Abstract
Purpose
Materials and Methods
Results
Conclusion
ACKNOWLEDGEMENTS
References
SUPPLEMENTARY MATERIALS
Supplementary Table 1
Supplementary Table 3
Supplementary Table 4
Supplementary Table 8
Supplementary Table 10
Fig. 2
Comparisons of ODI scores for functional improvement and VAS scores for back pain and leg pain between minimally invasive and open lumbar spinal fusion. Heterogeneity: ODI score [τ2=0.000; χ2=2.549, df=4 (p=0.636); I2=0.0%], VAS back pain [τ2=0.000; χ2=1.192, df=4 (p=0.879); I2=0.0%], and VAS leg pain [τ2=0.000; χ2=0.748, df=1 (p=0.387); I2=0.0%]. ODI, Oswestry Disability Index; VAS, visual analogue scale; df, degrees of freedom; CI, confidence interval.
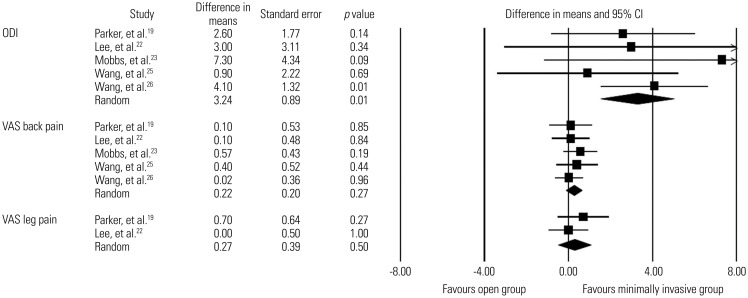
Fig. 3
Comparison of fusion rates between minimally invasive and open lumbar spinal fusion. Heterogeneity: fusion rates for PLF [τ2=0.000; χ2=0.000, df=0 (p=1.000); I2=0.0%], fusion rates for PLIF [τ2=0.000; χ2=0.701, df=1 (p=0.402); I2=0.0%], fusion rates for TLIF [τ2=0.000; χ2=1.836, df=4 (p=0.766); I2=0.0%], and overall fusion rates [τ2=0.000; χ2=3.925, df=7 (p=0.788); I2=0.0%]. PLF, posterolateral fusion; PLIF, posterior lumbar interbody fusion; TLIF, transforaminal lumbar interbody fusion; df, degrees of freedom; OR, odds ratio; CI, confidence interval.
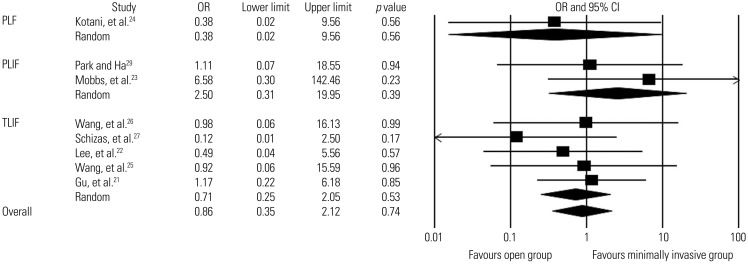
Fig. 4
Comparison of complications rates (hardware-related, neurological, and surgical-site complications) between minimally invasive and open lumbar spinal fusion. Heterogeneity: hardware-related complications [τ2=0.000; χ2=4.922, df=10 (p=0.896); I2=0.0%], neurological complications [τ2=0.000; χ2=3.909, df=9 (p=0.917); I2=0.0%], surgical-site complications [τ2=0.000; χ2=5.232, df=8 (p=0.732); I2=0.0%], and overall complication rates [τ2=0.000; χ2=16.605, df=29 (p=0.968); I2=0.0%]. df, degrees of freedom; OR, odds ratio; CI, confidence interval.
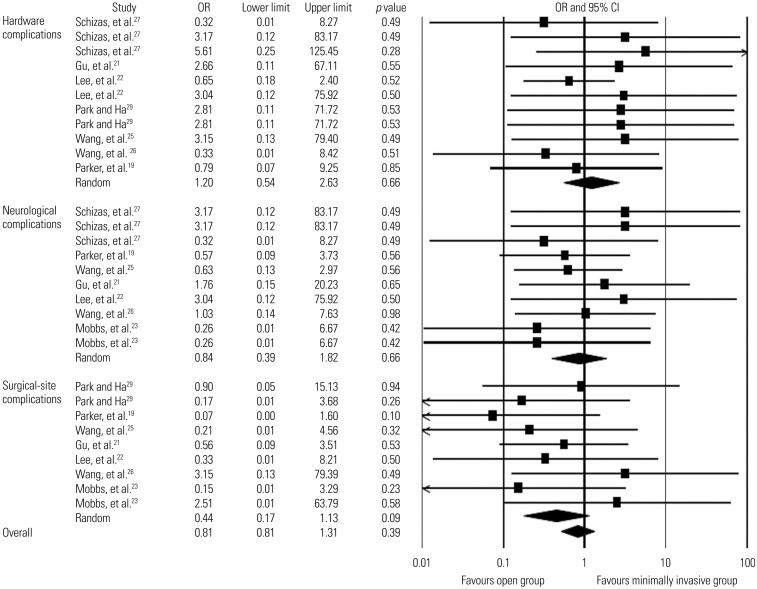
Fig. 5
Comparison of infection rates between minimally invasive and open lumbar spinal fusion. Heterogeneity: τ2=0.000; χ2=2.197, df=6 (p=0.901); I2=0.0%. df, degrees of freedom; CI, confidence interval; OR, odds ratio.
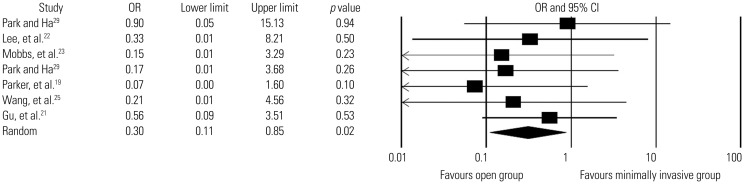
Fig. 6
Comparison of subsequent surgery rates between minimally invasive and open lumbar spinal fusion. Heterogeneity: Removal [τ2=1.204; χ2=2.938, df=2 (p=0.230); I2=31.9%], reoperation [τ2=0.774; χ2=3.965, df=3 (p=0.265); I2=24.3%], revision [τ2=0.000; χ2=1.154, df=3 (p=0.764); I2=0.0%], overall subsequent surgery rates [τ2=0.000; χ2=8.565, df=10 (p=0.574); I2=0.0%]. df, degrees of freedom; OR, odds ratio; CI, confidence interval.
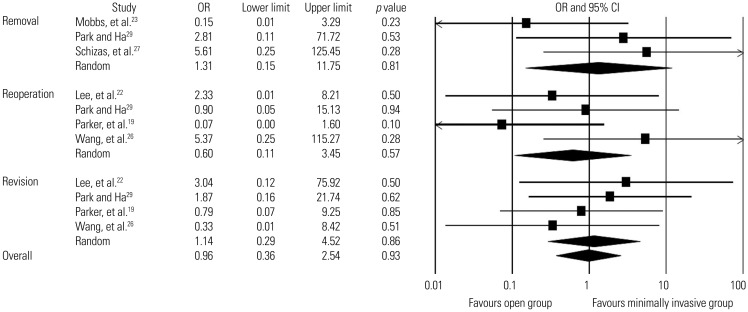
Fig. 7
Comparison of blood loss between minimally invasive and open lumbar spinal fusion. Heterogeneity: τ2=0.747; χ2=57.666, df=5 (p<0.0001); I2=91.3%. df, degrees of freedom; CI, confidence interval.
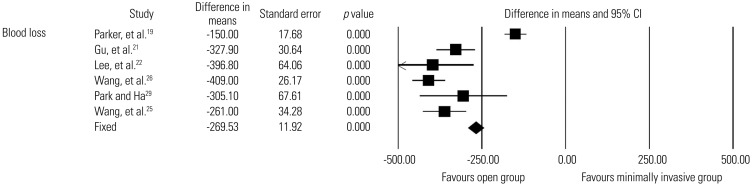
Fig. 8
Comparison of hospital stay, operation time, and radiation exposure time between minimally invasive and open lumbar spinal fusion. Heterogeneity: hospital stay [τ2=0.236; χ2=26.011, df=5 (p<0.0001); I2=80.8%], operation time [τ2=0.297; χ2=40.069, df=6 (p<0.0001); I2=85.0%], and radiation exposure time [τ2=0.240; χ2=14.309, df=3 (p=0.003); I2=79.0%]. df, degrees of freedom; CI, confidence interval.
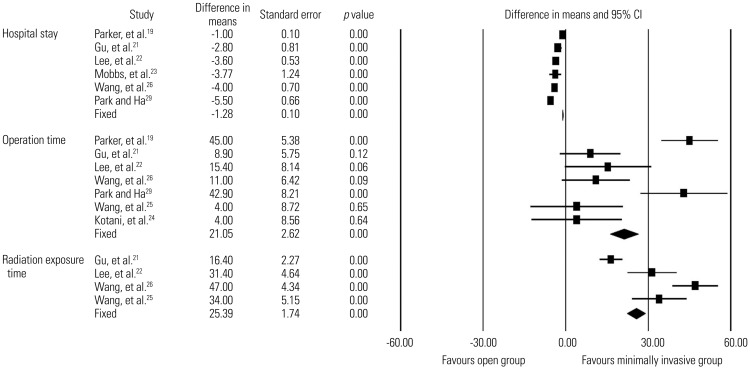
Table 1
Inclusion and Exclusion Criteria
Table 2
Characteristics of All Included Comparative Observational Studies
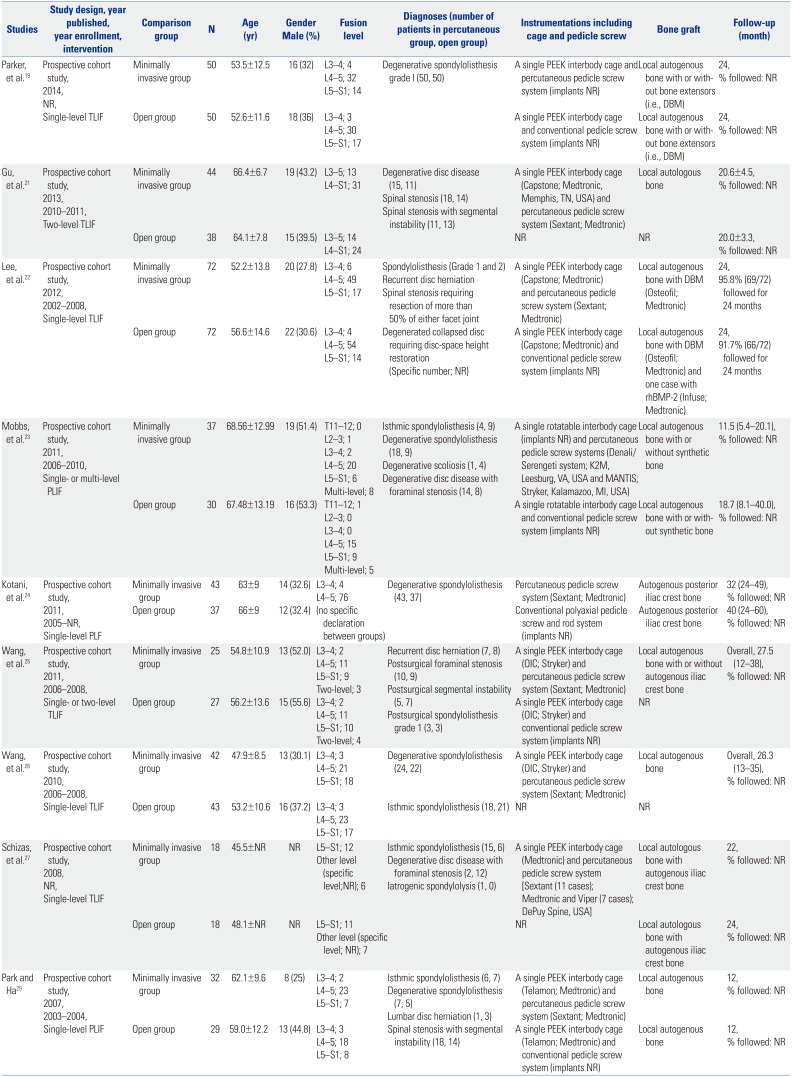
| Studies | Study design, year published, year enrollment, intervention | Comparison group | N | Age (yr) | Gender Male (%) | Fusion level | Diagnoses (number of patients in percutaneous group, open group) | Instrumentations including cage and pedicle screw | Bone graft | Follow-up (month) |
|---|---|---|---|---|---|---|---|---|---|---|
| Parker, et al.19 | Prospective cohort study, 2014, NR, Single-level TLIF | Minimally invasive group | 50 | 53.5±12.5 | 16 (32) | L3–4; 4 | Degenerative spondylolisthesis grade I (50, 50) | A single PEEK interbody cage and percutaneous pedicle screw system (implants NR) | Local autogenous bone with or without bone extensors (i.e., DBM) | 24, % followed: NR |
| L4–5; 32 | ||||||||||
| L5–S1; 14 | ||||||||||
| Open group | 50 | 52.6±11.6 | 18 (36) | L3–4; 3 | A single PEEK interbody cage and conventional pedicle screw system (implants NR) | Local autogenous bone with or without bone extensors (i.e., DBM) | 24, % followed: NR | |||
| L4–5; 30 | ||||||||||
| L5–S1; 17 | ||||||||||
| Gu, et al.21 | Prospective cohort study, 2013, 2010–2011, Two-level TLIF | Minimally invasive group | 44 | 66.4±6.7 | 19 (43.2) | L3–5; 13 |
Degenerative disc disease (15, 11) Spinal stenosis (18, 14) Spinal stenosis with segmental instability (11, 13) |
A single PEEK interbody cage (Capstone; Medtronic, Memphis, TN, USA) and percutaneous pedicle screw system (Sextant; Medtronic) | Local autologous bone | 20.6±4.5, % followed: NR |
| L4–S1; 31 | ||||||||||
| Open group | 38 | 64.1±7.8 | 15 (39.5) | L3–5; 14 | NR | NR | 20.0±3.3, % followed: NR | |||
| L4–S1; 24 | ||||||||||
| Lee, et al.22 | Prospective cohort study, 2012, 2002–2008, Single-level TLIF | Minimally invasive group | 72 | 52.2±13.8 | 20 (27.8) | L3–4; 6 |
Spondylolisthesis (Grade 1 and 2) Recurrent disc herniation Spinal stenosis requiring resection of more than 50% of either facet joint |
A single PEEK interbody cage (Capstone; Medtronic) and percutaneous pedicle screw system (Sextant; Medtronic) | Local autogenous bone with DBM (Osteofil; Medtronic) | 24, 95.8% (69/72) followed for 24 months |
| L4–5; 49 | ||||||||||
| L5–S1; 17 | ||||||||||
| Open group | 72 | 56.6±14.6 | 22 (30.6) | L3–4; 4 | Degenerated collapsed disc requiring disc-space height restoration (Specific number; NR) | A single PEEK interbody cage (Capstone; Medtronic) and conventional pedicle screw system (implants NR) | Local autogenous bone with DBM (Osteofil; Medtronic) and one case with rhBMP-2 (Infuse; Medtronic) | 24, 91.7% (66/72) followed for 24 months | ||
| L4–5; 54 | ||||||||||
| L5–S1; 14 | ||||||||||
| Mobbs, et al.23 | Prospective cohort study, 2011, 2006–2010, Single- or multi-level PLIF | Minimally invasive group | 37 | 68.56±12.99 | 19 (51.4) | T11–12; 0 |
Isthmic spondylolisthesis (4, 9) Degenerative spondylolisthesis (18, 9) Degenerative scoliosis (1, 4) Degenerative disc disease with foraminal stenosis (14, 8) |
A single rotatable interbody cage (implants NR) and percutaneous pedicle screw systems (Denali/ Serengeti system; K2M, Leesburg, VA, USA and MANTIS; Stryker, Kalamazoo, MI, USA) | Local autogenous bone with or without synthetic bone | 11.5 (5.4–20.1), % followed: NR |
| L2–3; 1 | ||||||||||
| L3–4; 2 | ||||||||||
| L4–5; 20 | ||||||||||
| L5–S1; 6 | ||||||||||
| Multi-level; 8 | ||||||||||
| Open group | 30 | 67.48±13.19 | 16 (53.3) | T11–12; 1 | A single rotatable interbody cage and conventional pedicle screw system (implants NR) | Local autogenous bone with or without synthetic bone | 18.7 (8.1–40.0), % followed: NR | |||
| L2–3; 0 | ||||||||||
| L3–4; 0 | ||||||||||
| L4–5; 15 | ||||||||||
| L5–S1; 9 | ||||||||||
| Multi-level; 5 | ||||||||||
| Kotani, et al.24 | Prospective cohort study, 2011, 2005–NR, Single-level PLF | Minimally invasive group | 43 | 63±9 | 14 (32.6) | L3–4; 4 | Degenerative spondylolisthesis (43, 37) | Percutaneous pedicle screw system (Sextant; Medtronic) | Autogenous posterior iliac crest bone | 32 (24–49), % followed: NR |
| Open group | 37 | 66±9 | 12 (32.4) | L4–5; 76 (no specific declaration between groups) | Conventional polyaxial pedicle screw and rod system (implants NR) | Autogenous posterior iliac crest bone | 40 (24–60), % followed: NR | |||
| Wang, et al.25 | Prospective cohort study, 2011, 2006–2008, Single- or two-level TLIF | Minimally invasive group | 25 | 54.8±10.9 | 13 (52.0) | L3–4; 2 |
Recurrent disc herniation (7, 8) Postsurgical foraminal stenosis (10, 9) Postsurgical segmental instability (5, 7) Postsurgical spondylolisthesis grade 1 (3, 3) |
A single PEEK interbody cage (OIC; Stryker) and percutaneous pedicle screw system (Sextant; Medtronic) | Local autogenous bone with or without autogenous iliac crest bone | Overall, 27.5 (12–38), % followed: NR |
| L4–5; 11 | ||||||||||
| L5–S1; 9 | ||||||||||
| Two-level; 3 | ||||||||||
| Open group | 27 | 56.2±13.6 | 15 (55.6) | L3–4; 2 | A single PEEK interbody cage (OIC; Stryker) and conventional pedicle screw system (implants NR) | NR | ||||
| L4–5; 11 | ||||||||||
| L5–S1; 10 | ||||||||||
| Two-level; 4 | ||||||||||
| Wang, et al.26 | Prospective cohort study, 2010, 2006–2008, Single-level TLIF | Minimally invasive group | 42 | 47.9±8.5 | 13 (30.1) | L3–4; 3 | Degenerative spondylolisthesis (24, 22) | A single PEEK interbody cage (OIC, Stryker) and percutaneous pedicle screw system (Sextant; Medtronic) | Local autogenous bone | Overall, 26.3 (13–35), % followed: NR |
| L4–5; 21 | ||||||||||
| L5–S1; 18 | ||||||||||
| Open group | 43 | 53.2±10.6 | 16 (37.2) | L3–4; 3 | Isthmic spondylolisthesis (18, 21) | NR | NR | |||
| L4–5; 23 | ||||||||||
| L5–S1; 17 | ||||||||||
| Schizas, et al.27 | Prospective cohort study, 2008, NR, Single-level TLIF | Minimally invasive group | 18 | 45.5±NR | NR | L5–S1; 12 |
Isthmic spondylolisthesis (15, 6) Degenerative disc disease with foraminal stenosis (2, 12) Iatrogenic spondylolysis (1, 0) |
A single PEEK interbody cage (Medtronic) and percutaneous pedicle screw system [Sextant (11 cases); Medtronic and Viper (7 cases); DePuy Spine, USA] | Local autologous bone with autogenous iliac crest bone | 22, % followed: NR |
| Other level (specific level;NR); 6 | ||||||||||
| Open group | 18 | 48.1±NR | NR | L5–S1; 11 | NR | Local autologous bone with autogenous iliac crest bone | 24, % followed: NR | |||
| Other level (specific level; NR); 7 | ||||||||||
| Park and Ha29 | Prospective cohort study, 2007, 2003–2004, Single-level PLIF | Minimally invasive group | 32 | 62.1±9.6 | 8 (25) | L3–4; 2 |
Isthmic spondylolisthesis (6, 7) Degenerative spondylolisthesis (7, 5) Lumbar disc herniation (1, 3) |
A single PEEK interbody cage (Telamon; Medtronic) and percutaneous pedicle screw system (Sextant; Medtronic) | Local autogenous bone | 12, % followed: NR |
| L4–5; 23 | ||||||||||
| L5–S1; 7 | ||||||||||
| Open group | 29 | 59.0±12.2 | 13 (44.8) | L3–4; 3 | Spinal stenosis with segmental instability (18, 14) | A single PEEK interbody cage (Telamon; Medtronic) and conventional pedicle screw system (implants NR) | Local autogenous bone | 12, % followed: NR | ||
| L4–5; 18 | ||||||||||
| L5–S1; 8 |
Table 3
Demographic and Surgical Data between the Two Surgical Groups
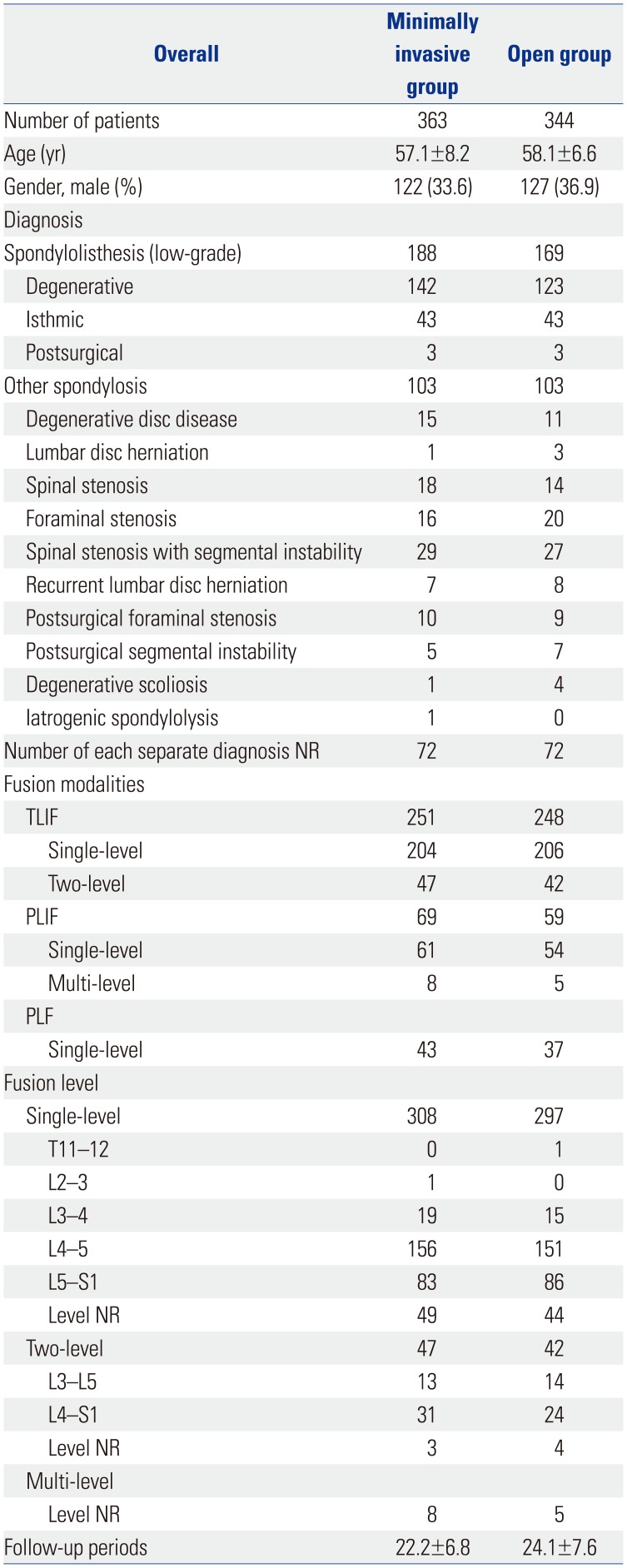
Table 4
Risk of Bias Assessment of Included Comparative Observational Studies
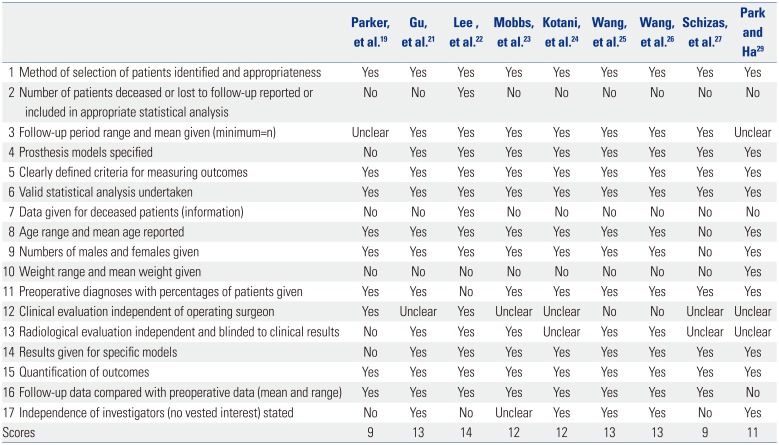
| Parker, et al.19 | Gu, et al.21 | Lee , et al.22 | Mobbs, et al.23 | Kotani, et al.24 | Wang, et al.25 | Wang, et al.26 | Schizas, et al.27 | Park and Ha29 | |
|---|---|---|---|---|---|---|---|---|---|
| 1 Method of selection of patients identified and appropriateness | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 2 Number of patients deceased or lost to follow-up reported or included in appropriate statistical analysis | No | No | Yes | No | No | No | No | No | No |
| 3 Follow-up period range and mean given (minimum=n) | Unclear | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclear |
| 4 Prosthesis models specified | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 5 Clearly defined criteria for measuring outcomes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 6 Valid statistical analysis undertaken | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 7 Data given for deceased patients (information) | No | No | Yes | No | No | No | No | No | No |
| 8 Age range and mean age reported | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes |
| 9 Numbers of males and females given | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes |
| 10 Weight range and mean weight given | No | No | No | No | No | No | No | No | Yes |
| 11 Preoperative diagnoses with percentages of patients given | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes |
| 12 Clinical evaluation independent of operating surgeon | Yes | Unclear | Yes | Unclear | Unclear | No | No | Unclear | Unclear |
| 13 Radiological evaluation independent and blinded to clinical results | No | Yes | Yes | Yes | Unclear | Yes | Yes | Unclear | Unclear |
| 14 Results given for specific models | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 15 Quantification of outcomes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| 16 Follow-up data compared with preoperative data (mean and range) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No |
| 17 Independence of investigators (no vested interest) stated | No | Yes | No | Unclear | Yes | Yes | Yes | No | Yes |
| Scores | 9 | 13 | 14 | 12 | 12 | 13 | 13 | 9 | 11 |
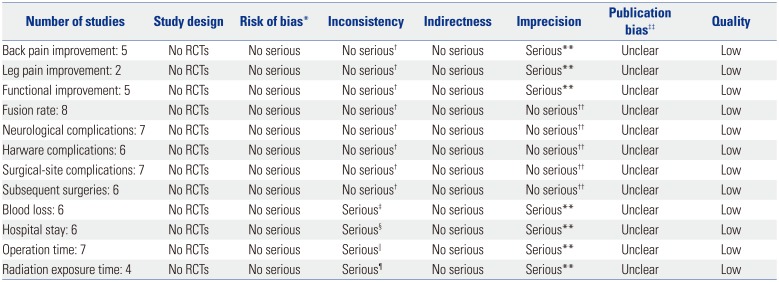
Table 5
The Quality Assessment of Evidence for Each Outcome
RCTs, randomized controlled trials; df, degrees of freedom.
*All studies fulfilled 9 or more criteria of checklist by Cowley and these studies were judged at low risk of bias, †Heterogeneity: I2=0%, ‡Heterogeneity: χ2=89.096, df=5 (p<0.0001); I2=94.4%, §Heterogeneity: χ2=91.483, df=5 (p<0.0001); I2=94.5%, ∥Heterogeneity: χ2=42.123, df=6 (p<0.0001); I2=85.8%, ¶Heterogeneity: χ2=44.986, df=3 (p<0.0001); I2=93.3%, **Weighted mean difference effect size crosses 0.5, ††Odds ratio effect size did not cross 2.5, ‡‡Publication bias was not calculated due to the small number of studies analyzed.
Table 6
Complications and Subsequent Surgeries between the Two Surgical Groups
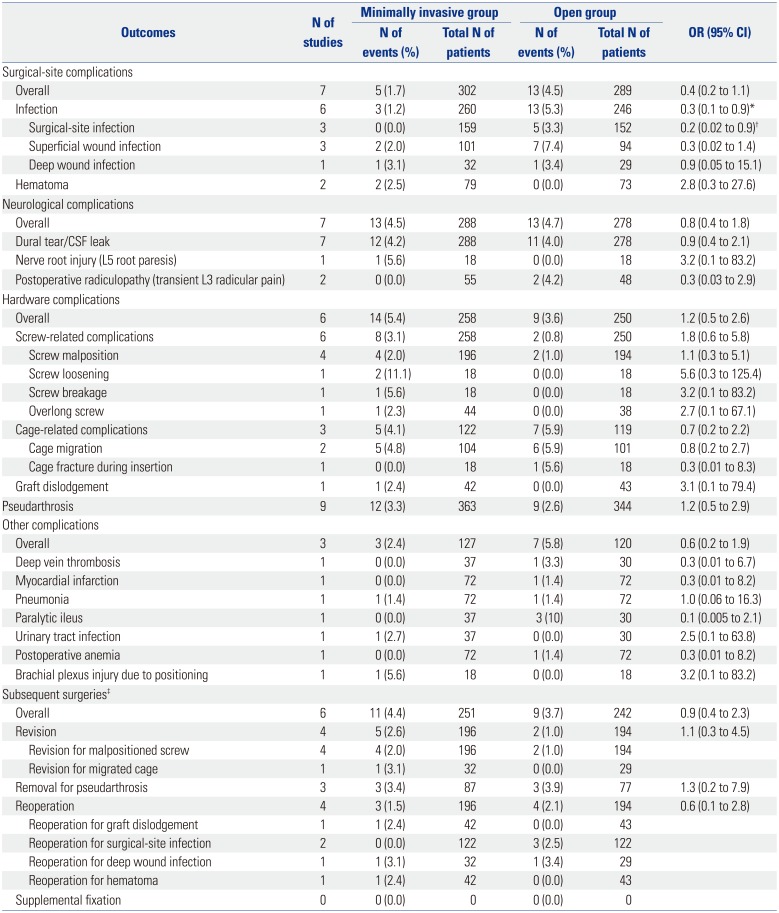
N, number; OR, odds ratio; CI, confidence interval; CSF, cerebrospinal fluid.
*p=0.02, †p=0.04, ‡Complications lead to a subsequent surgical intervention. Subsequent surgical intervention was categorized as follows: a revision is a procedure that adjusts or in any way modifies or removes part of the original implant configuration, with or without replacement of a component; a revision may also include adjusting the position of the original configuration (revision for migrated cage, removal of screws, etc.). A removal is a procedure where all of the original system configuration are removed with or without replacement (removal for pain at operative site but after fusion, for pseudarthrosis, etc.). A reoperation is any surgical procedure at the involved level(s) that does not removal, modification, or addition of any components to the system. A supplemental fixation is a procedure in which additional instrumentation not under study in the protocol is implanted.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download