; Is "chemobrain" a transient state? A prospective pilot study among persons with non-small cell lung cancer. J Support Oncol. 6:313–321.
2. Gan HK, Bernstein LJ, Brown J, Ringash J, Vakilha M, Wang L, Goldstein D, Kim J, Hope A, O'Sullivan B, Waldron J, Abdul Razak AR, Chen EX, Siu LL. 2011; Cognitive functioning after radiotherapy or chemoradiotherapy for head-and-neck cancer. Int J Radiat Oncol Biol Phys. 81:126–134. DOI: 10.1016/j.ijrobp.2010.05.004. PMID: 20708851.

3. Skoogh J, Steineck G, Stierner U, Cavallin-Ståhl E, Wilderäng U, Wallin A, Gatz M, Johansson B. Swenoteca. 2012; Testicular-cancer survivors experience compromised language following chemotherapy: findings in a Swedish population-based study 3-26 years after treatment. Acta Oncol. 51:185–197. DOI: 10.3109/0284186X.2011.602113. PMID: 21851186.

4. Erfani Majd N, Shahraki R, Tabandeh MR, Hosseinifar S. 2022; Protective effects of Aloe vera gel on cisplatin-induced oxidative stress, apoptosis and neurons structure in rat hippocampus. Vet Res Forum. 13:111–119.
5. Dasari S, Tchounwou PB. 2014; Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol. 740:364–378. DOI: 10.1016/j.ejphar.2014.07.025. PMID: 25058905. PMCID: PMC4146684.

6. Ghosh S. 2019; Cisplatin: the first metal based anticancer drug. Bioorg Chem. 88:102925. DOI: 10.1016/j.bioorg.2019.102925. PMID: 31003078.

7. Fung C, Dinh P Jr, Ardeshir-Rouhani-Fard S, Schaffer K, Fossa SD, Travis LB. 2018; Toxicities associated with cisplatin-based chemotherapy and radiotherapy in long-term testicular cancer survivors. Adv Urol. 2018:8671832. DOI: 10.1155/2018/8671832. PMID: 29670654. PMCID: PMC5835297.

8. Zhou W, Kavelaars A, Heijnen CJ. 2016; Metformin prevents cisplatin-induced cognitive impairment and brain damage in mice. PLoS One. 11:e0151890. DOI: 10.1371/journal.pone.0151890. PMID: 27018597. PMCID: PMC4809545.

9. Das A, Ranadive N, Kinra M, Nampoothiri M, Arora D, Mudgal J. 2020; An overview on chemotherapy-induced cognitive impairment and potential role of antidepressants. Curr Neuropharmacol. 18:838–851. DOI: 10.2174/1570159X18666200221113842. PMID: 32091339. PMCID: PMC7569321.

10. Umfress A, Speed HE, Tan C, Ramezani S, Birnbaum S, Brekken RA, Sun X, Plattner F, Powell CM, Bibb JA. 2021; Neuropathological effects of chemotherapeutic drugs. ACS Chem Neurosci. 12:3038–3048. DOI: 10.1021/acschemneuro.1c00338. PMID: 34370453. PMCID: PMC8713594.

11. Yoo KH, Tang JJ, Rashid MA, Cho CH, Corujo-Ramirez A, Choi J, Bae MG, Brogren D, Hawse JR, Hou X, Weroha SJ, Oliveros A, Kirkeby LA, Baur JA, Jang MH. 2021; Nicotinamide mononucleotide prevents cisplatin-induced cognitive impairments. Cancer Res. 81:3727–3737. DOI: 10.1158/0008-5472.CAN-20-3290. PMID: 33771896. PMCID: PMC8277702.

12. Langella S, Sadiq MU, Mucha PJ, Giovanello KS, Dayan E. Alzheimer's Disease Neuroimaging Initiative. 2021; Lower functional hippocampal redundancy in mild cognitive impairment. Transl Psychiatry. 11:61. DOI: 10.1038/s41398-020-01166-w. PMID: 33462184. PMCID: PMC7813821.

13. Amor S, Puentes F, Baker D, van der Valk P. 2010; Inflammation in neurodegenerative diseases. Immunology. 129:154–169. DOI: 10.1111/j.1365-2567.2009.03225.x. PMID: 20561356. PMCID: PMC2814458.

14. Koronyo-Hamaoui M, Gaire BP, Frautschy SA, Alvarez JI. 2022; Editorial: role of inflammation in neurodegenerative diseases. Front Immunol. 13:958487. DOI: 10.3389/fimmu.2022.958487. PMID: 35799792. PMCID: PMC9253757.

15. Englander EW. 2013; DNA damage response in peripheral nervous system: coping with cancer therapy-induced DNA lesions. DNA Repair (Amst). 12:685–690. DOI: 10.1016/j.dnarep.2013.04.020. PMID: 23684797. PMCID: PMC3733271.

16. Wang D, Wang B, Liu Y, Dong X, Su Y, Li S. 2019; Protective effects of ACY-1215 against chemotherapy-related cognitive impairment and brain damage in mice. Neurochem Res. 44:2460–2469. DOI: 10.1007/s11064-019-02882-6. PMID: 31571096.

17. Datta Chaudhuri A, Dasgheyb RM, DeVine LR, Bi H, Cole RN, Haughey NJ. 2020; Stimulus-dependent modifications in astrocyte-derived extracellular vesicle cargo regulate neuronal excitability. Glia. 68:128–144. DOI: 10.1002/glia.23708. PMID: 31469478.

18. Upadhya R, Zingg W, Shetty S, Shetty AK. 2020; Astrocyte-derived extracellular vesicles: neuroreparative properties and role in the pathogenesis of neurodegenerative disorders. J Control Release. 323:225–239. DOI: 10.1016/j.jconrel.2020.04.017. PMID: 32289328. PMCID: PMC7299747.

19. John JP, Sunyer B, Höger H, Pollak A, Lubec G. 2009; Hippocampal synapsin isoform levels are linked to spatial memory enhancement by SGS742. Hippocampus. 19:731–738. DOI: 10.1002/hipo.20553. PMID: 19140176.

20. Yang R, Chang Q, Meng X, Gao N, Wang W. 2018; Prognostic value of systemic immune-inflammation index in cancer: a meta-analysis. J Cancer. 9:3295–3302. DOI: 10.7150/jca.25691. PMID: 30271489. PMCID: PMC6160683.

21. Silva MS, de Andrade Gomes Y, de Sousa Cavalcante ML, Telles PVN, da Silva ACA, Severo JS, de Oliveira Santos R, Dos Santos BLB, Cavalcante GL, Rocha CHL, Palheta-Junior RC, de Cássia Meneses Oliveira R, Dos Santos RF, Sabino JPJ, Dos Santos AA, Tolentino Bento da Silva M. 2021; Exercise and pyridostigmine prevents gastric emptying delay and increase blood pressure and cisplatin-induced baroreflex sensitivity in rats. Life Sci. 267:118972. DOI: 10.1016/j.lfs.2020.118972. PMID: 33383052.

22. Almeida AA, Correia TML, Pires RA, Silva DAD, Coqueiro RS, Machado M, Magalhães ACM, Queiroz RF, Soares TJ, Pereira R. 2022; Nephroprotective effect of exercise training in cisplatin-induced renal damage in mice: influence of training protocol. Braz J Med Biol Res. 55:e12116. DOI: 10.1590/1414-431x2022e12116. PMID: 35976270. PMCID: PMC9377535.

23. Seo DY, Bae JH, Zhang D, Song W, Kwak HB, Heo JW, Jung SJ, Yun HR, Kim TN, Lee SH, Kim AH, Jeong DH, Kim HK, Han J. 2021; Effects of cisplatin on mitochondrial function and autophagy-related proteins in skeletal muscle of rats. BMB Rep. 54:575–580. DOI: 10.5483/BMBRep.2021.54.11.132. PMID: 34674798. PMCID: PMC8633523.

24. Nan X, Sun Q, Xu X, Yang Y, Zhen Y, Zhang Y, Zhou H, Fang H. 2022; Forsythoside B ameliorates diabetic cognitive dysfunction by inhibiting hippocampal neuroinflammation and reducing synaptic dysfunction in ovariectomized mice. Front Aging Neurosci. 14:974690. DOI: 10.3389/fnagi.2022.974690. PMID: 36389075. PMCID: PMC9650402.

25. Mille-Hamard L, Billat VL, Henry E, Bonnamy B, Joly F, Benech P, Barrey E. 2012; Skeletal muscle alterations and exercise performance decrease in erythropoietin-deficient mice: a comparative study. BMC Med Genomics. 5:29. DOI: 10.1186/1755-8794-5-29. PMID: 22748015. PMCID: PMC3473259.

26. Garcia JM, Scherer T, Chen JA, Guillory B, Nassif A, Papusha V, Smiechowska J, Asnicar M, Buettner C, Smith RG. 2013; Inhibition of cisplatin-induced lipid catabolism and weight loss by ghrelin in male mice. Endocrinology. 154:3118–3129. DOI: 10.1210/en.2013-1179. PMID: 23832960. PMCID: PMC3749475.

27. Conte E, Bresciani E, Rizzi L, Cappellari O, De Luca A, Torsello A, Liantonio A. 2020; Cisplatin-induced skeletal muscle dysfunction: mechanisms and counteracting therapeutic strategies. Int J Mol Sci. 21:1242. DOI: 10.3390/ijms21041242. PMID: 32069876. PMCID: PMC7072891.

28. Lin MT, Ko JL, Liu TC, Chao PT, Ou CC. 2018; Protective effect of D-methionine on body weight loss, anorexia, and nephrotoxicity in cisplatin-induced chronic toxicity in rats. Integr Cancer Ther. 17:813–824. DOI: 10.1177/1534735417753543. PMID: 29430988. PMCID: PMC6142074.

29. Amiri A, Chovanec M, Oliva V, Sedliak M, Mego M, Ukropec J, Ukropcová B. 2021; Chemotherapy-induced toxicity in patients with testicular germ cell tumors: the impact of physical fitness and regular exercise. Andrology. 9:1879–1892. DOI: 10.1111/andr.13078. PMID: 34245663.

30. Den H, Dong X, Chen M, Zou Z. 2020; Efficacy of probiotics on cognition, and biomarkers of inflammation and oxidative stress in adults with Alzheimer's disease or mild cognitive impairment - a meta-analysis of randomized controlled trials. Aging (Albany NY). 12:4010–4039. DOI: 10.18632/aging.102810. PMID: 32062613. PMCID: PMC7066922.

31. Kölliker-Frers R, Udovin L, Otero-Losada M, Kobiec T, Herrera MI, Palacios J, Razzitte G, Capani F. 2021; Neuroinflammation: an integrating overview of reactive-neuroimmune cell interactions in health and disease. Mediators Inflamm. 2021:9999146. DOI: 10.1155/2021/9999146. PMID: 34158806. PMCID: PMC8187052.

32. Lv H, Wang S, Tian M, Wang L, Gao J, Zhao Q, Li Z, Jia X, Yu Y. 2022; Exercise preconditioning ameliorates cognitive impairment in mice with ischemic stroke by alleviating inflammation and modulating gut microbiota. Mediators Inflamm. 2022:2124230. DOI: 10.1155/2022/2124230. PMID: 36262547. PMCID: PMC9576414.

33. Kwon HS, Koh SH. 2020; Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl Neurodegener. 9:42. DOI: 10.1186/s40035-020-00221-2. PMID: 33239064. PMCID: PMC7689983.

34. Mann CN, Devi SS, Kersting CT, Bleem AV, Karch CM, Holtzman DM, Gallardo G. 2022; Astrocytic α2-Na+/K+ ATPase inhibition suppresses astrocyte reactivity and reduces neurodegeneration in a tauopathy mouse model. Sci Transl Med. 14:eabm4107. DOI: 10.1126/scitranslmed.abm4107. PMID: 35171651. PMCID: PMC9161722.

35. de Senna PN, Xavier LL, Bagatini PB, Saur L, Galland F, Zanotto C, Bernardi C, Nardin P, Gonçalves CA, Achaval M. 2015; Physical training improves non-spatial memory, locomotor skills and the blood brain barrier in diabetic rats. Brain Res. 1618:75–82. DOI: 10.1016/j.brainres.2015.05.026. PMID: 26032744.

36. Chupel MU, Minuzzi LG, Furtado G, Santos ML, Hogervorst E, Filaire E, Teixeira AM. 2018; Exercise and taurine in inflammation, cognition, and peripheral markers of blood-brain barrier integrity in older women. Appl Physiol Nutr Metab. 43:733–741. DOI: 10.1139/apnm-2017-0775. PMID: 29474803.

37. Medhat E, Rashed L, Abdelgwad M, Aboulhoda BE, Khalifa MM, El-Din SS. 2020; Exercise enhances the effectiveness of vitamin D therapy in rats with Alzheimer's disease: emphasis on oxidative stress and inflammation. Metab Brain Dis. 35:111–120. DOI: 10.1007/s11011-019-00504-2. PMID: 31691146.

38. Wang YY, Zhou YN, Jiang L, Wang S, Zhu L, Zhang SS, Yang H, He Q, Liu L, Xie YH, Liang X, Tang J, Chao FL, Tang Y. 2023; Long-term voluntary exercise inhibited AGE/RAGE and microglial activation and reduced the loss of dendritic spines in the hippocampi of APP/PS1 transgenic mice. Exp Neurol. 363:114371. DOI: 10.1016/j.expneurol.2023.114371. PMID: 36871860.

39. Magee JC, Grienberger C. 2020; Synaptic plasticity forms and functions. Annu Rev Neurosci. 43:95–117. DOI: 10.1146/annurev-neuro-090919-022842. PMID: 32075520.

40. Lu C, Gao R, Zhang Y, Jiang N, Chen Y, Sun J, Wang Q, Fan B, Liu X, Wang F. 2021; S-equol, a metabolite of dietary soy isoflavones, alleviates lipopolysaccharide-induced depressive-like behavior in mice by inhibiting neuroinflammation and enhancing synaptic plasticity. Food Funct. 12:5770–5778. DOI: 10.1039/D1FO00547B. PMID: 34038497.

41. Lin L, Yang SS, Chu J, Wang L, Ning LN, Zhang T, Jiang Q, Tian Q, Wang JZ. 2014; Region-specific expression of tau, amyloid-β protein precursor, and synaptic proteins at physiological condition or under endoplasmic reticulum stress in rats. J Alzheimers Dis. 41:1149–1163. DOI: 10.3233/JAD-140207. PMID: 24787918.

42. Mirza FJ, Zahid S. 2018; The role of synapsins in neurological disorders. Neurosci Bull. 34:349–358. DOI: 10.1007/s12264-017-0201-7. PMID: 29282612. PMCID: PMC5856722.

43. Tassan Mazzocco M, Guarnieri FC, Monzani E, Benfenati F, Valtorta F, Comai S. 2021; Dysfunction of the serotonergic system in the brain of synapsin triple knockout mice is associated with behavioral abnormalities resembling synapsin-related human pathologies. Prog Neuropsychopharmacol Biol Psychiatry. 105:110135. DOI: 10.1016/j.pnpbp.2020.110135. PMID: 33058959.

44. Li SM, Li B, Zhang L, Zhang GF, Sun J, Ji MH, Yang JJ. 2020; A complement-microglial axis driving inhibitory synapse related protein loss might contribute to systemic inflammation-induced cognitive impairment. Int Immunopharmacol. 87:106814. DOI: 10.1016/j.intimp.2020.106814. PMID: 32707491.

45. Bilchak JN, Caron G, Côté MP. 2021; Exercise-induced plasticity in signaling pathways involved in motor recovery after spinal cord injury. Int J Mol Sci. 22:4858. DOI: 10.3390/ijms22094858. PMID: 34064332. PMCID: PMC8124911.

46. Gleeson M, Bishop NC, Stensel DJ, Lindley MR, Mastana SS, Nimmo MA. 2011; The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat Rev Immunol. 11:607–615. DOI: 10.1038/nri3041. PMID: 21818123.

47. He XF, Liu DX, Zhang Q, Liang FY, Dai GY, Zeng JS, Pei Z, Xu GQ, Lan Y. 2017; Voluntary exercise promotes glymphatic clearance of amyloid beta and reduces the activation of astrocytes and microglia in aged mice. Front Mol Neurosci. 10:144. DOI: 10.3389/fnmol.2017.00144. PMID: 28579942. PMCID: PMC5437122.

48. Rizzo FR, Guadalupi L, Sanna K, Vanni V, Fresegna D, De Vito F, Musella A, Caioli S, Balletta S, Bullitta S, Bruno A, Dolcetti E, Stampanoni Bassi M, Buttari F, Gilio L, Mandolesi G, Centonze D, Gentile A. 2021; Exercise protects from hippocampal inflammation and neurodegeneration in experimental autoimmune encephalomyelitis. Brain Behav Immun. 98:13–27. DOI: 10.1016/j.bbi.2021.08.212. PMID: 34391817.

49. Lourenco MV, Frozza RL, de Freitas GB, Zhang H, Kincheski GC, Ribeiro FC, Gonçalves RA, Clarke JR, Beckman D, Staniszewski A, Berman H, Guerra LA, Forny-Germano L, Meier S, Wilcock DM, de Souza JM, Alves-Leon S, Prado VF, Prado MAM, Abisambra JF, et al. 2019; Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer's models. Nat Med. 25:165–175. DOI: 10.1038/s41591-018-0275-4. PMID: 30617325. PMCID: PMC6327967.

50. Zare Z, Zarbakhsh S, Tehrani M, Mohammadi M. 2022; Neuroprotective effects of treadmill exercise in hippocampus of ovariectomized and diabetic rats. Neuroscience. 496:64–72. DOI: 10.1016/j.neuroscience.2022.06.012. PMID: 35700817.

51. Navazani P, Vaseghi S, Hashemi M, Shafaati MR, Nasehi M. 2021; Effects of treadmill exercise on the expression level of BAX, BAD, BCL-2, BCL-XL, TFAM, and PGC-1α in the hippocampus of thimerosal-treated rats. Neurotox Res. 39:1274–1284. DOI: 10.1007/s12640-021-00370-w. PMID: 33939098.

52. Vanzella C, Neves JD, Vizuete AF, Aristimunha D, Kolling J, Longoni A, Gonçalves CAS, Wyse ATS, Netto CA. 2017; Treadmill running prevents age-related memory deficit and alters neurotrophic factors and oxidative damage in the hippocampus of Wistar rats. Behav Brain Res. 334:78–85. DOI: 10.1016/j.bbr.2017.07.034. PMID: 28756215.

53. Rasoolijazi H, Norouzi Ofogh S, Ababzadeh S, Mehdizadeh M, Shabkhiz F. 2021; Comparing the effects of rosemary extract and treadmill exercise on the hippocampal function and antioxidant capacity in old rats. Basic Clin Neurosci. 12:361–372. DOI: 10.32598/bcn.12.3.2139.1. PMID: 34917295. PMCID: PMC8666924.

Fig. 2
Evaluation of cisplatin-induced toxicity.
(A) Weekly change of body weight. (B) Compared to CON group, the body weight (F = 26.07, p < 0.01) and (C) food intake (F = 71.58, p < 0.01) were significantly decreased in Cis and Cis-EXE groups in 3 weeks after cisplatin injection. Values are presented as mean ± SD. CON, control (n = 8); Cis, cisplatin injection (n = 8); Cis-EXE, aerobic exercise with cisplatin injection (n = 8). One-way analysis of variance and Tukey’s post-hoc were used for statistical analysis; ***p < 0.001.
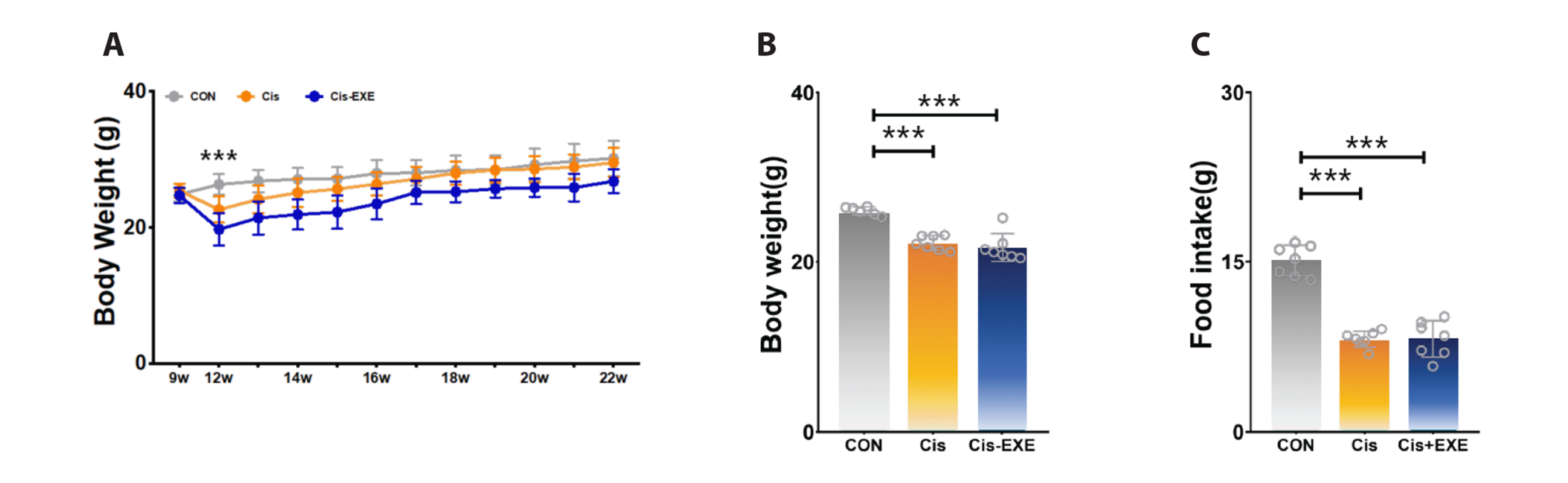
Fig. 3
Expression levels of neuroinflammation cytokines in the hippocampus after cisplatin injection.
(A) Representative Western blots of neuroinflammatory markers. Significant reduction in neuroinflammatory markers (B) IL-6 (F = 19.55, p < 0.01), (C) TNF-α (F = 45.43, p < 0.01), and (D) IL-1β (F = 37.75, p < 0.01) were observed in the Cis-EXE group compared to the Cis group. However, no significant difference were found in (E) IL-10 levels. Values are presented as mean ± SD. IL-6, interleukin 6; TNF-α, tumor necrosis factor alpha; IL-1β, interleukin 1 beta; IL-10, interleukin 10; GAPDH, glyceraldehyde-3-phosphate dehydrogenase. CON, control (n = 8); Cis, cisplatin injection (n = 8); Cis-EXE, aerobic exercise with cisplatin injection (n = 8). Bradford assay R2 = 0.993, ImageJ gamma value = 0.50. One-way analysis of variance and Tukey’s post-hoc were used for statistical analysis; **p < 0.01, ***p < 0.001.
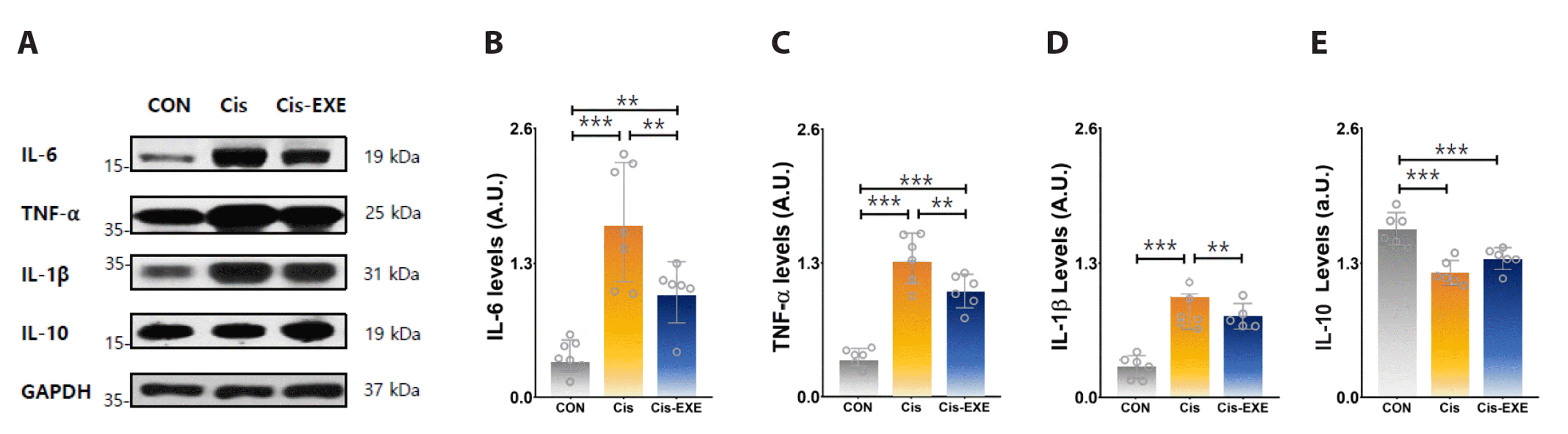
Fig. 4
Expression levels of synaptic plasticity-related factors in the hippocampus after cisplatin injection.
(A) Representative Western blots of synapse-related proteins. Compared to the Cis group, expression of the (B) synapsin-1 protein (F = 55.46, p < 0.01) was significantly increased in Cis-EXE group. Meanwhile, no significant difference were found in (C) synapsin-2 levels. Values are presented as mean ± SD. GAPDH, glyceraldehyde-3-phosphate dehydrogenase. CON, control (n = 8); Cis, cisplatin injection (n = 8); Cis-EXE, aerobic exercise with cisplatin injection (n = 8). Bradford assay R2 = 0.993, ImageJ gamma value = 0.50. One-way analysis of variance and Tukey’s post-hoc were used for statistical analysis; **p < 0.01, ***p < 0.001.
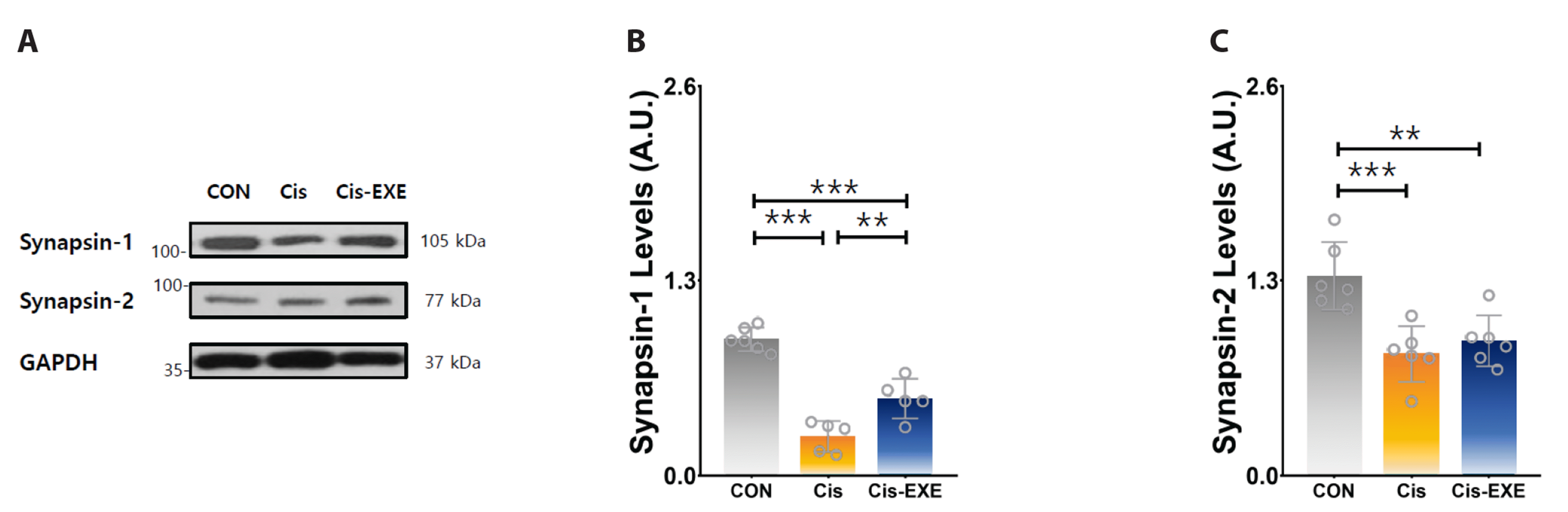
Fig. 5
Expression levels of antioxidant enzyme and apoptosis-related factors in the hippocampus after cisplatin injection.
(A) Representative Western blots of antioxidant and apoptosis-related factors. (B) SOD was significantly increased in the Cis-EXE group compared to the Cis group (F = 57.71, p < 0.01). Despite there is no significant difference in the levels of apoptosis-related marker (C) caspase 3, there is a significant decrease in the level of (D) Bax in the Cis-EXE group compared to the Cis group (F = 31.56, p < 0.01) while there is a significant increase in (E) Bcl-2 levels (F = 20.12, p < 0.01). Values are presented as mean ± SD. SOD, super oxide dismutase; GAPDH, glyceraldehyde-3-phosphate dehydrogenase. CON, control (n = 8); Cis, cisplatin injection (n = 8); Cis-EXE, aerobic exercise with cisplatin injection (n = 8). Bradford assay R2 = 0.993, ImageJ gamma value = 0.50. One-way analysis of variance and Tukey’s post-hoc were used for statistical analysis; *p < 0.05, **p < 0.01, ***p < 0.001.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download