Eosinophilic myositis (EM) is a collective term used in meat inspection to designate diseases of clinically healthy animals that have focal, greencolored muscular lesions of unknown origin [7,10]. EM is a relatively rare condition in cattle and sheep of all ages [7,8]. Previous reports have provided evidence that Sarcocystis are directly associated with the lesions and contribute to rejection and down-grading of carcasses at meat-processing plants [1,2,5]. Bovine EM has been only rarely described. The cause of EM remains unknown since most cases were detected during a routine postmortem inspection and because there is little tissue reaction in the intermediate host, cattle, in which asexual development of Sarcocystis occurs.
Presently, we describe a case of EM in a 3-year-old cow with a normal history and no pre-existing clinical conditions at the time of slaughter. A routine postmortem inspection of the carcass revealed generalized whitish and pale skeletal muscles in the cervical region (Fig. 1). Carcasses with locally whitish and pale lesions that had been rejected by inspectors were acquired for comparative purposes. Representative sections of cervical skeletal muscles were fixed immediately in 10% neutral buffered formalin, processed routinely, and embedded in paraffin. Tissue sections 4 µm in thickness were cut and stained with hematoxylin and eosin and periodic acid-Schiff (PAS) for detection of pathogen.
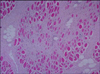
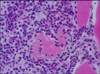
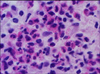
Histopathological examination of the lesions revealed interstitial fibrosis, more developed fibrous septa, and augmented numbers of fibroblasts between the myofibers as compared with normal skeletal muscles (Fig. 2). The carcass was whitish and pale (Fig. 1). The skeletal muscles showed histopathological features of eosinophilic myositis that contained focal granulomatous lesions with multifocal calcified necrotic fibers, mixed inflammatory cells mainly consistent with eosinophils, and the infiltration of a multinucleated giant cell. Massive intramuscular infiltration of the eosinophils was identified in the specimens; as well, most myofibers were hypercontracted, ruptured, and were replaced by eosinophils (Fig. 3). Eosinophils were present within and adjacent to the affected myofibers (Fig. 4). There was no evidence of infection from parasites, or sarcocystosis. Especially considering the appearance of tissues following PAS staining, a diagnosis of bovine idiopathic EM was made.
EM is a group of three inflammatory disorders - eosinophilic polymyositis, eosinophilic perimyositis and focal eosinophilic myositis - characterized pathologically by an eosinophilic cell infiltrate in the skin and skeletal muscles [4,6]. The pathogenesis of EM varies; a correlation between serum interleukin-5 levels and disease severity [9] suggests a role for this interleukin species in eosinophil activation.
Sarcocystis spp. has long been suspected as an etiological agent of EM [7]. Recent reports have provided evidence that sarcocysts are directly associated with lesions and contribute to the rejection and down grading of carcasses at meat-processing plants [1,2,5,10]. In one study, up to 5% of carcasses rejected in the United States were positive for sarcocysts [5]. Presently, generalized inflammatory reactions were common in sections of all the acquired rejected carcasses, and sections of all lesions revealed granulomatous reactions. Most of the granulomas observed in the EM carcasses may have resulted from sarcocysts that served as chronic inflammatory stimuli. It is not surprising that sarcocysts were found in only a small proportion of granulomas.
Currently, there are no tests that can predict the presence of EM prior to slaughter. Immunoglobulins that are specific to species of Sarcocystis have been found in both affected and unaffected cattle [2]. Speciation of the responsible pathogen in cattle is unclear. Regarding the respective occurrence of EM and sarcocytosis, EM has little economic importance while sarcocystosis has great economic importance to the meat-producing and meat packing industries, and to the overall public health.
There have been a few reports of carcasses with numerous disseminated lesions that were rejected as human food sources and presented a public health threat. Although carcasses with a few localized or excisable lesions can be approved, nearly all grossly affected carcasses are assumed to contain disseminated lesions, even though such lesions may be difficult to find.
In the present study, a cow with pale and whitish skeletal muscles grossly evident in the loggisimus capitus was observed at the slaughter house. Histosections of carcass tissue revealed lesions that were characterized by a massive infiltration of eosinophils between the myofibers, hypercontraction and rupture of the muscle bundles, and augmented fibrous septae. As well, focally granulomatous inflammatory lesions were observed. Our case may well represent Sarcocystis spp. EM, although the direct cause was not ascertained.
When confronted by EM, we suggest that not only rejected carcass should be examined, but the entire animal stock of the farm as well. While it is likely that the cause of EM a Sarcocystis spp. infection, livestock officials can prevent or reduce sarcocystosis by controlling the movement of working dogs and cats, and eliminating stray and wild animals from cattle and sheep pastures, feedlots, and feedmills to avoid feed and water-being contaminated with sporocysts. Eliminating bovine and ovine raw muscles and viscera from the diets of dogs and cats is prudent to prevent infecting the definitive hosts. Moreover, human defecation in or near feed or water that could be consumed by cattle should be restricted so as to avoid the transmission of Sarcocystis spp. oocysts to cattle.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download