Abstract
Tamsulosin is an effective therapeutic option for lower urinary tract symptoms, as it selectively blocks α1A- and α1D-adrenoceptors in the bladder and prostate. The purpose of this study was to evaluate the bioequivalence in the pharmacokinetics (PK) of two 0.2 mg tamsulosin formulations when administered as the reference formulation (Yuropa® sustained-release tablet) vs. the test formulation (Yutanal® capsule) in healthy male subjects. A randomized, open-label, single-dose, two-way, two-period, crossover study was conducted in 37 healthy volunteers. The 0.2 mg of tamsulosin as the test or the reference formulation were administered during each period, and serial blood samples were collected up to 36 hours after dosing for PK analyses. A non-compartmental analysis was used to estimate the PK parameters. Geometric mean ratios (GMR) and 90% confidence intervals (CIs) were calculated for the two formulations to compare the maximum concentration (Cmax) and the area under the concentration-time curve from time zero to the time of the last quantifiable concentration (AUClast). The mean Cmax and AUClast for the test formulation were 6.19 μg/L and 71.30 μg.h/L, respectively, and 5.76 μg/L and 70.38 μg.h /L for the reference formulation, respectively. The GMRs (90% CIs) of the Cmax and AUClast between the two formulations were 1.09 (1.01–1.17) and 1.03 (0.96–1.10), respectively. Tamsulosin 0.2 mg as the test formulation exhibited bioequiva-lent PK profiles to those of the reference formulation. Therefore, the test formulation is expected to be an alternative to the reference formulation without concerns about differences in drug exposure.
References
1. Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, et al. The standardisation of terminology in lower urinary tract function: report from the standardisation sub-committee of the International Continence Society. Urology. 2003; 61:37–49. doi: 10.1016/S0090-4295 (02)02243-4.

2. Lyseng-Williamson KA, Jarvis B, Wagstaff AJ. Tamsulosin: an update of its role in the management of lower urinary tract symptoms. Drugs. 2002; 62:135–167. doi: 10.2165/00003495-200262010-00006.
3. Irwin DE, Kopp ZS, Agatep B, Milsom I, Abrams P. Worldwide prevalence estimates of lower urinary tract symptoms, overactive bladder, urinary incontinence and bladder outlet obstruction. BJU Int. 2011; 108:1132–1138. doi: 10.1111/j.1464-410X.2010.09993.x.

4. Irwin DE, Milsom I, Kopp Z, Abrams P, Artibani W, Herschorn S. Prevalence, Severity, and symptom bother of lower urinary tract symptoms among men in the EPIC study: impact of overactive bladder. Eur Urol. 2009; 56:14–20. doi: 10.1016/j.eururo.2009.02.026.

5. Kannan H, Radican L, Turpin RS, Bolge SC. Burden of illness associated with lower urinary tract symptoms including overactive bladder/urinary incontinence. Urology. 2009; 74:34–38.

6. Andersson K-E. Mode of action of α-adrenoceptor antagonists in the treatment of lower urinary 225 tract symptoms. Current Prostate Reports. 2005; 3:28–33. doi: 10.1007/s11918-996-0012-1.
7. Schwinn DA, Michelotti GA. α1-adrenergic receptors in the lower urinary tract and vascular bed: potential role for the α1d subtype in filling symptoms and effects of ageing on vascular expression. BJU Int. 2000; 85(Suppl 2):6–11. doi: 10.1046/j.1464-410X.2000.00061.x.
8. http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/020579s027lbl.pdfAccessedMay25. 2015.
9. O'Leary MP. Tamsulosin: current clinical experience. Urology. 2001; 58(Suppl 1):42–48.
10. Lee K, Choi S, Jeon H, Lee B, Kim H, Lee J, et al. Controlled release of tamsulosin from enteric coated sustained-release matrices with aqueous microchannels. J Kor Pharm Sci. 2004; 34:471–475.
11. Wilde M, McTavish D. Tamsulosin. A review of its pharmacological properties and therapeutic potential in the management of symptomatic benign prostatic hyperplasia. Drugs. 1996; 52:883–898. doi: 10.2165/00003495-199652060-00012.
12. Kim MS, Kim JS, You YH, Park HJ, Lee S, Park JS, et al. Development and 236 optimization of a novel oral controlled delivery system for tamsulosin hydrochloride using response surface methodology. Int J Pharm. 2007; 341:97–104.
13. Kamimura H, Oishi S, Matsushima H, Watanabe T, Higuchi S, Hall M, et al. Identification of cytochrome P450 isozymes involved in metabolism of the α1-adrenoceptor blocker tamsulosin in human liver microsomes. Xenobiotica. 1998; 28:909–922.
14. Ding L, Li L, Tao P, Yang J, Zhang Z. Quantitation of tamsulosin in human plasma by liquid chromatography–electrospray ionization mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2002; 767:75–81.

15. Prasaja B, Harahap Y, Lusthom W, Setiawan EC, Ginting MB, Hardiyanti , et al. A bioequivalence study of two tamsulosin sustained-release tablets in Indonesian healthy volunteers. Eur J Drug Metab Pharmacokinet. 2011; 36:109–113.

16. Nithiyananthan T, Shankarananth V, Rajasekhar K, Hareesh G, Kumar PN, Reddy RSP. Formulation and evaluation of tamsulosin hydrochloride as sustained release matrix tablet. Int J Chem Tech Res. 2009; 1:1278–1290.
17. Matsushima H, Kamimura H, Soeishi Y, Watanabe T, Higuchi S, Tsunoo M. Pharmacokinetics and plasma protein binding of tamsulosin hydrochloride in rats, dogs, and humans. Drug Metab Dispos. 1998; 26:240–245.
Figure 1.
Mean ± SD plasma concentration-time profiles of tamsulosin after single doses of the test (black circles) and the reference formulations (white circles) of tamsulosin hydrochloride 0.2 mg in healthy subjects (A: linear scale B: log scale).
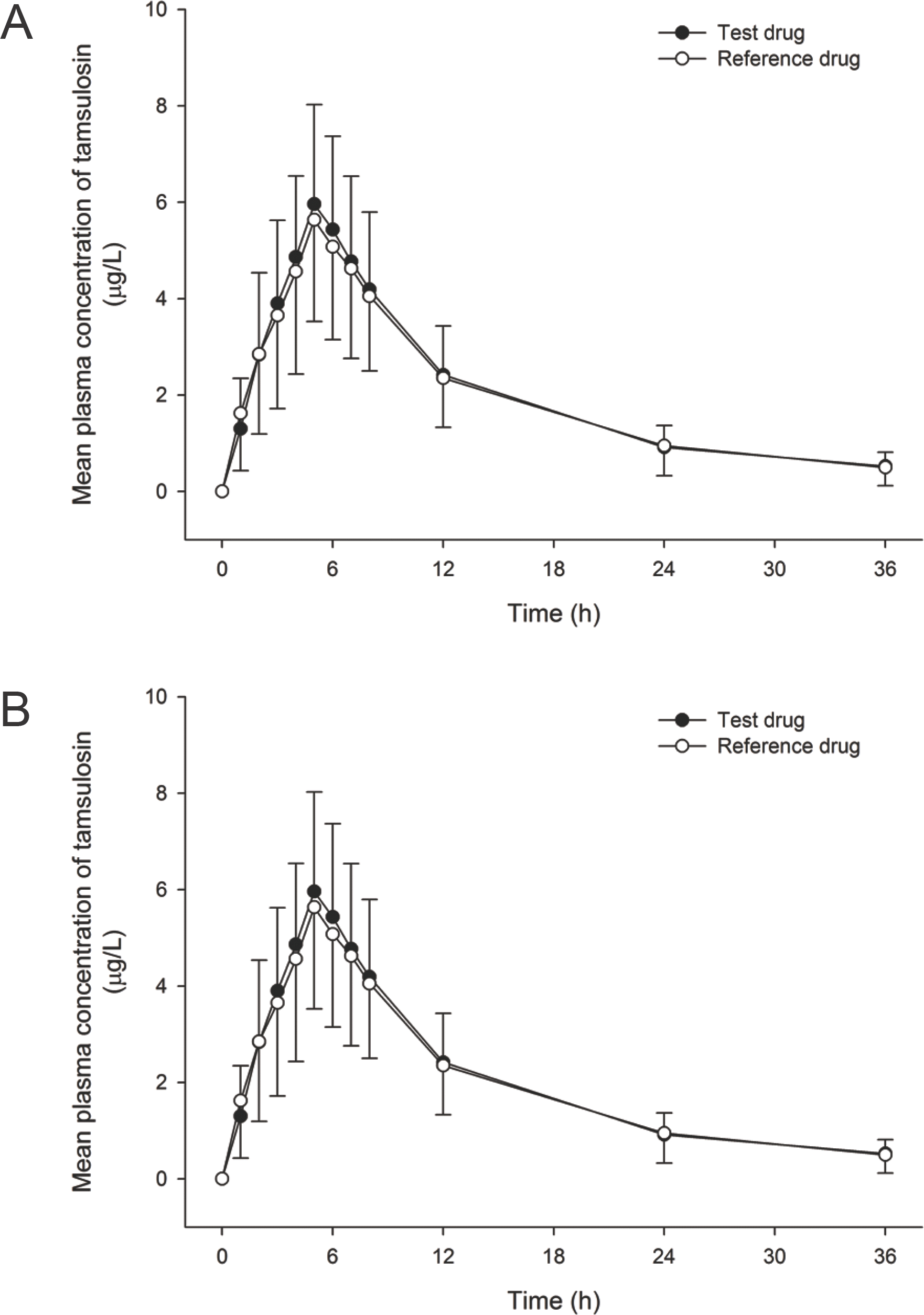
Figure 2.
Individual (A) AUClast and (B) Cmax of tamsulosin after single doses of the test and the reference formulations of tamsulosin hydrochloride 0.2 mg in healthy subjects.
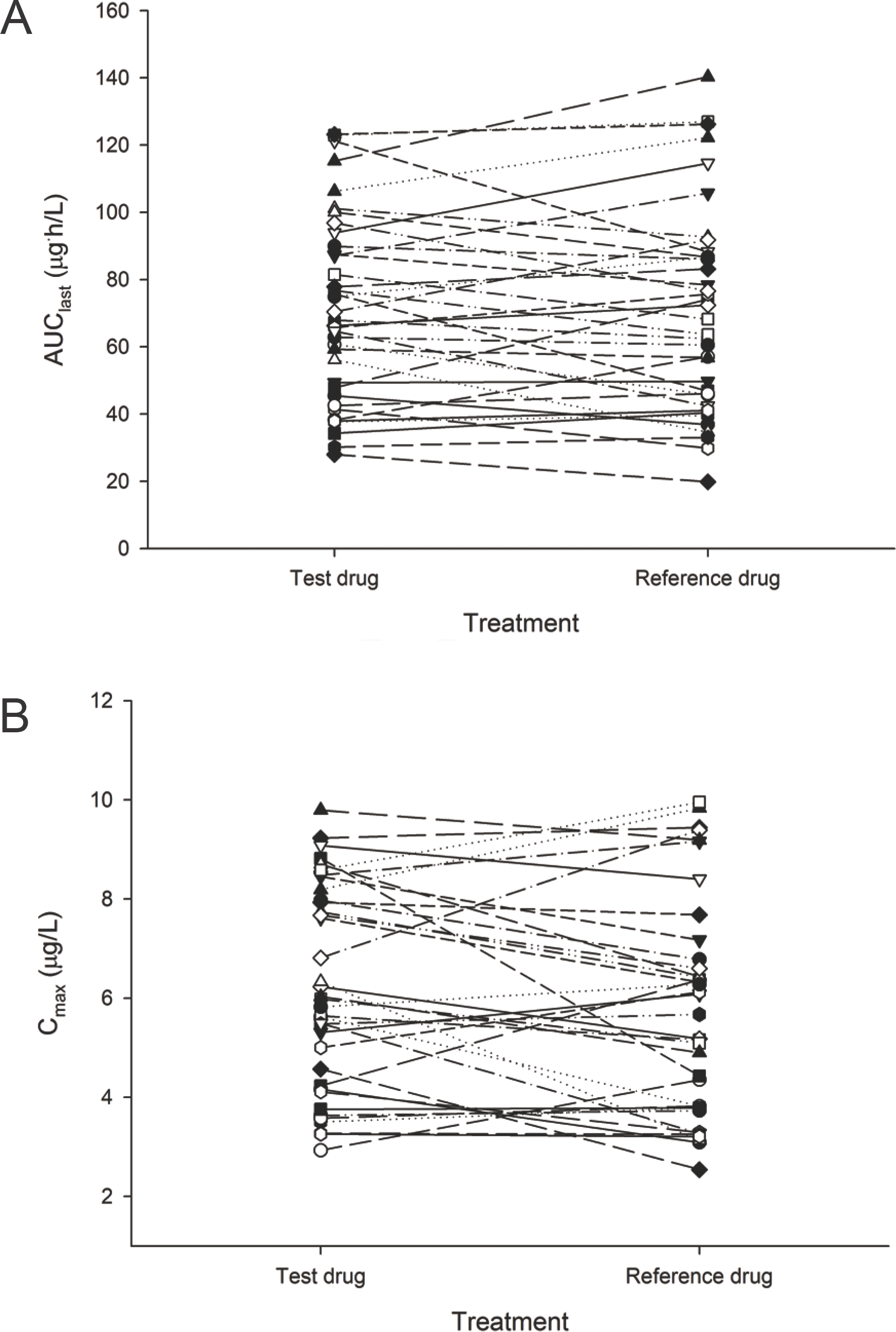
Table 1.
Pharmacokinetic parameters of tamsulosin in 37 subjects after they received a single dose of tamsulosin hydrochloride 0.2 mg of the test and the reference formulations
| Test formulation | Reference formulation | Geometric mean ratio (90% confidence interval) | |
|---|---|---|---|
| Tmax(h)a | 5.0 [2.0–7.0] | 5.0 [3.0–6.0] | |
| Cmax (μg/L) | 6.19±2.04 | 5.76±2.21 | 1.09 (1.01–1.17) |
| AUClast (μg·h/L) | 71.30±27.55 | 70.38±30.52 | 1.03 (0.96–1.10) |
| AUCinf (μg·h/L) | 78.04±30.89 | 77.66±37.58 | |
| t1/2 (h) | 8.67±2.03 | 8.85±2.98 | |
| CL/F (L/h) | 3.01±1.28 | 3.23±1.63 | |
| Vd/F (L) | 36.30±15.38 | 36.85±12.52 |
a Median [range]. Tmax, time to reach the maximum blood concentration after administration of drug; Cmax, maximum plasma concentration of drug; AUClast, area under the plasma concentration-time curve from time 0 to the time of the last quantifiable concentration; AUCinf, area under the plasma concentration-time curve from time 0 to infinity; t1/2, terminal elimination half-life; CL/F, apparent clearance; Vd/ F, apparent volume of distribution.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download