TO THE EDITOR: Hairy cell leukemia (HCL) is a rare B-cell neoplasm with an indolent clinical course. It typically presents with progressive cytopenias, splenomegaly, diffuse bone marrow infiltration and rarely lymphadenopathy. The involvement of extranodal sites is infrequent, and presentation as an isolated extranodal mass without involvement of peripheral blood, bone marrow, and spleen is extremely rare. When HCL presents as an isolated extranodal mass, the diagnosis can be challenging since the classic leukemic component may be absent. In such cases, a biopsy of the mass is necessary to establish the diagnosis. Immunohistochemistry and flow cytometry can be useful tools in confirming the diagnosis of HCL. Presentation of HCL as an isolated extranodal mass without involvement of the peripheral blood, bone marrow and spleen is extremely rare. We report a case of hairy cell leukemia presenting as a pleural mass and discuss the diagnostic challenges when a classic leukemic component is absent.
Hairy cell leukemia (HCL) is a neoplasm of small mature B-cells with circumferential cytoplasmic hairy projections. HCL presents in middle age (median age 58) with a male predominance, accounting for 2% of lymphoid leukemias [1]. Clinical presentation with cytopenias, including characteristic monocytopenia and splenomegaly, is common. The bone marrow is diffusely involved, and the aspirate is often hemodiluted, resulting in a “dry-tap” due to reticulin fibrosis. Extranodal lesions caused by HCL have been reported as isolated case reports; however, most cases had a preceding diagnosis with a leukemic component. Only rare cases of hairy cell leukemia without peripheral blood, bone marrow, and spleen involvement have been reported. We present this case report and discuss the potential diagnostic pitfalls and challenges with this rare and unusual presentation.
A 60-year-old male presented with right lower chest pain for four years. He has had no weight loss, fevers, or night sweats. There was no history of smoking. His physical exam was unremarkable, without any lymphadenopathy or hepato-splenomegaly. Laboratory testing included a complete blood count (CBC), full chemistry panel, serum protein electrophoresis, hepatitis and human immunodeficiency virus panels, and lactate dehydrogenase were normal.
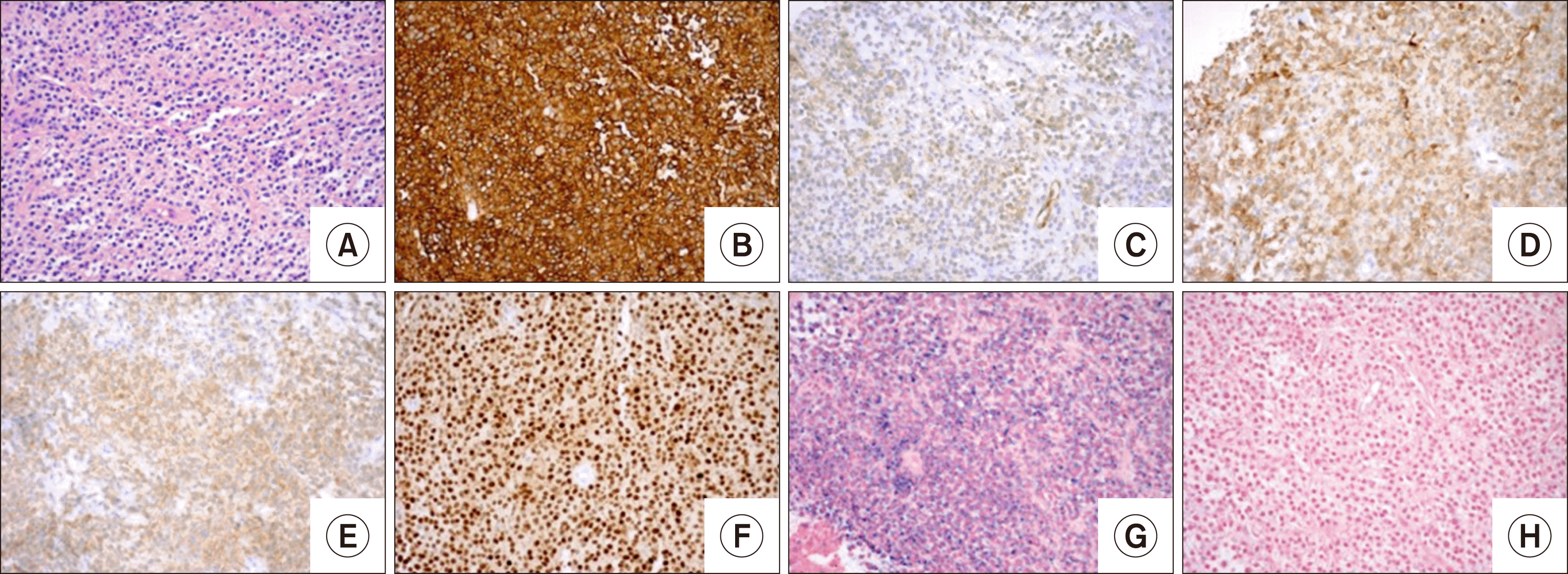
Chest imaging revealed a pleural-based mass (8.3×12.8× 10.4 cm) in the right midlung. A biopsy of the mass showed focal small lymphoid nodules and sheets of monocytic lymphocytes with eccentric nuclei. Scattered plasma cells were also seen. The differential diagnoses of marginal zone lymphoma and lymphoplasmacytic lymphoma were considered by morphology. The B-cells were strongly positive for CD19, CD20, Pax-5, and kappa light chains and were negative for CD5, CD10, and CD138. Immunostaining for Cyclin-D1 was also positive (Fig. 1).
Fluorescence in situ hybridization (FISH) studies for IGH/CCND1 were performed to rule out mantle cell lymphoma and were negative. Additional stains were performed to characterize the B-cell lymphoma further, and the cells were weakly positive for Annexin-1, BRAF, and CD123. This phenotype raised concern for hairy cell leukemia, although the location and presentation were very unusual, and the patient’s CBC was currently within the reference range. A flow cytometry study was recommended to evaluate hairy cell markers (CD11c, CD25, and CD103). Serum protein electrophoresis was also suggested.
Subsequently, the patient underwent a bone marrow biopsy which was negative for malignancy.
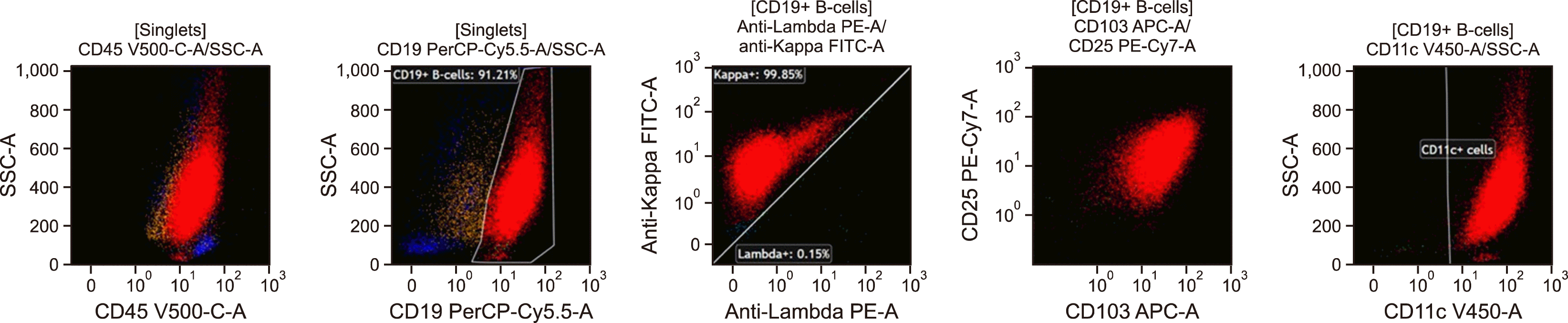
A thoracic surgery referral was recommended to evaluate the pleural mass further. The decision was made for a right video-assisted thoracoscopic surgery (VATS) incisional biopsy of the pleural mass. Tissue was sent for morphology, flow cytometry, cytogenetic and molecular analysis. Flow cytometry analysis detected a population of mature B-cells with high-side scatter, expressing CD19, moderate to high-density CD20, CD38, CD123, high-density expression of CD11c, CD25, and CD103 and moderate-density monotypic kappa light chains present at 94% of lymphocytes (68% of total cells). The B-cells were negative for CD5, CD10, and CD23 and were intermediate in size by forward angle light scatter (Fig. 2).
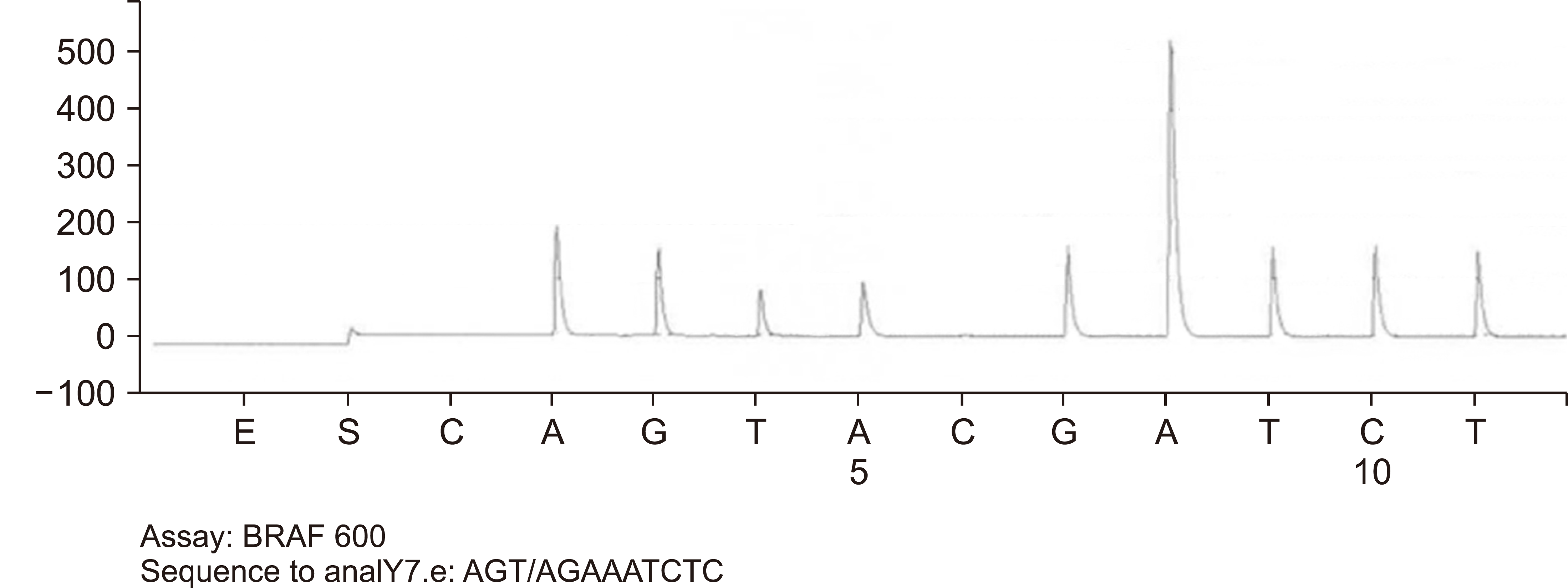
DNA amplification and pyrosequencing analysis identified BRAF molecular mutation (p.Val600Glu; p.V600E) in codon 600 of the BRAF gene (Fig. 3). Cytogenetic studies by routine chromosome analysis were non-contributory due to a lack of available metaphases for evaluation. The tumor morphology, immunoprofile, and presence of BRAF mutation were diagnostic of classical hairy cell leukemia. The patient received Cladribine for five days with no difficulties. Then he was placed on Rituximab weekly for 12 weeks.
Hairy cell leukemia is a rare B-cell neoplasm with an annual incidence rate of 0.32 cases per 100,000 population in the United States. The median age at diagnosis is 58 years old, and the male-to-female ratio is 4:1 [1]. It tends to have a classic pattern of immunohistochemical (IHC) staining, which includes positive staining for B-cell markers (e.g., CD19, CD20, and CD22), CD11c, CD25, CD103, and CD123 while CD5, CD10, and CD23 are negative [2]. BRAFV600E mutation, identified by polymerase chain reaction (PCR) analysis and IHC staining, has been reported as a disease-defining feature [3].
The diagnosis of HCL can be made without difficulty when the presentation is classic with cytopenias and diffuse bone marrow involvement. However, the diagnosis can be challenging when the presentation is non-classic. Extranodal HCL leukemia has been reported in different locations, such as the parotid gland, femur, breast, lumbar spine, and pleura [4-6]. In contrast to the typical presentation of HCL, our patient presented with a pleural mass without any peripheral blood cytopenia or splenomegaly.
The morphological features of extranodal HCL can overlap with marginal zone lymphoma, mantle cell lymphoma, and lymphoplasmacytic lymphoma [7]. Based on the morphology and initial immunophenotype in our case (strong positivity for CD20, CD19, PAX5, and Kappa light chains and negative for CD138, CD10, and CD5), the differential diagnosis included marginal zone lymphoma and lymphoplasmacytic lymphoma. Additional staining revealed that the tumor cells were also positive for cyclin-D1, which is unusual for both entities. FISH cytogenetic studies for IGH/CCND1 fusion were negative, which argues against mantle cell lymphoma. The diagnosis of hairy cell leukemia was confirmed by positive BRAF immunostaining and the presence of BRAF mutation. Most hairy cell leukemia patients achieve complete remission with a single 5-day course of cladribine [8, 9].
The unusual tumor mass presentation of HCL without leukemic component and the morphology can overlap with other low-grade B-cell lymphomas. As illustrated by our case, cyclin-D1 positivity can be beneficial in the initial differential diagnosis workup, and FISH cytogenetic studies should be performed to rule out mantle cell lymphoma. Additional studies, including flow cytometry for hairy cell markers (CD11c, CD25, and CD103), immunostaining for Annexin and BRAF, and molecular analysis for BRAF mutation, are all essential tools to establish the diagnosis of hairy cell leukemia.
REFERENCES
1. Swerdlow SH, Campo E, Pileri SA, et al. 2016; The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 127:2375–90. DOI: 10.1182/blood-2016-01-643569. PMID: 26980727. PMCID: PMC4874220.

2. Dores GM, Matsuno RK, Weisenburger DD, Rosenberg PS, Anderson WF. 2008; Hairy cell leukaemia: a heterogeneous disease? Br J Haematol. 142:45–51. DOI: 10.1111/j.1365-2141.2008.07156.x. PMID: 18477040. PMCID: PMC4009349.

3. Tiacci E, Trifonov V, Schiavoni G, et al. 2011; BRAF mutations in hairy-cell leukemia. N Engl J Med. 364:2305–15. DOI: 10.1056/NEJMoa1014209. PMID: 21663470. PMCID: PMC3689585.
4. Cortazar JM, DeAngelo DJ, Pinkus GS, Morgan EA. 2017; Mor-phological and immunophenotypical features of hairy cell leukaemia involving lymph nodes and extranodal tissues. Histopathology. 71:112–24. DOI: 10.1111/his.13206. PMID: 28261866.

5. Hammond WA, Swaika A, Menke D, Tun HW. 2017; Hairy cell lymphoma: a potentially under-recognized entity. Rare Tumors. 9:6518. DOI: 10.4081/rt.2017.6518. PMID: 28458788. PMCID: PMC5379228. PMID: 37c1d1d23bb54e14887c8abc7cfdabda.

6. Rosen DS, Smith S, Gurbuxani S, Yamini B. 2008; Extranodal hairy cell leukemia presenting in the lumbar spine. J Neurosurg Spine. 9:374–6. DOI: 10.3171/SPI.2008.9.10.374. PMID: 18939925.

7. Choi WW, Weisenburger DD, Greiner TC, et al. 2009; A new immunostain algorithm classifies diffuse large B-cell lymphoma into molecular subtypes with high accuracy. Clin Cancer Res. 15:5494–502. DOI: 10.1158/1078-0432.CCR-09-0113. PMID: 19706817. PMCID: PMC7289055.

8. Chadha P, Rademaker AW, Mendiratta P, et al. 2005; Treatment of hairy cell leukemia with 2-chlorodeoxyadenosine (2-CdA): long-term follow-up of the Northwestern University experience. Blood. 106:241–6. DOI: 10.1182/blood-2005-01-0173. PMID: 15761021.

9. Saven A, Burian C, Koziol JA, Piro LD. 1998; Long-term follow-up of patients with hairy cell leukemia after cladribine treatment. Blood. 92:1918–26. DOI: 10.1182/blood.V92.6.1918. PMID: 9731048.

Fig. 1
Pleural-based mass involved by Hairy cell leukemia. All stains at ×20. Hematoxylin & eosin (A) stain shows small to medium sized cells with interspersed histiocytes. Immunohistochemical studies confirmed these cells to be CD20 (B), CD123 (C), annexin A1 (D) BRAFV600E (E), and cyclin D1 (F). Kappa (G) and Lambda (H) in situ hybridization demonstrates kappa light chains restriction.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download