Abstract
Purpose
To investigate the role of nitric oxide (NO) on the migration of cultured human Tenon's capsule fibroblasts (HTCF) and contraction of collagen gel.
Methods
After artificial wounding, the primarily cultured HTCF were exposed to an NO donor such as sodium nitroprusside (SNP), S-Nitroso-N-acetylpenicillamine (SNAP), or dexamethasone at various concentrations. The cellular migration was measured up to five days. After embedding the cells in the collagen gels, the amount of contraction by the gels was also measured. Cellular survival and NO production were measured with MTT assay and Griess assay, respectively.
Results
Cellular survival was decreased by both NO donors but not by dexamethasone. SNP inhibited migration of HTCF in a dose-dependent manner and enhanced contraction of collagen gels. However, SNAP had no effect on the cellular migration or gel contraction. Dexamethasone inhibited cellular migration but did not affect the contraction of collagen gels.
References
1. Addicks EM, Quigley HA, Green WR, Robin AL. Histologic characteristics of filtering blebs. Arch Ophthalmol. 1983; 101:795–8.
2. Skuta GL, Parrish RK. Wound healing in glaucoma filtering surgery. Surv Ophthalmol. 1987; 32:149–70.

4. Moncada S, Palmer RM, Higgs EA. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev. 1991; 43:109–42.
5. Bredt DS, Snyder SH. Nitric oxide: a physiologic messenger molecule. Annu Rev Biochem. 1994; 63:175–95.

6. Park GC, Kwon NS, Kim YM, Kim JC. The role of nitric oxide in ocular surface diseases. Korean J Ophthalmol. 2001; 15:59–66.

7. Haefliger IO, Dettmann E, Liu R, et al. Potential role of nitric oxide and endothelin in the pathogenesis of glaucoma. Surv Ophthalmol. 1999; 43:S51–8.

8. Nathanson JA, McKee M. Identification of an extensive system of nitric oxide‐ producing cells in the ciliary muscle and outflow pathway of the human eye. Invest Ophthalmol Vis Sci. 1995; 36:1765–73.
9. Nathanson JA, McKee M. Alterations of ocular nitric oxide synthase in human glaucoma. Invest Ophthalmol Vis Sci. 1995; 36:1774–84.
10. Schaffer MR, Tantry U, Gross SS, et al. Nitric oxide regulates wound healing. J Surg Res. 1996; 63:237–40.
11. Kiviluto T, Watanabe S, Hirose M, et al. Nitric oxide donors retard wound healing in cultured rabbit gastric epithelial cell monolayers. Am J Physiol Gastrointest Liver Physiol. 2001; 281:G1151–7.
12. Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J Immunol Methods. 1983; 65:55–63.

13. Green LC, Wagner DA, Glogowski J, et al. Analysis of nitrate, nitrite and [15 N] nitrate in biological fluids. Anal Biochem. 1982; 126:131–8.
14. Liu XD, Skold CM, Umino T, et al. Sodium nitroprusside augments human lung fibroblast collagen gel contraction independently of NO‐ cGMP pathway. Am J Physiol Lung Cell Mol Physiol. 2000; 278:L1032–8.
15. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein‐ dye binding. Anal Biochem. 1976; 72:248–54.
16. Grierson I, Joseph J, Miller M, Day JE. Wound repair: The fibroblast and the inhibition of scar formation. Eye. 1988; 2:135–48.

17. Khaw PT, Occleston NL, Schultz G, et al. Activation and suppression of fibroblast function. Eye. 1994; 8:188–95.

18. Grant MD, Jones RC, Wilson SE, et al. Single dose cephalosporin prophylaxis in high‐ risk patients undergoing surgical treatment of the biliary tract. Surg Gynecol Obstet. 1992; 274:347–54.
19. Kim YM, Bombeck CA, Billiar TR. Nitric oxide as a bifunctional regulator of apoptosis. Circ Res. 1999; 84:253–6.

20. Beckman JS, Koppenol WH. Nitric oxide, superoxide, and peroxynitrite: the good, bad, and ugly. Am J Physiol. 1996; 271:1424–37.
22. Gabbiani GB, Hirschel GB, Ryan PR, et al. Granulation tissue as a contractile organ: a study of structure and function. J Exp Med. 1972; 135:719–33.
23. Elsdale T, Bard J. Collagen substrata for studies on cell behavior. J Cell Biol. 1972; 54:626–37.

24. Bell E, Ivarsson B, Merrill C. Production of a tissue‐ like structure by contraction of collagen lattices by human fibroblasts of different proliferative potential in vitro. Proc Natl Acad Sci U S A. 1979; 76:1274–8.
25. Harris AK, Stopak D, Wild P. Fibroblast traction as a mechanism for collagen morphogenesis. Nature. 1981; 290:249–51.

26. Skold CM, Liu X, Zhu YK, et al. Glucocorticoids augment fibroblast mediated contraction of collagen gels by inhibition of endogenous PGE production. Proc Assoc Am Physicians. 1999; 111:249–58.
27. Kim JW, Kim KH, Cho BJ. Effect of nitric oxide on the proliferation of cultured human Tenon capsule fibroblasts exposed to benzalkonium chloride. J Korean Ophthalmol Soc. 2003; 1908–13.
28. Kanno M, Araie M, Koibuchi H, Masua K. Effects of topical nipradilol, a β blocking agent with α blocking and nitroglycerin‐ like activities, on intraocular pressure and aqueous dynamics in humans. Br J Ophthalmol. 2000; 84:293–9.
29. Mizuno K, Koide T, Yoshimura M, et al. Neuroprotective effect and intraocular penetration of nipradilol, a β– blocker with nitric oxide donative action. Invest Ophthalmol Vis Sci. 2001; 42:688–94.
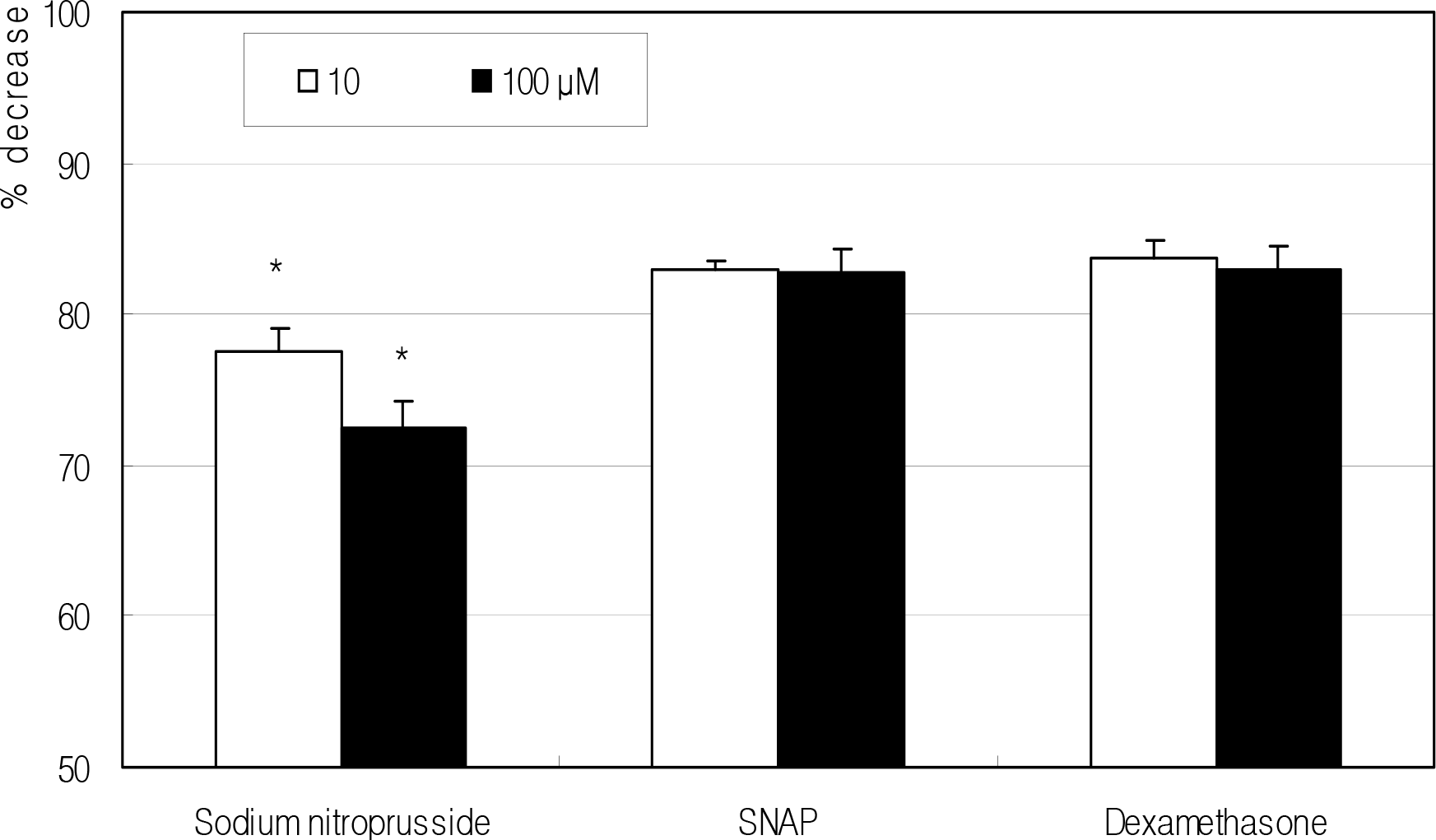
Figure 1.
Effect of NO donors and dexamethasone on the survival of fibroblasts. NO donors decreased cellular survival in a dose-dependent manner compared to non-exposed control. (*; p<0.05)
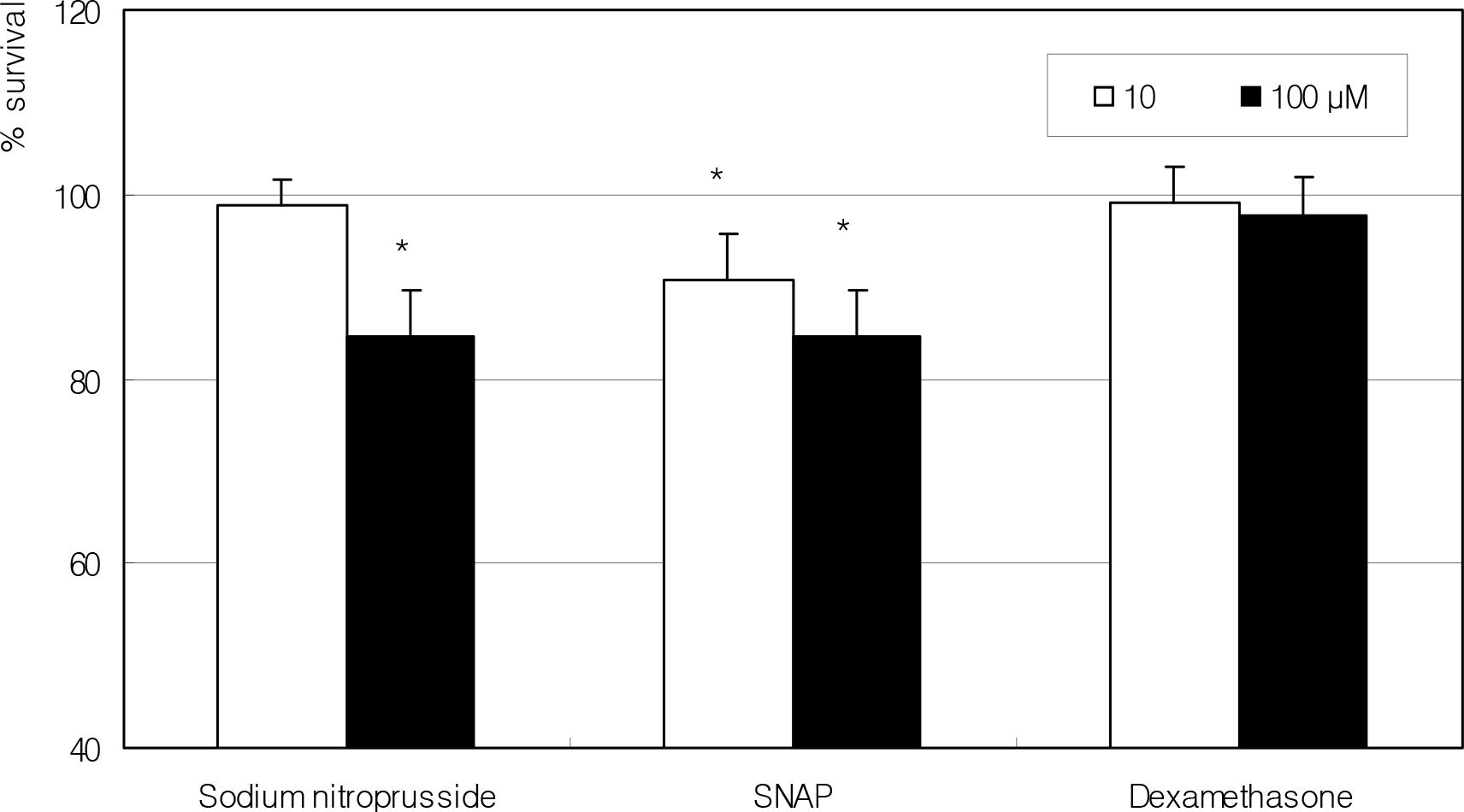
Figure 2.
Effect of NO donors on the production of nitric oxide. Both NO donor increased NO in a dose-dependent manner. (*; p<0.05)
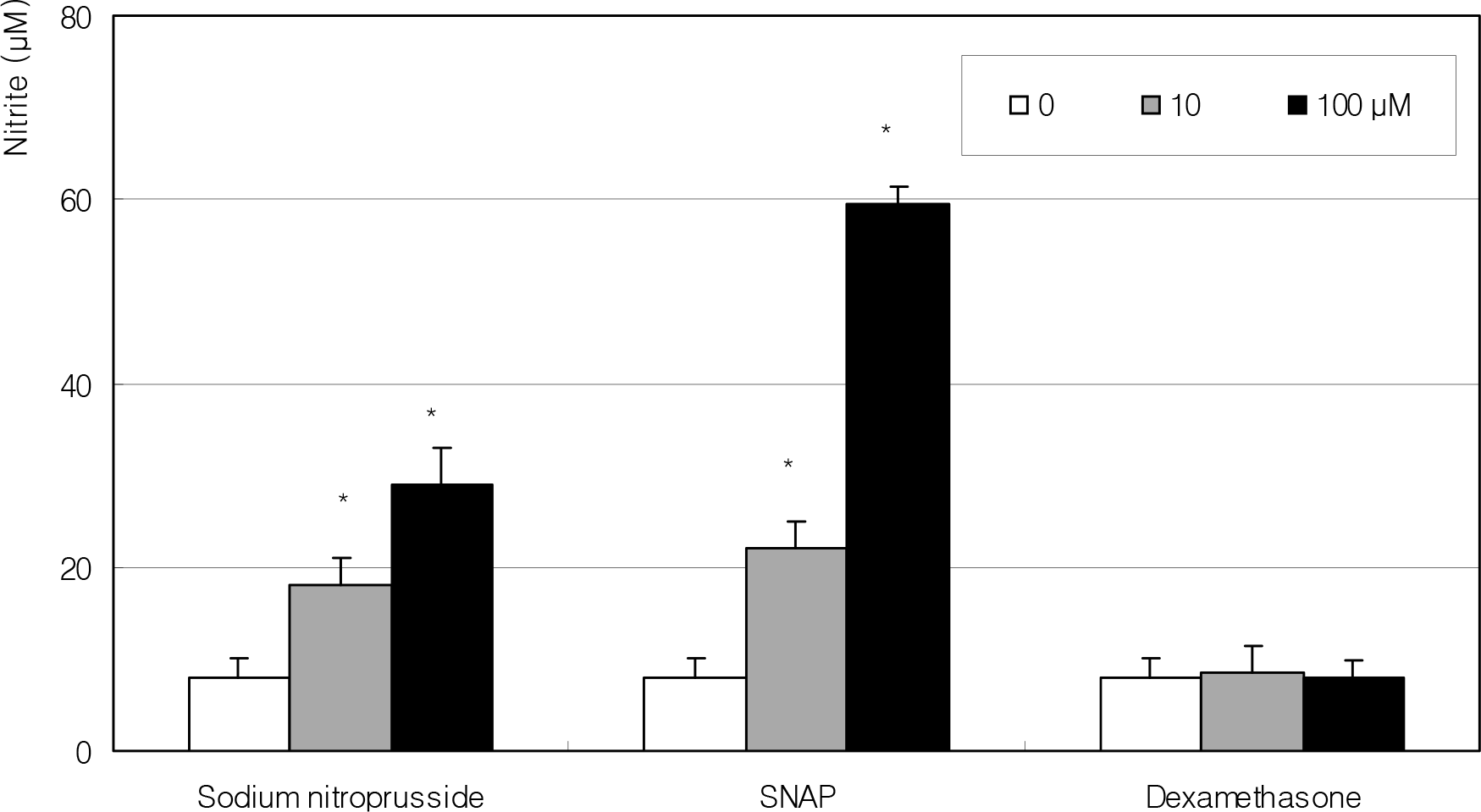
Figure 3.
Effect of sodium nitroprusside on the migration of fibroblasts. Sodium nitroprusside inhibited migration of fibroblasts in a dose-dependent manner from 10 µM of concentration after 3 days. (*; p<0.05)
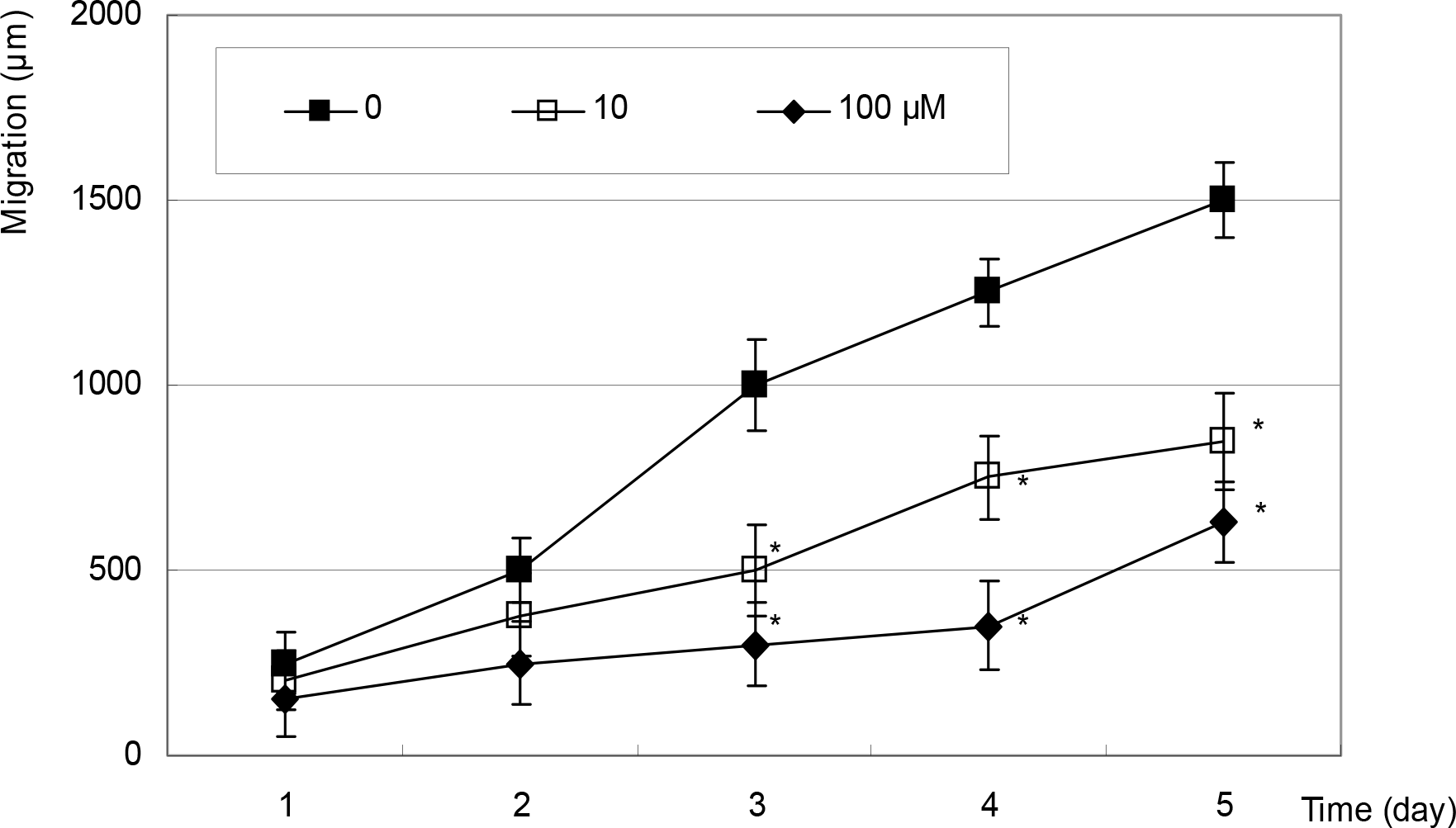
Figure 4.
Effect of SNAP on the migration of fibroblsts. SNAP did not affect on the migration of fibroblasts up to 5 days compared to non-exposed controls.
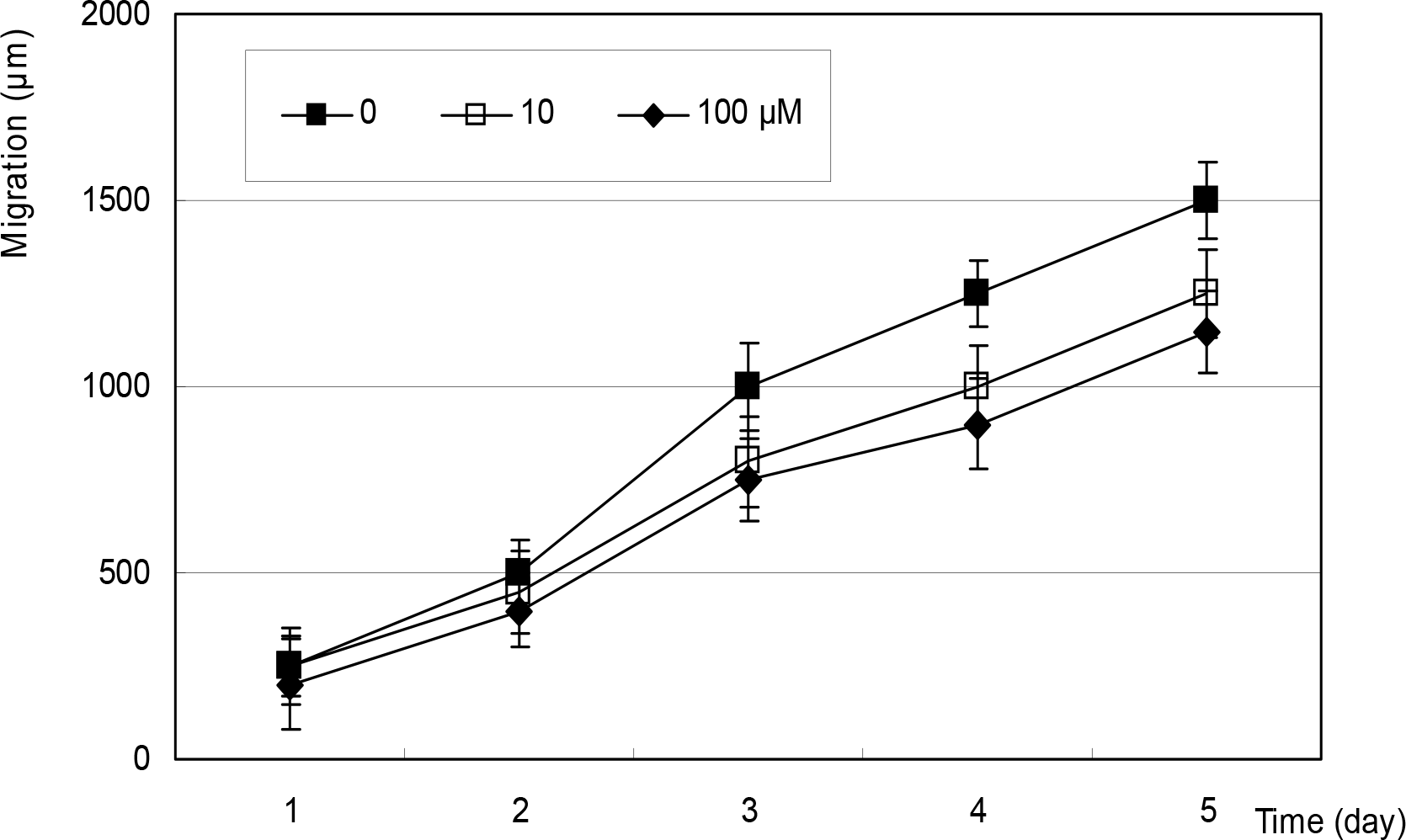
Figure 5.
Effect of dexamethasone on the migration of fibroblasts. Dexamethasone inhibited the migration of fibroblasts after 4 days at 10 µM and after 3 days at 100 µM, compared to non-exposed controls. (*; p<0.05)
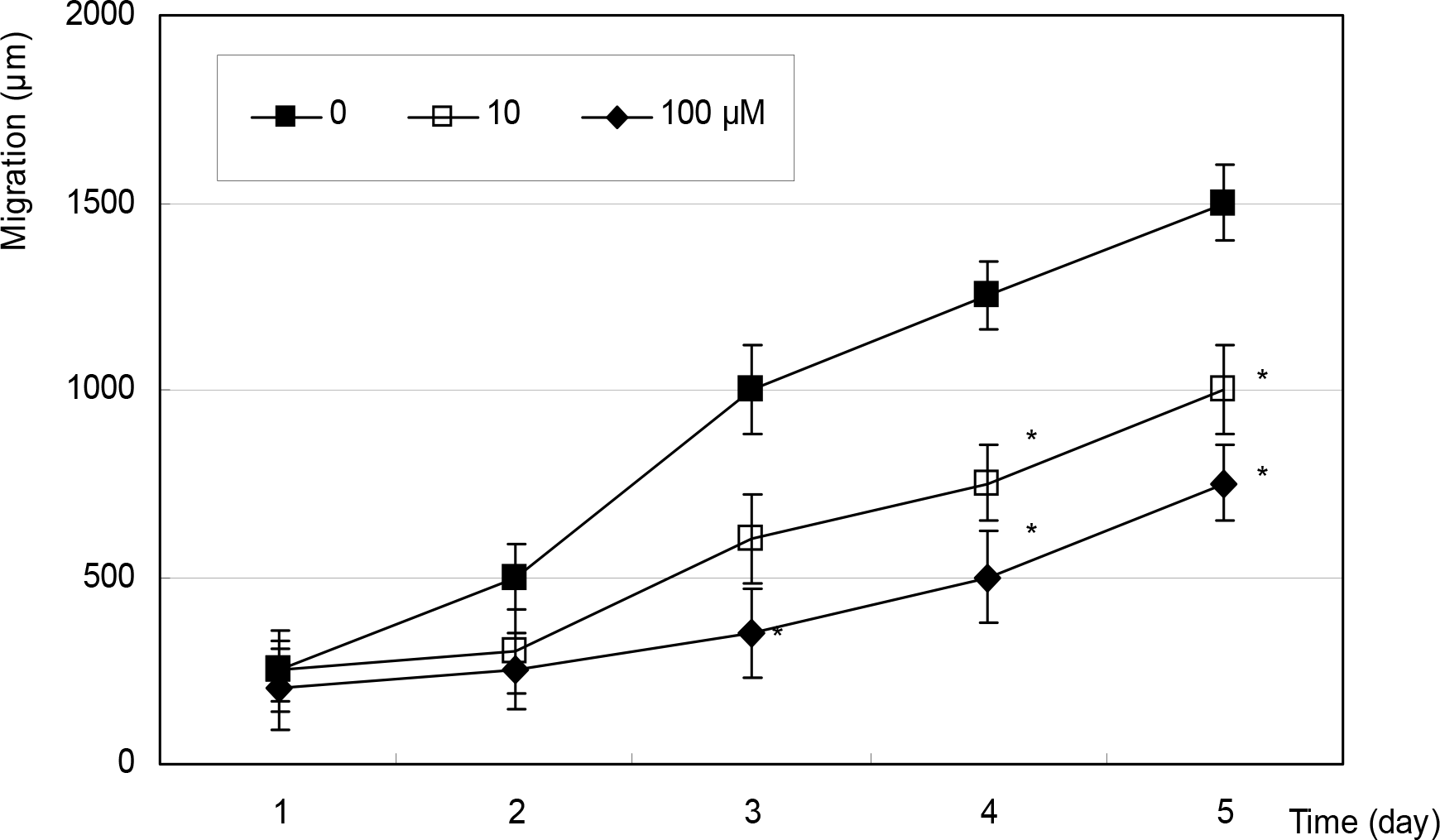
Figure 6.
Effect of sodium nitroprusside on the speed of migration in cultured Tenon fibroblasts. Sodium nitroprusside decreased the speed of migration significantly compared to non-exposed control up to 5 days. (*; p<0.05)
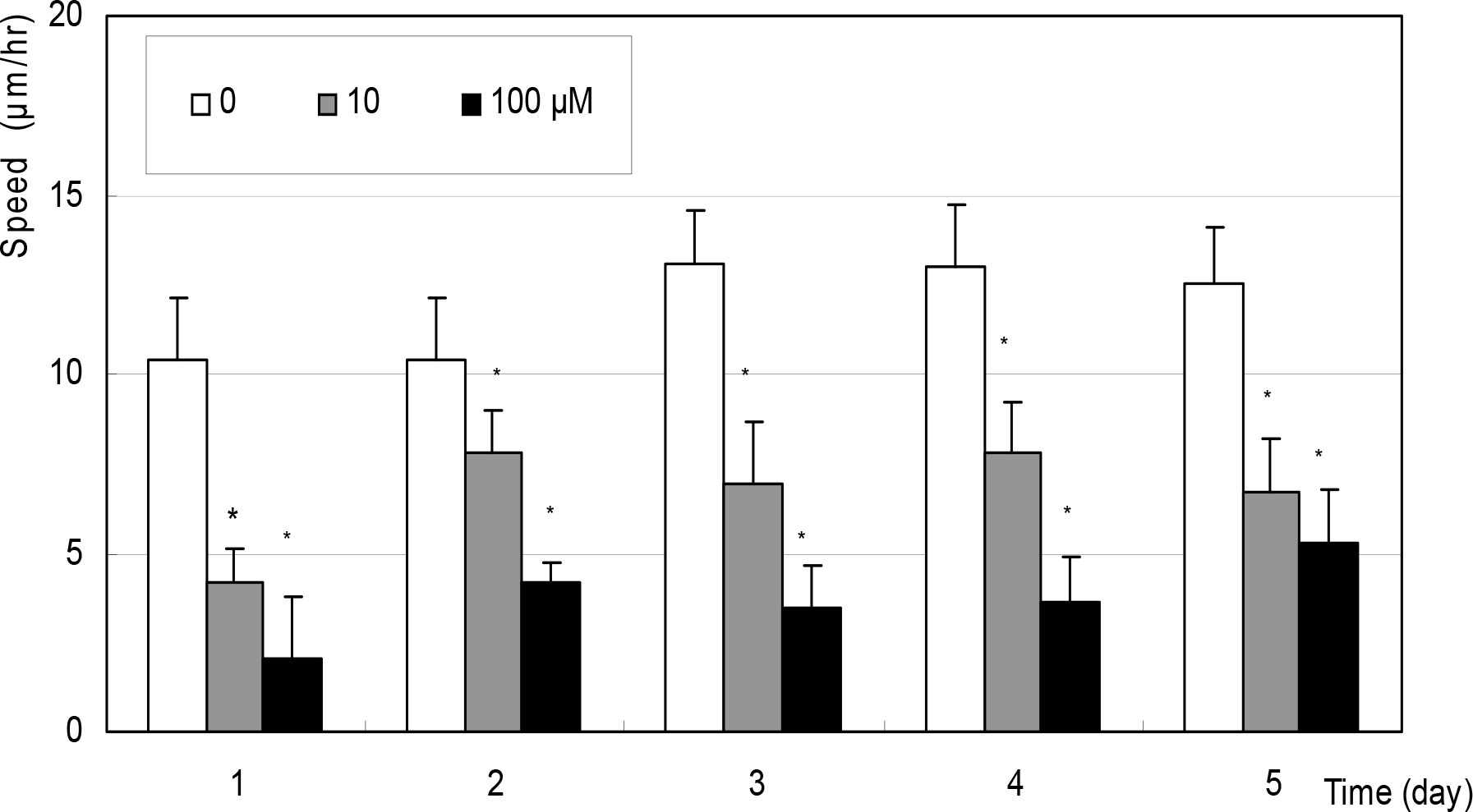
Figure 7.
Effect of SNAP on the speed of migration in cultured Tenon capsule fibroblasts. SNAP did not affect on the speed of migration significantly compared to non-exposed control.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download