Abstract
There is persuasive epidemiological and experimental evidence that dietary polyphenols have anti-inflammatory activity. Aspirin and other non-steroidal anti-inflammatory drugs (NSAIDs) have long been used to combat inflammation. Recently, cyclooxygenase (COX) inhibitors have been developed and recommended for treatment of rheumatoid arthritis (RA) and osteoarthritis (OA). However, two COX inhibitors have been withdrawn from the market due to unexpected side effects. Because conventional therapeutic and surgical approaches have not been able to fully control the incidence and outcome of many inflammatory diseases, there is an urgent need to find safer compounds and to develop mechanism-based approaches for the management of these diseases. Polyphenols are found in many dietary plant products, including fruits, vegetables, beverages, herbs, and spices. Several of these compounds have been found to inhibit the inflammation process as well as tumorigenesis in experimental animals; they can also exhibit potent biological properties. In addition, epidemiological studies have indicated that populations who consume foods rich in specific polyphenols have lower incidences of inflammatory disease. This paper provides an overview of the research approaches that can be used to unravel the biology and health effects of polyphenols. Polyphenols have diverse biological effects, however, this review will focus on some of the pivotal molecular targets that directly affect the inflammation process.
Inflammation is the immunological defense mechanism by which the body fights infection or injury from bacteria, viruses and other pathogens. The defining clinical features of inflammation are known in Latin as rubor (redness), calor (warmth), tumor (swelling) and dolor (pain). These hallmarks of inflammation were first described by Aurelius Cornelius, a Roman physician and medical writer who lived from about 30 B.C. to 45 A.D. The redness and swelling that occurs at the site of a wound is due to the widening blood vessels to allow specialized white blood cells to enter and battle the pathogens. Following this process, the inflammation fades and healing begins. Excessive or inadequate activation of the system can have serious effects, as can the failure of the inactivation mechanism. In the early 1970s, aspirin and other non-steroidal anti-inflammatory drugs (NSAIDs) were found to inhibit cyclooxygenase (COX), which produces prostaglandins (PGs), a class of physiological and pathophysiological inflammatory mediators.1 Since then, the biosynthetic cascade of arachidonic acid (AA) has been the subject of intense research. This is because AA, having been previously liberated from phospholipids using various stimuli, can be metabolized by the COX pathway into PGs and thromboxane A2, or by the lipoxygenase (LOX) pathways to hydroperoxyeicosateraenoic acids (HpETEs), hydroxyeicosateraenoic acids (HETEs) and leukotrienes (LTs) (Fig. 1). These two proteins play a vital role in inflammation by controlling the intensity and duration of pain, as well as the occurrence of fever, swelling and heat of an affected area. Although steroidal anti-inflammatory drugs and NSAIDs are currently used to treat acute inflammation, these drugs have not been successful in curing chronic inflammatory diseases, such as bronchiectasis, rheumatoid arthritis (RA), osteoarthritis (OA), and inflammatory bowel disease (ulcerative colitis and Crohn's disease). Recently, Vioxx and Celebrex, two blockbuster drugs used to treat arthritis, were withdrawn from the drug market due to their cardiovascular side effects. These cases provide examples of the unknown side effects produced by synthetic drugs, and suggest that an evaluation of the long-term effects of drugs may be required.
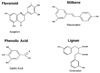
Alternatively, a large number of dietary polyphenols have been consumed in food, and their anti-inflammatory activities have been reported. Polyphenols can be classified into four different groups: flavonoids, stillbenes, lignas, and phenolic acids (Fig. 2). These compounds have received considerable attention from the public. In fact, 12 out of the 40 anti-inflammatory drugs approved between 1983 and 1994 were derived from or based on these natural products.2 These molecules are secondary metabolites of plants and are generally involved in the defense mechanism against ultraviolet radiation and insects. Some of the polyphenols found to be beneficial to health were characterized as naturally-occurring toxicants in plants. In excess, some polyphenols resulted in growth depression in poultry, livestock and laboratory animals. However, the intake of small amounts of polyphenols in food has a potent effect on reducing chronic diseases in animal models. Epidemiological studies have indicated that populations who consume foods rich in specific polyphenols have a lower incidence of chronic inflammatory diseases. Kuhnau et al. calculated that dietary polyphenol intake in the US was ~1 g/day.3 Some studies have subsequently provided more precise individual data regarding the intake of various classes of dietary polyphenols. Flavonols (one of the flavonoids) have been more extensively studied, and the consumption of these substances has been estimated at 20-25 mg/day in the US.4 The plasma concentrations of flavonoids are usually <10 µM and the steady-state levels are typically <1 µM.5 However, the concentrations of total polyphenols may be higher than those of flavonoids.
We have been studying the effects of several dietary polyphenol compounds (including resveratrol, genistein, catechin, and indole-3-carbinol) on chronic diseases in humans, such as inflammation and cancer.6-9 The potential molecular mechanisms of their anti-inflammatory activities have also been suggested to include, but not limited to, the inhibition of enzymes related to inflammation, such as cyclooxygenase and lipoxygenase, and many others including PPAR, NOS, NF-κB, and NAG-1. Thus, polyphenols can influence many pathways, and other compounds may also affect the same pathways. The detailed mechanisms by which each polyphenol induces anti-inflammation remain to be elucidated. In this review, we summarized the current findings of the molecular targets of polyphenols as anti-inflammatory compounds. We focused on two molecular aspects: the arachidonic acid (AA)-dependent pathway and the AA-independent pathway. Cyclooxygenase, lipoxygenase, and PLA2 are discussed as AA-dependent pathway proteins by polyphenols, whereas NOS, NF-κB, PPAR, and NAG-1 are discussed as AA-independent pathway proteins. There is great potential for dietary polyphenols to become the next generation of dietary factors to confer health effects for inflammation beyond synthetic drugs. Further, dietary polyphenols may provide an excellent model system for the development of more effective drugs in the future.
An anti-inflammatory effect is believed to result from inhibiting the formation of prostaglandins by prostaglandin H synthase (COX, also called cyclooxygenase), which converts arachidonic acid (AA) released by membrane phospholipids into prostaglandins. Two isoforms of prostaglandin H synthase, COX-1 and COX-2, have been identified, and one variant form (COX-3) has recently been reported as well.10 COX-1 is constitutively expressed in many tissues, while the expression of COX-2 is regulated by mitogens, tumor promoters, and growth factors.11 NSAIDs are widely used in the treatment of inflammatory diseases. Since aspirin was synthesized, a number of aspirin-like drugs have also been developed. Aspirin and other NSAIDs (also known as conventional NSAIDs that block COX-1 and COX-2) induce gastric ulcerization and kidney failure due to COX-1 inhibition. COX-1 is necessary for the protection of the stomach lining, so interfering with its activity can cause gastrointestinal disturbances ranging from simple discomfort to bleeding ulcers. This hypothesis was confirmed when newly developed drugs that specifically inhibited COX-2 were shown to have a lower rate of gastrointestinal bleeding when compared to conventional NSAIDs.12 However, the "good" COX-1 versus "bad" COX-2 dichotomy has proven overly simplistic. COX-2 has been shown to exert normal physiological effects, including the regulation of vascular and renal blood flows.13 The identification of COX-2 was not expected to increase the effectiveness of NSAID therapy, since conventional NSAIDs inhibited both COX-1 and COX-2. However, the identification of cyclooxygenase 3 (COX-3) may contribute to the elucidation of the inflammatory process.10 COX-3 has also raised many questions by researchers studying the effects of acetaminophen. The weak level of COX-3 inhibition induced by acetaminophen probably does not occur with standard oral dosages of acetaminophen (0.5-1 g/day). Thus, other hypotheses regarding acetaminophen's mechanisms of action should be considered. Although the marketing of new COX-2 inhibitors has emphasized the advantages of not blocking the necessary COX-1 pathway, infrequent reports of gastrointestinal problems still exist from the use of these medications. Both Celebrex and Vioxx were withdrawn from the market in 2005 due to an increase in cardiovascular risks, likely caused by the thromboxane formed via the COX-1 pathway.14 Therefore, a clinical trial that involves patients switching from one synthetic agent to a different agent could be valuable. Several compounds that are consumed daily in various foods may provide alternative tools for treating inflammatory diseases by acting as COX inhibitors.
In 1980, Baumann et al. were the first to report, in a study that assessed rat medullar COX activity, that some dietary polyphenols, such as galangin and luteolin, inhibit AA peroxidation.15 Since then, several researchers have reported that many dietary polyphenols inhibit COX activity at the transcriptional level as well as at the enzyme level. For instance, Landolfi et al. found that flavone, chrysin, apigenein, and phloretin depressed COX activity and inhibited platelet aggregation.16 The flavonoids, 6-hydroxykaempferol, and quercetagenin isolated from T. parthenium (feverfew), and 6-hydroxyluteolin and scutellarein isolated from T. vulgaris (tansy) were shown to inhibit COX activity in leukocytes.17 The triterpenes sasanquol isolated from C. sasanqua (Theaceae) and 3β-p-hydroxybenzoyldehydrotumulosic acid from the fungus P. cocos (Polyporaceae) produced activity against 12-O-tetradecanoylphobol-13-acetate (TPA) and also AA-induced ear inflammation in mice,18 which was probably caused by the COX-2 inhibition. Pre-treatment with green tea extract enriched with catechin and epigallocatechin gallate (EGCG) by gavage inhibited COX-2 expression that was induced by the tumor promoter 12-O-tetradecanoylphorbol-13-acetate (TPA) in mouse skin. Similarly, EGCG down-regulated COX-2 in TPA-stimulated human mammary epithelial cells (MCF-10A) in culture.19 Both the green tea catechin and the EGCG displayed COX inhibition activity in LPS-induced macrophages20 and inhibited IL-1β-dependent pro-inflammatory signal transduction in cultured respiratory epithelial cells.21 The stillbene compound resveratol possesses anti-inflammatory activity because it suppresses carragenen-induced pedal edema via the inhibition of COX activity.22 Furthermore, resveratrol inhibits TPA-induced COX-2 expression at the transcriptional level.23 Thus, the mechanism by which resveratrol exerts anti-inflammatory activity may be related to the inhibition of either COX transcription or COX activity. Studies using isolated bovine COX-1 and COX-2 enzymes showed that curcumin had significantly higher inhibitory effects on the peroxidase activity of COX-1 than that of COX-2.24 EGCG and resveratrol were the most effective COX-1 inhibitors among those tested in the study.20 Genistein down-regulates COX-2 promoter activity in colon cancer cells transfected with a COX-2 reporter gene system.25 Wogonin and sophoraflavanone-G down-regulate COX-2 expression from TNF-α treated NIH/3T3 cells and LPS treated RAW cells, respectively. This suggests that the COX-2 down-regulation of skin fibroblasts may in part be one of the anti-inflammatory mechanisms these compounds exert against skin inflammation such as atopic dermatitis.26,27 Although many studies have reported that polyphenols inhibit COX-1 or COX-2, it has not yet been reported that polyphenols inhibit COX-3. Collectively, several polyphenols were reported to have anti-inflammatory effects, as assessed by COX inhibition. COX inhibition by polyphenols may account for these anti-inflammatory effects, which reduce prostaglandin synthesis. Therefore, it should be noted that the concurrent use of polyphenols and NSAIDs could be beneficial or deleterious, and thus necessitates constant attention by healthcare providers.
LOXs are the enzymes responsible for generating hydroxyl acids and leukotrienes (LTs) from AA (Fig. 1). There are three distinct LOX isozymes in that affect different cells and tissues. 15-LOX synthesizes anti-inflammatory 15-HETE, 5; 12-LOX is involved in provoking inflammatory/allergic disorders; and 5-LOX produces 5-HETE and LTs, which are potent chemoattractants and lead to the development of asthma. 12-LOX synthesizes 12-HETE, which aggregates platelets and induces the inflammatory response. Therefore, the effect of polyphenols on 5- and 12-LOXs has been extensively studied in order to elucidate the anti-inflammatory properties. Flavonols, including kaempferol, quercetin, morin and myricetin, were found to be 5-LOX inhibitors.28 With IC50 values ranging from 1.0 to 18.7 µM, hamamelitannin and the galloylated proanthocyanidins were found to be the most potent inhibitors of 5-LOX.29 Some prenylated flavonoids, such as artonin E, are the most effective inhibitors of porcine leukocyte 5-LOX.30 An anti-inflammatory neolignan sibyllenone from Ocotea bullata (black stinkwood) was also active against 5-LOX.31 Baicalein was reported to selectively inhibit platelet 5-LOX.32 Curcumin blocks the activity of human recombinant 5-LOX.24 In contrast, there are few reports regarding 12-LOX inhibition. Using bovine PMNs and 12-LOX from bovine platelets, kuwanson C and quercetin potently inhibit 12-LOX activity,33 with IC50 values of 19 and 12 µM, respectively. In comparison, the IC50 value of the known LOX inhibitor nordihydroguaiaretic acid (NDGA) is 2.6 µM.
The LOX pathway produces leukotrienes. When only COX-2 is blocked, the LOX pathway still produces the potent mediators of inflammation. The condition may even be exacerbated by blocking only the inflammatory cascade of COX-2, which allows the LOX branch to accelerate the formation of leukotrienes. Dual inhibition of LOX/COX has been suggested to be a desirable approach in the development of new drugs for anti-inflammation.34 Thus far, researchers have not been able to bypass certain limiting barriers to produce such a medication. This is important since some researchers have found that COX-2 inhibition alone will exacerbate the production of pro-inflammatory cytokines such as TNF-α and IL-1β, both of which cause destructive damage to inflammatory sites. Hong et al. demonstrated that curcumin fits in this category of agents.24 Many other polyphenols are also the inhibitors of both COX and LOX. Subsequently, a general structure rule has been found: more COX inhibitions and less LOX inhibitions with polyphenols contain few hydroxyl substituents.35
Phospholipase A2 (PLA2), the enzyme that cleaves phospholipids producing lysophospholipids and free fatty acids, was originally identified as an intracellular protein involved in cell signaling and in the production of free fatty acids, such as arachidonic acid.36 It is known that PLA2 plays an important role in the inflammation process. The inhibition of PLA2 could be a potential therapeutic agent for curing human inflammatory diseases (Fig. 1). Phospholipases are mainly classified into three large groups: secretory PLA2 (sPLA2), cytosolic PLA2 (cPLA2), and calcium-independent PLA2 (iPLA2). It is now known that this family is comprised of at least 10 members with distinct cellular distributions and growing therapeutic potential.37 Specifically, sPLA2-V and sPLA2-X are selectively expressed in the epithelium of the human airway. sPLA2-IIA is low but becomes highly expressed during inflammation and sepsis as a result of LPS, cytokine and NF-κB induction. This enzyme is now associated with allergic rhinitis, rheumatoid arthritis, and septic shock. Finally, the selective expression of sPLA2-V and sPLA2-X suggests that these enzymes should be evaluated as targets for airway dysfunction. Thus, the PLA2 family represents a therapeutic target with ever-increasing potential. It is likely that PLA2 is an important intra- and extracellular mediator of inflammation. The modulation of sPLA2 and/or cPLA2 activity is important in controlling the inflammatory process.
Quercetin was found to be an effective inhibitor of PLA2 in human38 and rabbit39 leukocytes. It was also demonstrated that quercetin selectively inhibited sPLA2-II, compared to its lower inhibition of sPLA2-IB.40 Quercetagetin, kaempferol-3-O-galactoside, and scutellarein inhibited human recombinant synovial PLA2 with IC50 values ranging from 12.2 to 17.6 µM.41 Morelloflavone inhibited sPLA2 with its selectivity for Group II and III enzymes in vitro, including a high potency for the human recombinant synovial PLA2.42 Curcumin affects AA metabolism by blocking the phosphorylation of cPLA2, resulting in decreased COX-2 expression.24 Since PLA2 is coupled with COXs and LOXs (depending on the cells), the inhibitors of PLA2 may also cause the inhibition of COX or LOX activity. PLA2 inhibition of some polyphenols may contribute to their anti-inflammatory property in vivo as well.43
Many inflammatory cytokines are produced and regulated at the transcriptional level, which can either enhance or inhibit the inflammation process. One such molecular link between proinflammatory cytokines and transcription factor is peroxisome proliferator-activated receptors (PPARs). PPARs are nuclear hormone receptors that are activated by specific endogenous and exogenous ligands.44 Three isoforms (α, β/δ, and γ) have been identified and are encoded by separate genes. Among those, PPARα activation is responsible for the pleiotropic effects of peroxisome proliferators, such as enzyme induction, peroxisome proliferation, liver enlargement and anti-inflammation.45 PPARα also plays a critical role in the regulation of cellular uptake and β-oxidation of fatty acids.46 In contrast, PPARδ (also known as PPARβ) is widely expressed with relatively higher levels in the brain, colon, and skin. Although there have been extensive studies on PPARα and inflammation, very little is known about the effect of PPARδ on inflammation. In contrast, PPARγ has been further divided into three subtypes: PPARγ1, γ2, and γ3.47,48 All PPAR isotypes form a heterodimeric complex with the retinoid X receptor (RXR), and the complex binds to the PPAR response element (PPRE), which functions as the central regulator of cellular differentiation,49 apoptosis,50,51 inflammatory responses,52,53 lipid metabolism and metabolic disease.54 The first study that provided evidence of a potential role for PPARs in the inflammatory response showed that the duration of the inflammatory response is prolonged in PPARα-KO mice in response to LTB4.55 Since then, a growing body of evidence has suggested that PPAR ligands inhibit pro-inflammatory cytokines in monocytes.52 Thus, the activation of PPAR results in anti-inflammatory activity.
Genistein increased the expression of genes involved in lipid catabolism; this effect is not estrogen receptor-dependent, but instead is PPARα-dependent.56 EGCG also binds to PPARα.57 Amentoflavone up-regulates PPARγ expression in A549 human lung epithelial cells.58 Few studies have regarded polyphenols as PPAR ligands, but it is probable that polyphenols may also affect PPAR protein expression, which results in the activation of the PPAR pathway. It is notable that the PPAR pathways are tightly connected to other inflammatory pathways including NF-κB, COX-2 expression, and pro-inflammatory cytokines. These results demonstrate that the activation of PPARs by certain polyphenols represent another possibility the treatment of chronic inflammatory diseases.
Nitric oxide (NO), a gaseous free radical, is released by a family of enzymes, including endothelia NOS (eNOS), neuronal NOS (nNOS) and inducible NOS (iNOS), with the formation of stoichiometric amounts of L-citrulline from L-arginine.59 Excessive and prolonged iNOS-mediated NO generation has attracted attention because of its relevance to inflammation. Since NO is one of the inflammatory mediators, the inhibition of NO by the extracts of edible plants has been reported.60 Using LPS/cytokine-treated macrophages or macrophage-like cell lines, quercetin was found to inhibit the production of NO.61 However, mechanism studies have shown that compounds down-regulate iNOS expression, which subsequently results in reducing the amount of NO production. Several flavonoid derivatives, including apigenin, quercetin, and morin, have also inhibited NO production from LPS/interferonγ-activated C6 astrocytes.62 Quercetin markedly inhibits the production of TNF-α and NO by LPS-activated macrophages, and TNF-α inhibition may occur post-transcriptionally, whereas iNOS inhibition usually occurs at the transcriptional level.63 Many polyphenols, including 6-gingerol,64 EGCG,65 resveratrol,66 indole-3-carbinol,67 and oroxylin A,68 inhibit NOS expression in LPS-induced RAW264.7 cells. These effects are probably mediated by NF-κB inhibition.69
Nuclear transcription factor κB (NF-κB), which was discovered by David Baltimore in 1986, is a ubiquitous factor that resides in the cytoplasm. When it becomes activated, it is translocated to the nucleus, where it induces gene transcription. NF-κB is activated by free radicals, inflammatory stimuli, carcinogens, tumor promoters, endotoxins, γ-radiation, ultraviolet (UV) light, and X-rays. Upon activation, NF-κB induces the expression of more than 200 genes that have been shown to suppress apoptosis and induce cellular transformation, proliferation, invasion, metastasis, chemoresistance, radio-resistance, and inflammation. The activated form of NF-κB has been found to mediate cancer, atherosclerosis, myocardial infarction, diabetes, allergy, asthma, arthritis, and other inflammatory diseases. It is not surprising that NF-κB has been linked to this wide variety of diseases, because most diseases are caused by poorly regulated inflammation. Therefore, agents that can suppress NF-κB activation, in principle, have the potential to prevent, delay the onset of, or treat inflammatory diseases.
Several sesquiterpene lactones demonstrated anti-inflammatory properties by inhibiting the binding of the transcription factor NF-κB, as assessed by a gel shift assay.70 Quercetin was reported to suppress TNF-α induced expression of IL-8 and monocyte chemoattractant protein (MCP-1) due to its ability to inhibit the activation of NF-κB.71 Quercetin also inhibited the NF-κB pathway without any modification of c-Jun N-terminal kinase activity both in vivo and in vitro.72 Caffeic acid phenethyl ester (CAFE), a natural product secreted by the honeybee, inhibited cerebral inflammatory responses by reducing the activity of NF-κB.73 Resveratrol has also been known to inhibit NF-κB activity, although resveratrol may possess anti-inflammatory properties via an NF-κB-independent mechanism.74 Recently, it has been reported that indole-3-carbinol, found in Brassica sp. vegetables (such as cabbage, cauliflower, and brussel spouts), inhibits NF-κB and NF-κB-regulated gene expression.75 The inhibition of NF-κB by several polyphenols may explain the antiinflammatory effects of these compounds.
NAG-1 (NSAID activated gene-1) was identified by PCR-based subtractive hybridization from an NSAID-induced library in cyclooxygenase-negative cells as a divergent member of the TGF-β superfamily.76 The induction of NAG-1 by NSAIDs is an important COX-independent mechanism by which some of these proven anti-inflammatory compounds mediate their effects.77 NAG-1 has been identified by other groups, as macrophage inhibitory cytokine-1 (MIC-1),78 placental transformation growth factor-β (PTGFB),79 prostate derived factor (PDF),80 growth differentiation factor 15 (GDF-15),81 and placental bone morphogenetic protein (PLAB).82 The diversity of biological functions represented by this nomenclature suggests that the NAG-1 protein has broad activity in inflammation, cancer, and differentiation. The molecular mechanisms responsible for these functions have yet to be determined in detail. Experimental evidence suggests that NAG-1 may share at least some of the common functions of TGF-β superfamily cytokines. For instance, TGF-β1 induces apoptosis and cell growth arrest in epithelial cells, and mice treated with TGF-β1 died of widespread inflammation. Indeed, NAG-1 expression reduces TNF-α secretion in macrophages,78 and inhibits the proliferation of primitive hemopoietic progenitors83 and several epithelial cancer cell lines,76,77,79,84,85 reflecting the activity of a multifunctional cytokine. In contrast to other members of the TGF-β superfamily, NAG-1 is induced not only by several NSAIDs, but also by several dietary compounds. As described in Table 1, NAG-1 was initiated by many of the polyphenols described in this review. All of the listed compounds induced NAG-1 expression, except EC and EGC, which indicates the structure-specific expression of NAG-1 induction. Overall, NAG-1 could prove to be a good molecular target protein for examining the anti-inflammatory and/or anti-cancer activity of other compounds.
Dietary polyphenols comprise a vast array of biologically active compounds that are ubiquitous in plants, many of which have been used in traditional Oriental medicine for thousands of years. In this review, we summarized the different targets of dietary polyphenols by their anti-inflammatory effects. Given that certain polyphenols are known to affect more than one protein, it is necessary consider possibility that any single compound may affect multiple mechanisms. However, most of the previous clinical studies have used foods or beverages containing a mixture of different polyphenols, and the exact mechanisms of the most active polyphenol components remain largely unknown. Therefore, these factors should be taken into consideration along with bioavailability in order to improve the experimental design and interpret the observed effects. In addition, since absorption, metabolism, and elimination vary widely among polyphenols, these factors should also be considered Furthermore, such characterization of these compounds could validate the importance of dietary polyphenol compounds in human health and encourage their use as templates for further structural development of more effective and safe chemopreventive compounds.
Figures and Tables
Fig. 1
Arachidonic acid (AA) pathways. AA can be converted to either prostaglandins or leukotrienes, depending on the enzymes that are present. Each metabolite affects its biological action through receptors.

References
1. Flower RJ, Vane JR. Inhibition of prostaglandin synthetase in brain explains the anti-pyretic activity of paracetamol (4-acetamidophenol). Nature. 1972. 240:410–411.
2. Cragg GM, Newman DJ, Weiss RB. Coral reefs, forests, and thermal vents: the worldwide exploration of nature for novel antitumor agents. Semin Oncol. 1997. 24:156–163.
3. Kuhnau J. The flavonoids. A class of semi-essential food components: their role in human nutrition. World Rev Nutr Diet. 1976. 24:117–191.
4. Sampson L, Rimm E, Hollman PC, de Vries JH, Katan MB. Flavonol and flavone intakes in US health professionals. J Am Diet Assoc. 2002. 102:1414–1420.
5. Scalbert A, Williamson G. Dietary intake and bioavailability of polyphenols. J Nutr. 2000. 130:8S Suppl. 2073S–2085S.
6. Baek SJ, Kim JS, Jackson FR, Eling TE, McEntee MF, Lee SH. Epicatechin gallate-induced expression of NAG-1 is associated with growth inhibition and apoptosis in colon cancer cells. Carcinogenesis. 2004. 25:2425–2432.
7. Baek SJ, Wilson LC, Eling TE. Resveratrol enhances the expression of non-steroidal anti-inflammatory drug-activated gene (NAG-1) by increasing the expression of p53. Carcinogenesis. 2002. 23:425–434.
8. Lee SH, Kim JS, Yamaguchi K, Eling TE, Baek SJ. Indole-3-carbinol and 3,3'-diindolylmethane induce expression of NAG-1 in a p53-independent manner. Biochem Biophys Res Commun. 2005. 328:63–69.
9. Wilson LC, Baek SJ, Call A, Eling TE. Nonsteroidal anti-inflammatory drug-activated gene (NAG-1) is induced by genistein through the expression of p53 in colorectal cancer cells. Int J Cancer. 2003. 105:747–753.
10. Chandrasekharan NV, Dai H, Roos KL, Evanson NK, Tomsik J, Elton TS, et al. COX-3, a cyclooxygenase-1 variant inhibited by acetaminophen and other analgesic/antipyretic drugs: cloning, structure, and expression. Proc Natl Acad Sci USA. 2002. 99:13926–13931.
11. Herschman HR. Prostaglandin synthase 2. Biochim Biophys Acta. 1996. 1299:125–140.
12. Silverstein FE, Faich G, Goldstein JL, Simon LS, Pincus T, Whelton A, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: The CLASS study: A randomized controlled trial. Celecoxib Long-term Arthritis Safety Study. JAMA. 2000. 284:1247–1255.
13. Brater DC, Harris C, Redfern JS, Gertz BJ. Renal effects of COX-2-selective inhibitors. Am J Nephrol. 2001. 21:1–15.
14. Fitzgerald GA. Coxibs and cardiovascular disease. N Engl J Med. 2004. 351:1709–1711.
15. Baumann J, von Bruchhausen F, Wurm G. Flavonoids and related compounds as inhibition of arachidonic acid peroxidation. Prostaglandins. 1980. 20:627–639.
16. Landolfi R, Mower RL, Steiner M. Modification of platelet function and arachidonic acid metabolism by bioflavonoids. Structure-activity relations. Biochem Pharmacol. 1984. 33:1525–1530.
17. Williams CA, Harborne JB, Geiger H, Hoult JR. The flavonoids of Tanacetum parthenium and T. vulgare and their anti-inflammatory properties. Phytochemistry. 1999. 51:417–423.
18. Yasukawa K, Kaminaga T, Kitanaka S, Tai T, Nunoura Y, Natori S, et al. 3 beta-p-hydroxybenzoyldehydrotumulosic acid from Poria cocos, and its anti-inflammatory effect. Phytochemistry. 1998. 48:1357–1360.
19. Kundu JK, Na HK, Chun KS, Kim YK, Lee SJ, Lee SS, et al. Inhibition of phorbol ester-induced COX-2 expression by epigallocatechin gallate in mouse skin and cultured human mammary epithelial cells. J Nutr. 2003. 133:3805S–3810S.
20. Gerhauser C, Klimo K, Heiss E, Neumann I, Gamal-Eldeen A, Knauft J, et al. Mechanism-based in vitro screening of potential cancer chemopreventive agents. Mutat Res. 2003. 523-524:163–172.
21. Wheeler DS, Catravas JD, Odoms K, Denenberg A, Malhotra V, Wong HR. Epigallocatechin-3-gallate, a green tea-derived polyphenol, inhibits IL-1 beta-dependent proinflammatory signal transduction in cultured respiratory epithelial cells. J Nutr. 2004. 134:1039–1044.
22. Jang M, Pezzuto JM. Cancer chemopreventive activity of resveratrol. Drugs Exp Clin Res. 1999. 25:65–77.
23. Subbaramaiah K, Chung WJ, Michaluart P, Telang N, Tanabe T, Inoue H, et al. Resveratrol inhibits cyclooxygenase-2 transcription and activity in phorbol estertreated human mammary epithelial cells. J Biol Chem. 1998. 273:21875–21882.
24. Hong J, Bose M, Ju J, Ryu JH, Chen X, Sang S, et al. Modulation of arachidonic acid metabolism by curcumin and related beta-diketone derivatives: effects on cytosolic phospholipase A(2), cyclooxygenases and 5-lipoxygenase. Carcinogenesis. 2004. 25:1671–1679.
25. Mutoh M, Takahashi M, Fukuda K, Matsushima-Hibiya Y, Mutoh H, Sugimura T, et al. Suppression of cyclooxygenase-2 promoter-dependent transcriptional activity in colon cancer cells by chemopreventive agents with a resorcin-type structure. Carcinogenesis. 2000. 21:959–963.
26. Chi YS, Kim HP. Suppression of cyclooxygenase-2 expression of skin fibroblasts by wogonin, a plant flavone from Scutellaria radix. Prostaglandins Leukot Essent Fatty Acids. 2005. 72:59–66.
27. Kim DW, Chi YS, Son KH, Chang HW, Kim JS, Kang SS, et al. Effects of sophoraflavanone G, a prenylated flavonoid from Sophora flavescens, on cyclooxygenase-2 and in vivo inflammatory response. Arch Pharm Res. 2002. 25:329–335.
28. Laughton MJ, Evans PJ, Moroney MA, Hoult JR, Halliwell B. Inhibition of mammalian 5-lipoxygenase and cyclo-oxygenase by flavonoids and phenolic dietary additives. Relationship to antioxidant activity and to iron ion-reducing ability. Biochem Pharmacol. 1991. 42:1673–1681.
29. Hartisch C, Kolodziej H, von Bruchhausen F. Dual inhibitory activities of tannins from Hamamelis virginiana and related polyphenols on 5-lipoxygenase and lyso-PAF: acetyl-CoA acetyltransferase. Planta Med. 1997. 63:106–110.
30. Reddy GR, Ueda N, Hada T, Sackeyfio AC, Yamamoto S, Hano Y, et al. A prenylflavone, artonin E, as arachidonate 5-lipoxygenase inhibitor. Biochem Pharmacol. 1991. 41:115–118.
31. Zschocke S, Drewes SE, Paulus K, Bauer R, van Staden J. Analytical and pharmacological investigation of Ocotea bullata (black stinkwood) bark and leaves. J Ethnopharmacol. 2000. 71:219–230.
32. Sekiya K, Okuda H. Selective inhibition of platelet lipoxygenase by baicalein. Biochem Biophys Res Commun. 1982. 105:1090–1095.
33. Chi YS, Jong HG, Son KH, Chang HW, Kang SS, Kim HP. Effects of naturally occurring prenylated flavonoids on enzymes metabolizing arachidonic acid: cyclooxygenases and lipoxygenases. Biochem Pharmacol. 2001. 62:1185–1191.
34. Fiorucci S, Meli R, Bucci M, Cirino G. Dual inhibitors of cyclooxygenase and 5-lipoxygenase. A new avenue in anti-inflammatory therapy? Biochem Pharmacol. 2001. 62:1433–1438.
35. Moroney MA, Alcaraz MJ, Forder RA, Carey F, Hoult JR. Selectivity of neutrophil 5-lipoxygenase and cyclooxygenase inhibition by an anti-inflammatory flavonoid glycoside and related aglycone flavonoids. J Pharm Pharmacol. 1988. 40:787–792.
36. Pruzanski W, Vadas P. Phospholipase A2--a mediator between proximal and distal effectors of inflammation. Immunol Today. 1991. 12:143–146.
37. Six DA, Dennis EA. The expanding superfamily of phospholipase A2 enzymes: classification and characterization. Biochim Biophys Acta. 2000. 1488:1–19.
38. Lee TP, Matteliano ML, Middleton E Jr. Effect of quercetin on human polymorphonuclear leukocyte lysosomal enzyme release and phospholipid metabolism. Life Sci. 1982. 31:2765–2774.
39. Lanni C, Becker EL. Inhibition of neutrophil phospholipase A2 by p-bromophenylacyl bromide, nordihydroguaiaretic acid, 5,8,11,14-eicosatetraynoic acid and quercetin. Int Arch Allergy Appl Immunol. 1985. 76:214–217.
40. Lindahl M, Tagesson C. Selective inhibition of group II phospholipase A2 by quercetin. Inflammation. 1993. 17:573–582.
41. Gil B, Sanz MJ, Terencio MC, Ferrandiz ML, Bustos G, Paya M, et al. Effects of flavonoids on Naja naja and human recombinant synovial phospholipases A2 and inflammatory responses in mice. Life Sci. 1994. 54:PL333–PL338.
42. Gil B, Sanz MJ, Terencio MC, Gunasegaran R, Paya M, Alcaraz MJ. Morelloflavone, a novel biflavonoid inhibitor of human secretory phospholipase A2 with anti-inflammatory activity. Biochem Pharmacol. 1997. 53:733–740.
43. Sapirstein A, Bonventre JV. Specific physiological roles of cytosolic phospholipase A2 as defined by gene knockouts. Biochim Biophys Acta. 2000. 1488:139–148.
44. Schoonjans K, Martin G, Staels B, Auwerx J. Peroxisome proliferator-activated receptors, orphans with ligands and functions. Curr Opin Lipidol. 1997. 8:159–166.
45. Klaunig JE, Babich MA, Baetcke KP, Cook JC, Corton JC, David RM, et al. PPARalpha agonist-induced rodent tumors: modes of action and human relevance. Crit Rev Toxicol. 2003. 33:655–780.
46. Marx N, Duez H, Fruchart JC, Staels B. Peroxisome proliferator-activated receptors and atherogenesis: regulators of gene expression in vascular cells. Circ Res. 2004. 94:1168–1178.
47. Fajas L, Auboeuf D, Raspe E, Schoonjans K, Lefebvre AM, Saladin R, et al. The organization, promoter analysis, and expression of the human PPARgamma gene. J Biol Chem. 1997. 272:18779–18789.
48. Fajas L, Fruchart JC, Auwerx J. PPARgamma3 mRNA: a distinct PPARgamma mRNA subtype transcribed from an independent promoter. FEBS Lett. 1998. 438:55–60.
49. Tontonoz P, Singer S, Forman BM, Sarraf P, Fletcher JA, Fletcher CD, et al. Terminal differentiation of human liposarcoma cells induced by ligands for peroxisome proliferator-activated receptor gamma and the retinoid X receptor. Proc Natl Acad Sci USA. 1997. 94:237–241.
50. Heaney AP, Fernando M, Yong WH, Melmed S. Functional PPAR-gamma receptor is a novel therapeutic target for ACTH-secreting pituitary adenomas. Nat Med. 2002. 8:1281–1287.
51. Yoshizawa K, Cioca DP, Kawa S, Tanaka E, Kiyosawa K. Peroxisome proliferator-activated receptor gamma ligand troglitazone induces cell cycle arrest and apoptosis of hepatocellular carcinoma cell lines. Cancer. 2002. 95:2243–2251.
52. Jiang C, Ting AT, Seed B. PPAR-gamma agonists inhibit production of monocyte inflammatory cytokines. Nature. 1998. 391:82–86.
53. Ricote M, Li AC, Willson TM, Kelly CJ, Glass CK. The peroxisome proliferator-activated receptor-gamma is a negative regulator of macrophage activation. Nature. 1998. 391:79–82.
54. Nolan JJ, Ludvik B, Beerdsen P, Joyce M, Olefsky J. Improvement in glucose tolerance and insulin resistance in obese subjects treated with troglitazone. N Engl J Med. 1994. 331:1188–1193.
55. Devchand PR, Keller H, Peters JM, Vazquez M, Gonzalez FJ, Wahli W. The PPARalpha-leukotriene B4 pathway to inflammation control. Nature. 1996. 384:39–43.
56. Kim S, Shin HJ, Kim SY, Kim JH, Lee YS, Kim DH, et al. Genistein enhances expression of genes involved in fatty acid catabolism through activation of PPARalpha. Mol Cell Endocrinol. 2004. 220:51–58.
57. Kaul D, Sikand K, Shukla AR. Effect of green tea polyphenols on the genes with atherosclerotic potential. Phytother Res. 2004. 18:177–179.
58. Banerjee T, Valacchi G, Ziboh VA, van der Vliet A. Inhibition of TNFalpha-induced cyclooxygenase-2 expression by amentoflavone through suppression of NF-kappaB activation in A549 cells. Mol Cell Biochem. 2002. 238:105–110.
59. Nathan C, Xie QW. Nitric oxide synthases: roles, tolls, and controls. Cell. 1994. 78:915–918.
60. Kim OK, Murakami A, Nakamura Y, Ohigashi H. Screening of edible Japanese plants for nitric oxide generation inhibitory activities in RAW 264.7 cells. Cancer Lett. 1998. 125:199–207.
61. Chen YC, Shen SC, Lee WR, Hou WC, Yang LL, Lee TJ. Inhibition of nitric oxide synthase inhibitors and lipopolysaccharide induced inducible NOS and cyclooxygenase-2 gene expressions by rutin, quercetin, and quercetin pentaacetate in RAW 264.7 macrophages. J Cell Biochem. 2001. 82:537–548.
62. Soliman KF, Mazzio EA. In vitro attenuation of nitric oxide production in C6 astrocyte cell culture by various dietary compounds. Proc Soc Exp Biol Med. 1998. 218:390–397.
63. Wadsworth TL, Koop DR. Effects of the wine polyphenolics quercetin and resveratrol on pro-inflammatory cytokine expression in RAW 264.7 macrophages. Biochem Pharmacol. 1999. 57:941–949.
64. Ippoushi K, Azuma K, Ito H, Horie H, Higashio H. [6]-Gingerol inhibits nitric oxide synthesis in activated J774.1 mouse macrophages and prevents peroxynitriteinduced oxidation and nitration reactions. Life Sci. 2003. 73:3427–3437.
65. Lin YL, Lin JK. (-)-Epigallocatechin-3-gallate blocks the induction of nitric oxide synthase by down-regulating lipopolysaccharide-induced activity of transcription factor nuclear factor-kappaB. Mol Pharmacol. 1997. 52:465–472.
66. Tsai SH, Lin-Shiau SY, Lin JK. Suppression of nitric oxide synthase and the down-regulation of the activation of NFkappaB in macrophages by resveratrol. Br J Pharmacol. 1999. 126:673–680.
67. Chen YH, Dai HJ, Chang HP. Suppression of inducible nitric oxide production by indole and isothiocyanate derivatives from Brassica plants in stimulated macrophages. Planta Med. 2003. 69:696–700.
68. Chen Y, Yang L, Lee TJ. Oroxylin A inhibition of lipopolysaccharide-induced iNOS and COX-2 gene expression via suppression of nuclear factor-kappaB activation. Biochem Pharmacol. 2000. 59:1445–1457.
69. Surh YJ. Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer. 2003. 3:768–780.
70. Castro V, Murillo R, Klaas CA, Meunier C, Mora G, Pahl HL, et al. Inhibition of the transcription factor NF-kappa B by sesquiterpene lactones from Podachaenium eminens. Planta Med. 2000. 66:591–595.
71. Sato M, Miyazaki T, Kambe F, Maeda K, Seo H. Quercetin, a bioflavonoid, inhibits the induction of interleukin 8 and monocyte chemoattractant protein-1 expression by tumor necrosis factor-alpha in cultured human synovial cells. J Rheumatol. 1997. 24:1680–1684.
72. Comalada M, Camuesco D, Sierra S, Ballester I, Xaus J, Galvez J, et al. In vivo quercitrin anti-inflammatory effect involves release of quercetin, which inhibits inflammation through down-regulation of the NF-kappaB pathway. Eur J Immunol. 2005. 35:584–592.
73. Montpied P, de Bock F, Rondouin G, Niel G, Briant L, Courseau AS, et al. Caffeic acid phenethyl ester (CAPE) prevents inflammatory stress in organotypic hippocampal slice cultures. Brain Res Mol Brain Res. 2003. 115:111–120.
74. Birrell MA, McCluskie K, Wong S, Donnelly LE, Barnes PJ, Belvisi MG. Resveratrol, an extract of red wine, inhibits lipopolysaccharide induced airway neutrophilia and inflammatory mediators through an NF-kappa;B-independent mechanism. FASEB J. 2005. 19:840–841.
75. Takada Y, Andreeff M, Aggarwal BB. Indole-3-carbinol suppresses NF-{kappa}B and I{kappa}B{alpha} kinase activation causing inhibition of expression of NF-{kappa}B-regulated antiapoptotic and metastatic gene products and enhancement of apoptosis in myeloid and leukemia cells. Blood. 2005. 106:641–649.
76. Baek SJ, Kim KS, Nixon JB, Wilson LC, Eling TE. Cyclooxygenase inhibitors regulate the expression of a TGF-beta superfamily member that has proapoptotic and antitumorigenic activities. Mol Pharmacol. 2001. 59:901–908.
77. Baek SJ, Wilson LC, Lee CH, Eling TE. Dual function of nonsteroidal anti-inflammatory drugs (NSAIDs): inhibition of cyclooxygenase and induction of NSAID-activated gene. J Pharmacol Exp Ther. 2002. 301:1126–1131.
78. Bootcov MR, Bauskin AR, Valenzuela SM, Moore AG, Bansal M, He XY, et al. MIC-1, a novel macrophage inhibitory cytokine, is a divergent member of the TGF-beta superfamily. Proc Natl Acad Sci USA. 1997. 94:11514–11519.
79. Li PX, Wong J, Ayed A, Ngo D, Brade AM, Arrowsmith C, et al. Placental transforming growth factor-beta is a downstream mediator of the growth arrest and apoptotic response of tumor cells to DNA damage and p53 overexpression. J Biol Chem. 2000. 275:20127–20135.
80. Paralkar VM, Vail AL, Grasser WA, Brown TA, Xu H, Vukicevic S, et al. Cloning and characterization of a novel member of the transforming growth factor-beta/bone morphogenetic protein family. J Biol Chem. 1998. 273:13760–13767.
81. Bottner M, Laaff M, Schechinger B, Rappold G, Unsicker K, Suter-Crazzolara C. Characterization of the rat, mouse, and human genes of growth/differentiation factor-15/macrophage inhibiting cytokine-1 (GDF-15/MIC-1). Gene. 1999. 237:105–111.
82. Hromas R, Hufford M, Sutton J, Xu D, Li Y, Lu L. PLAB, a novel placental bone morphogenetic protein. Biochim Biophys Acta. 1997. 1354:40–44.
83. Detmer K, Steele TA, Shoop MA, Dannawi H. Lineage-restricted expression of bone morphogenetic protein genes in human hematopoietic cell lines. Blood Cells Mol Dis. 1999. 25:310–323.
84. Baek SJ, Kim JS, Moore SM, Lee SH, Martinez J, Eling TE. Cyclooxygenase inhibitors induce the expression of the tumor suppressor gene EGR-1, which results in the up-regulation of NAG-1, an antitumorigenic protein. Mol Pharmacol. 2005. 67:356–364.
85. Tan M, Wang Y, Guan K, Sun Y. PTGF-beta, a type beta transforming growth factor (TGF-beta) superfamily member, is a p53 target gene that inhibits tumor cell growth via TGF-beta signaling pathway. Proc Natl Acad Sci USA. 2000. 97:109–114.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download