Abstract
Purpose
To evaluate the safety and effects of intracameral triamcinolone acetonide injection in rabbit corneas.
Methods
Triamcinolone acetonide in the amounts of 0.5, 1, and 2 mg was injected into the anterior chamber of rabbit eyes, and intraocular pressure, corneal thickness, and endothelial cell counts were evaluated on days 1, 3, 7, 14, and 28. Twenty-eight days after triamcinolone acetonide injection, the eyes were enucleated and examined after TUNEL staining.
Results
No statistically significant differences were found among control, 0.5, and 1 mg triamcinolone-injected eyes in central corneal thickness, endothelial cell density, pleomorphism, and polymegathism. There was no difference between 2 mg triamcinolone-injected eyes and control eyes for corneal thickness and cell density, but there were statistically significant differences between these two groups for pleomorphism (p<0.05) and polymegathism (p<0.05).
References
1. Gordon DM. Prednisone and prednisolone in ocular disease. Am J Ophthalmol. 1956; 41:593–600.
2. Machemer R, Sugita G, Tano Y. Treatment of intraocular proliferations with intravitreal steroids. Trans Am Ophthalmol Soc. 1979; 77:171–80.
3. Kok H, Lau C, Maycock N, et al. Outcome of intravitreal triamcinolone in uveitis. Ophthalmology. 2005; 112:1916.

4. Arevalo JF, Garcia RA, Mendoza AJ. Indocyanine green-mediated photothrombosis with intravitreal triamcinolone acetonide for subfoveal choroidal neovascularization in age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2005; 243:1180–5.

5. Chieh JJ, Roth DB, Liu M, et al. Intravitreal triamcinolone acetonide for diabetic macular edema. Retina. 2005; 25:828–34.

6. Cekic O, Chang S, Tseng JJ, et al. Intravitreal triamcinolone injection for treatment of macular edema secondary to branch retinal vein occlusion. Retina. 2005; 25:851–5.

7. Jonas JB, Kreissig I, Degenring RF. Intravitreal triamcinolone acetonide for pseudophakic cystoid macular edema. Am J Ophthalmol. 2003; 136:384–6.

8. Oh J-Y, Wee WR, Lee JH, Kim MK. Short-term effect of intracameral triamcinolone acetonide on corneal endothelium using the rabbit model. Eye. 2007; 21:812–8.

9. Hernaez‐ Ortega MC, Soto‐ Pedre E. Removal of benzyl alcohol from a commercially available triamcinolone acetonide suspension for intravitreal use. Ophthalmic Surg Lasers Imaging. 2006; 37:162–4.

10. Jonas JB, Degenring RF, Kreissig I, et al. Intraocular pressure elevation after intravitreal triamcinolone acetonide injection. Ophthalmology. 2005; 112:593–8.

11. Chan CK, Fan DS, Chan WM, et al. Ocular-hypertensive response and corneal endothelial changes after intravitreal triamcinolone injections in Chinese subjects: a 6-month follow-up study. Eye. 2005; 19:625–30.

12. Jonas JB, Kreissig I, Degenring RF. Intraocular pressure after intravitreal injection of triamcinolone acetonide. Br J Ophthalmol. 2003; 87:24–7.

13. Vajpayee RB, Sharma N, Dada T, et al. Management of posterior capsule tears. Surv Ophthalmol. 2001; 45:473–88.

14. Akura J, Hatta S, Kaneda S, et al. Management of posterior capsule rupture during phacoemulsification using the dry technique. J Cataract Refract Surg. 2001; 27:982–9.

15. Yamakiri K, Uchino E, Kimura K, Sakamoto T. Intracameral triamcinolone helps to visualize and remove the vitreous body in anterior chamber in cataract surgery. Am J Ophthalmol. 2004; 138:650–2.

16. Jonas JB. Intraocular availability of triamcinolone acetonide after intravitreal injection. Am J Ophthalmol. 2004; 137:560–2.

17. Chen TY, Yang CM, Liu KR. Intravitreal triamcinolone staining observation of residual undetached cortical vitreous after posterior vitreous detachment. Eye. 2006; 20:423–7.

18. Furino C, Micelli Ferrari T, Boscia F, et al. Triamcinolone-assisted pars plana vitrectomy for proliferative vitreoretinopathy. Retina. 2003; 23:771–6.

19. Walker TD. Benzalkonium toxicity. Clin Experiment Ophthalmol. 2004; 32:657.
20. Morrison VL, Koh HJ, Cheng L, et al. Intravitreal toxicity of the kenalog vehicle (benzyl alcohol) in rabbits. Retina. 2006; 26:339–44.

21. Garcia-Arumi J, Boixadera A, Giralt J, et al. Comparison of different techniques for purification of triamcinolone acetonide suspension for intravitreal use. Br J Ophthalmol. 2005; 89:1112–4.

22. Nishimura A, Kobayashi A, Segawa Y, et al. Isolating triamcinolone acetonide particles for intravitreal use with a porous membrane filter. Retina. 2003; 23:777–9.

24. Misra A, Burton RL. Incidence of intraoperative complications during phacoemulsification in vitrectomized and nonvitrectomized eyes: prospective study. J Cataract Refract Surg. 2005; 31:1011–4.

25. Mason JO 3rd, Somaiya MD, Singh RJ. Intravitreal concentration and clearance of triamcinolone acetonide in nonvitrectomized human eyes. Retina. 2004; 24:900–4.

26. Zinn KM. Iatrogenic intraocular injection of depot corticosteroid and its surgical removal using the pars plana approach. Ophthalmology. 1981; 88:13–7.
27. Van Horn DL, Sendele DD, Seideman S, Buco PJ. Regenerative capacity of the corneal endothelium in rabbit and cat. Invest Ophthalmol Visual Sci. 1977; 10:597–613.
Figure 1.
Anterior segment photographs after different concentrations of intracameral triamcinolone acetonide injections at 1, 7, and 28 days after 0.5 mg (A, B, C), 1 mg (D, E, F) and 2 mg (G, H, I) triamcinolone acetonide injection into the anterior chamber.

Figure 2.
Specular micrographs at one day after 1 mg (A) and 2 mg (B) triamcinolone acetonide injection; CD=cell density; CV=coefficient of variation; 6A=hexagonality.
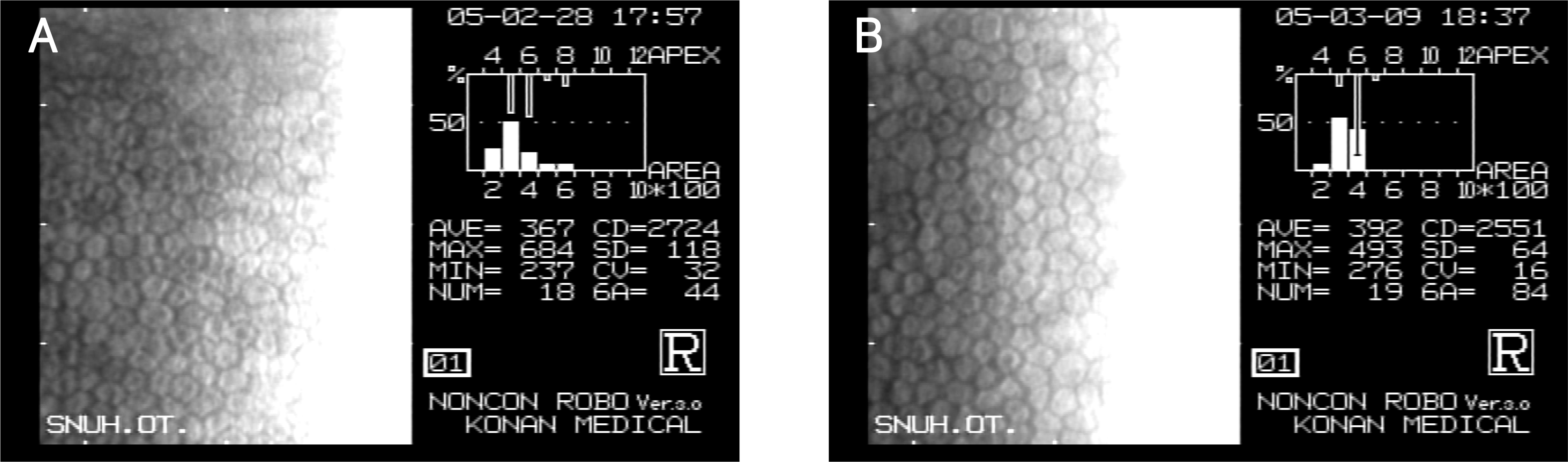
Figure 3.
Hematoxylin & eosin staining (A) and TUNEL staining (B) of the central cornea in control, and in rabbit with 0.5, 1, and 2 mg of triamcinolone acetonide injection at 28 days. (×200)

Table 1.
The average corneal thickness (µm) according to triamcinolone acetonide injection dose and time
Table 2.
The average corneal endothelial cell density (cell/mm2) according to triamcinolone acetonide injection dose and time
Table 3.
The average corneal endothelial polymegathism (coefficient of variation) according to triamcinolone acetonide injection dose and time
| | Pre-operative | Day 1 | Day 3 | Day 7 | Day 14 | Day 28 | Statistical significance |
|---|---|---|---|---|---|---|---|
| BSS (n=5) | 18.0±1.2 | 22.8±3.4 | 19.6±4.6 | 18.2±2.0 | 18.0±1.5 | 19.8±3.1 | NS |
| TA 0.5 mg (n=5) | 19.4±0.8 | 25.0±5.7 | 21.2±3.9 | 20.2±3.2 | 20.5±2.4 | 20.2±4.2 | NS |
| TA 1.0 mg (n=5) | 21.0±1.8 | 27.4±5.5 | 21.4±4.0 | 22.0±3.2 | 21.4±2.8 | 19.8±2.6 | NS |
| TA 2.0 mg (n=5) | 19.8±1.3 | 37.0±4.8 | 26.0±3.1 | 21.4±2.0 | 21.8±2.2 | 20.8±2.9 | P=0.007* P=0.02† |
Table 4.
The average corneal endothelial pleomorphism (%, hexagonality) according to triamcinolone acetonide injection dose and time
| | Pre-operative | Day 1 | Day 3 | Day 7 | Day 14 | Day 28 | Statistical significance |
|---|---|---|---|---|---|---|---|
| BSS (n=5) | 67.6±7.9 | 72.6±4.9 | 72.2±3.8 | 72.0±4.1 | 70.6±3.9 | 71.2±3.1 | NS |
| TA 0.5 mg (n=5) | 66.6±6.9 | 72.8±7.4 | 68.8±4.4 | 68.8±3.2 | 69.0±1.7 | 69.4±2.0 | NS |
| TA 1.0 mg (n=5) | 67.6±8.0 | 68.6±4.5 | 68.6±6.1 | 69.0±6.2 | 70.4±5.3 | 68.6±4.2 | NS |
| TA 2.0 mg (n=5) | 70.0±11.0 | 57.8±5.4 | 61.6±4.8 | 63.8±3.4 | 63.4±3.8 | 69.6±4.8 | P=0.036* P=0.02† |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download