This article has been
cited by other articles in ScienceCentral.
Abstract
A marker chromosome (mar) is a structurally abnormal chromosome in which no part can be identified. The significance of a marker varies, depending on the material contained within the marker. Very few reports have been published of marker chromosomes associated with male infertility. Here, we report the case of an infertile man with a rare variant of a marker chromosome of a mos 47,XY,+mar[25]/46,XY[25] karyotype.
Keywords: Azoospermia, Chromosome aberrations, Infertility, male
INTRODUCTION
Male infertility represents one of the clear examples of a complex phenotype with a substantial genetic basis. Karyotypic abnormalities are detected in 5% of patients with fertility problems, and the prevalence increases to 13% when only considering men with azoospermia [
12]. Most chromosomal abnormalities associated with male infertility involve the sex chromosomes, with Klinefelter syndrome (47,XXY) being the most commonly detected karyotypic abnormality in infertile men. Other structural changes are related to male infertility, such as translocation [
3]. Since the transmission of a haploid chromosomal asset is fundamental for embryonic vitality and development, the study of sperm chromosomes has become fundamental because recent assisted reproductive technologies such as intracytoplasmic sperm injection (ICSI) could allow fertilization even in cases of severe male infertility [
4]. A marker chromosome (mar) is a structurally abnormal chromosome in which no part can be identified. Small supernumerary marker chromosomes (sSMCs) are relatively common in the field of clinical cytogenetics, and their prevalence is estimated to be 0.05% of unselected live-born infants. The significance of a marker is variable, depending on the material contained within the marker. Very few reports have been published of marker chromosomes associated with male infertility [
5]. Here, we report the case of an infertile man with a rare variant of a marker chromosome of a mos 47,XY,+mar[25]/46,XY[25] karyotype.
CASE REPORT
A 34-year-old patient, born to a 30-year-old woman and her 39-year-old husband, was referred to our center for infertility due to azoospermia. Physical examination revealed a healthy-looking male with a normal male appearance, body height of 174.5 cm, and a body mass index of 30 kg/m2.
The patient had a normal male pattern of body hair and no gynecomastia. Examination of the external genitalia revealed both testes to be normally positioned in scrotum, with normal consistency when palpated, and with a volume of 14 mL each. There were no abnormalities of the epididymis and the vas deferens. No varicocele, erectile dysfunction, diabetes, or reduced muscle strength or malformation was noted. Mild mental retardation was suspected, but the patient refused further evaluation. There was no family history of infertility. Based on 2 semen analyses with centrifugation, the patient was diagnosed with cryptozoospermia, with only very small number of immotile sperms.
A blood test for reproductive hormones and a genetic test was conducted. The serum follicle-stimulating hormone level was 10.0 mIU/mL, and the serum testosterone was 3.78 ng/mL. Next, karyotyping was performed. Metaphase spreads from lymphocytes were prepared and trypsin-Giemsa banded according to standard techniques [
6]. DNA isolation from peripheral blood leukocytes was performed using the QuickGene DNA blood kit (Kurabo, Osaka, Japan). All procedures were followed according to the manufacturer's instructions. Molecular investigations to determine the mechanism of formation were performed using the new technical approach of glass-needle microdissection of 10 copies of the marker chromosome from metaphases, whole genome amplification (WGA) with the GenomePlex Single Cell WGA kit (Sigma-Aldrich, St. Louis, MO, USA), conventional Xq microsatellite marker polymerase chain reaction, gel electrophoresis, and visualization by gel electrophoresis. His karyotype revealed mos 47,XY,+mar[25]/46,XY[25] on 50 metaphase chromosomes (
Fig. 1). To identify the origin of the marker chromosome, nucleolar organizing region staining and fluorescence
in situ hybridization (FISH) using whole chromosome paint Y probe (Vysis, Abbott Park, IL, USA) was performed with negative results. Additional FISH analysis with a CEP 1 (D1Z1) probe (Vysis) showed that the marker chromosome originated from the centromeric region of chromosome 1 (
Fig. 2).
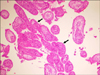
A diagnostic testicular biopsy was performed. The histology of the biopsy revealed different-sized seminiferous tubules with thick basement membranes containing Sertoli cells without germ cells and moderate hyperplasia of the Leydig cells (
Fig. 3). The patient underwent genetic counseling, and microsurgical testicular sperm extraction was discussed. However, he was lost to further follow-up.
DISCUSSION
This was a very rare case of male infertility associated with a mosaic marker chromosome 1. The most common sSMC in humans is derived from chromosome 15, accounting for as much as 50% to 60% of all sSMCs observed [
7]. The significance of a marker chromosome varies, depending on the material contained within the marker. Sometimes, the marker is composed of inactive genetic material and has little or no effect. Often, markers are not passed down, as they can be lost during cell division due to their small size. Conducting specialized studies to identify the genetic material allows more conclusions to be drawn about the effects of the marker.
Spermatogenesis is governed by the parallel and serial actions of thousands of genes. Alterations in any of these genes or their expression may cause spermatogenic failure. The formation of normal bivalents during meiosis is disrupted in patients with structural abnormalities (mainly with respect to translocations), leading to impaired meiosis and the arrest of spermatogenesis at the maturation stage [
8]. Since the transmission of a haploid chromosomal asset is fundamental for embryonic vitality and development, the study of genetic causes of severe male infertility is very important, especially because recent assisted reproductive technologies, such as ICSI could allow fertilization even in cases of severe male infertility. Genetic causes of male infertility include karyotypic abnormalities, Y chromosome microdeletions, and genetic mutations. In general, chromosomal abnormalities account for approximately 5% of infertility in males, and the prevalence increases to 15% in the population of azoospermic males. Therefore, chromosomal errors are a pertinent area of research to determine the role of genetic factors in male factor infertility [
910].
Proper genetic counseling is recommended for infertile patients with a marker chromosome. If needed, surgical sperm retrieval and the possibility of attempting to achieve a pregnancy with the patient's sperm may be discussed prudently. Preimplantation genetic screening to identify at-risk embryos also should be considered.
Figures and Tables
Fig. 1
Karyotyping (GTG banding, ×1,000) revealed a mos 47,XY,+mar[25]/46,XY[25] with a supernumerary marker chromosome of unknown origin.
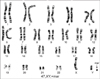
Fig. 2
Fluorescence in situ hybridization analysis with a CEP 1 (D1Z1) probe showed that the marker chromosome originated from the centromeric region of chromosome 1.

Fig. 3
The histology of a testes biopsy revealed different-sized seminiferous tubules with thick basement membranes containing Sertoli cells with minimal germ cells (arrows) (H&E ×100).
ACKNOWLEDGEMENTS
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant No. HI14C0106020014).
References
1. Ferlin A, Raicu F, Gatta V, Zuccarello D, Palka G, Foresta C. Male infertility: role of genetic background. Reprod Biomed Online. 2007; 14:734–745.

2. Carrell DT, De Jonge C, Lamb DJ. The genetics of male infertility: a field of study whose time is now. Arch Androl. 2006; 52:269–274.

3. O'Flynn O'Brien KL, Varghese AC, Agarwal A. The genetic causes of male factor infertility: a review. Fertil Steril. 2010; 93:1–12.
4. Silber SJ, Repping S. Transmission of male infertility to future generations: lessons from the Y chromosome. Hum Reprod Update. 2002; 8:217–229.

5. Manvelyan M, Riegel M, Santos M, Fuster C, Pellestor F, Mazaurik ML, et al. Thirty-two new cases with small supernumerary marker chromosomes detected in connection with fertility problems: detailed molecular cytogenetic characterization and review of the literature. Int J Mol Med. 2008; 21:705–714.

6. Buckton KE, O'Riordan ML, Ratcliffe S, Slight J, Mitchell M, McBeath S, et al. A G-band study of chromosomes in liveborn infants. Ann Hum Genet. 1980; 43:227–239.

7. Koç A, Onur SO, Ergün MA, Perçin EF. Supernumerary marker chromosome 15 in a male with azoospermia and open bite deformity. Asian J Androl. 2009; 11:617–622.

8. Carrell DT. Contributions of spermatozoa to embryogenesis: assays to evaluate their genetic and epigenetic fitness. Reprod Biomed Online. 2008; 16:474–484.

9. Oates RD. The genetic basis of male reproductive failure. Urol Clin North Am. 2008; 35:257–270. ix

10. Brugh VM 3rd, Maduro MR, Lamb DJ. Genetic disorders and infertility. Urol Clin North Am. 2003; 30:143–152.








