Sclerotherapy with aluminum potassium sulfate and tannic acid (ALTA) has a potent effect on internal hemorrhoids. In this retrospective study, we compared the effects of endoscopic ALTA therapy and standard ALTA therapy.
We investigated patients who underwent treatment for internal hemorrhoids at our institution between 2014 and 2016. They were divided into a standard ALTA group (
There were no intergroup differences in background factors. The mean ALTA dose was 21.9±7.2 mL and 17.8±3.4 mL in the standard and endoscopic ALTA groups, respectively (
Endoscopic ALTA sclerotherapy is equivalent to standard ALTA therapy in terms of efficacy, adverse events, and recurrence. Therefore, it is a useful non-surgical option for patients with internal hemorrhoids who prefer a less invasive treatment.
Zione® (J-Dolph Co., Osaka, Japan) is a combination drug containing aluminum potassium sulfate and tannic acid (ALTA) as the active ingredients. It was created by modifying the components of Xiaozhiling, a sclerosant that is approved for injection sclerotherapy of internal hemorrhoids in China [
Although non-surgical treatment does not improve internal hemorrhoids to the same extent as surgical treatment, it may be more effective than conservative treatment with suppositories and ointments. Non-surgical treatment is less invasive than surgical treatment and is preferred by many patients. The current non-surgical treatments for internal hemorrhoids include rubber band ligation (RBL), sclerotherapy, and infrared coagulation. These treatments are primarily performed for internal hemorrhoids of Goligher grade II or lower [
ALTA therapy was first covered by the Japanese national health insurance system in 2005. It is effective for both bleeding internal hemorrhoids and prolapsed internal hemorrhoids and thus may be a useful alternative to surgery [
In ALTA therapy, internal hemorrhoids are subjected to a 4-step injection procedure that is performed using a proctoscope [
In this study, we retrospectively compared the therapeutic effect between endoscopic ALTA therapy and standard ALTA therapy.
Patients who underwent treatment of internal hemorrhoids at our institution between 2014 and 2016 were investigated. The study population consisted of 33 and 48 patients who were treated with standard ALTA therapy and endoscopic ALTA therapy, respectively.
ALTA therapy was indicated for patients with Goligher grade II/III internal hemorrhoids presenting with prolapse and bleeding. Patients with external hemorrhoids or anal polyps were excluded, as were pregnant or lactating women and patients with kidney dysfunction. In patients taking antithrombotic agents, ALTA therapy was performed without discontinuing antithrombotic treatment [
ALTA therapy was performed using a 4-step injection procedure. In the 1st step, ALTA was injected into the upper pole of the hemorrhoid. In the 2nd and 3rd steps, ALTA was injected into the center of the hemorrhoid. In the 4th step, ALTA was injected into the lower pole of the hemorrhoid. After the injections, the injection sites were massaged (by the physician inserting his/her fingers into the anus of the patient) to diffuse the drug solution [
Standard ALTA therapy was carried out in the operation theater. After the application of local anesthesia to the periphery of the anus, with the patient in Sims’ position, standard ALTA therapy was performed using a Z-type proctoscope (Arakawa Seisakujo, Tokyo, Japan) and a special needle (ALTA-only needle).
Endoscopic ALTA sclerotherapy was conducted in the endoscopy room after bowel preparation through intestinal lavage. Sedation was induced with pethidine hydrochloride instead of perianal anesthesia, and endoscopic ALTA therapy was conducted using an endoscopic injection needle (25 G, 3 mm) by attaching a transparent hood to the end of the scope. According to the 4-step injection technique, the scope was retroflexed in the rectum, and ALTA was injected into the upper pole of the hemorrhoid (1st step). After returning the retroflexed scope to the normal position, ALTA was injected into the center of the hemorrhoid and then into the lower pole (2nd, 3rd, and 4th steps). Before endoscopic ALTA therapy, total colonoscopy was performed to detect other diseases; however, an insurance claim for endoscopy was not submitted. When a lesion required further treatment, endoscopy was promptly performed to confirm the effect of endoscopic ALTA therapy [
Analgesia and antibiotics were prescribed after ALTA therapy. Short-term admission (overnight stay) was required in standard ALTA therapy, whereas endoscopic ALTA therapy was performed as a day procedure. Follow-up was performed at 1 week, 1 month, and 1 year after ALTA therapy.
Standard or endoscopic ALTA therapy was selected on the basis of each patient’s preference and the attending physician’s evaluation after explaining the anesthetic/treatment procedures and admission period to the patients.
We compared the background data of the patients, dose of ALTA administered, complications, adverse events, patients’ satisfaction with the therapeutic effect, and frequency of recurrence between the standard ALTA therapy group and the endoscopic ALTA therapy group. The therapeutic effect was evaluated through interviews, digital anal examination, and anoscopy at 1 month after ALTA therapy. A “cure” was defined as the absence of residual symptoms with almost complete resolution of the internal hemorrhoids, whereas “improvement” meant no residual symptoms, with the internal hemorrhoids being reduced in size but still visible on anoscopy. “Failure” was defined as no improvement of symptoms, with the internal hemorrhoids still being visible (no shrinkage). Patient satisfaction was evaluated on a 3-grade scale (“very satisfied”, “satisfied”, or “dissatisfied”) at 1 month after ALTA therapy.
Informed consent was obtained from all patients. This study was conducted after approval by the ethical review board of our hospital (approval no. 488). Results are expressed as means±standard deviations, except for the follow-up period (expressed as the median). Statistical analysis was performed using the chi-square test or
There were no differences in background factors between the standard ALTA therapy group and the endoscopic ALTA therapy group, including age, sex, primary symptoms, grade of internal hemorrhoids, and frequency of antithrombotic treatment (
The mean dose of ALTA in the standard ALTA therapy group and endoscopic ALTA therapy group was 21.9±7.2 mL and 17.8±3.4 mL, respectively (
At 1 month after ALTA therapy, the percentage of patients who achieved a “cure” was higher in the standard ALTA therapy group (30.3%) than in the endoscopic ALTA therapy group (18.7%). On the other hand, the percentage of patients exhibiting “improvement” was higher in the endoscopic ALTA therapy group (77.1%) than in the standard ALTA group (66.7%). The cases of 1 patient (3.0%) in the standard ALTA therapy group and 2 patients (4.2%) in the endoscopic ALTA therapy group were classified as “failure”.
The patients’ satisfaction with the therapeutic effect at 1 month after the procedure was good in both groups (>90% of patients were “very satisfied” or “satisfied” with the result). In the standard ALTA therapy group, a greater proportion of patients were “very satisfied” with the results and a lower proportion of patients were “dissatisfied” compared with the endoscopic ALTA therapy group.
None of the patients developed intra-procedural complications such as hypotension or bradycardia. Post-procedural adverse events were observed in 4 patients (12.1%) from the standard ALTA therapy group and 6 patients (12.5%) from the endoscopic ALTA therapy group. However, all of these events improved with conservative treatment. The adverse events included mild fever, anal pain, urination disorders, and ulcers. In 1 patient, an asymptomatic ulcer was found during colonoscopy conducted 3 months after endoscopic ALTA therapy for the resection of a colorectal polyp that was detected during the ALTA procedure (
Recurrent hemorrhoids were detected in 2 patients (6.3%) from the standard ALTA therapy group and in 4 patients (8.3%) from the endoscopic ALTA therapy group. One patient (3.0%) from the standard ALTA therapy group and 3 patients (6.3%) from the endoscopic ALTA therapy group underwent surgical hemorrhoidectomy within 1 year because they were not satisfied with the therapeutic effects. One patient from the standard ALTA therapy group used suppositories for the treatment of recurrent hemorrhoids. Endoscopic ALTA therapy was repeated in 1 of the 4 patients with recurrent hemorrhoids in the standard group. Recurrence of symptoms was not observed in the other patients who were followed up. The therapeutic effect after 1 year was assessed using anoscopy in 37 patients (45.7%) and endoscopy in 18 patients (22.2%). The follow-up of the patients was discontinued at the time of termination of outpatient care on the basis of self-evaluation or because of death from another disease. The median follow-up period was 498 days in the standard ALTA therapy group and 676 days in the endoscopic ALTA therapy group (
Endoscopic ALTA therapy was performed in a 70-year-old man with prolapsed internal hemorrhoids (
ALTA sclerotherapy was first covered by the Japanese national health insurance system as a non-surgical treatment for hemorrhoids in 2005. Since then, it has been performed for many patients in Japan [
Treatment of internal hemorrhoids using an endoscope instead of a proctoscope is not a new procedure, and endoscopes have also been used for sclerotherapy, RBL, and infrared coagulation [
In the current study, it is possible that more patients underwent endoscopic ALTA therapy rather than standard ALTA therapy because the endoscopic method is a day procedure, thereby being more attractive to the patients. Although standard ALTA therapy can also be performed as a day procedure, it was carried out in the operating theater in the present study and short-term admission (overnight stay) was required. Furthermore, the endoscopic ALTA group had a percentage of female patients, possibly because the endoscopic method reduces the embarrassment associated with the treatment.
ALTA was injected at a volume of 7–10 mL per internal hemorrhoid, although the dose differed between the 2 groups. Because endoscopic ALTA therapy has not been recommended in ALTA sclerotherapy workshops, we performed it carefully and used a similar bowel preparation protocol to that used for colonoscopy in order to prevent accidents. In addition, we covered the tip of the endoscope with a transparent hood to add anoscopic functionality and confirmed that ALTA was injected into the submucosal layer while observing distension at the injection site on a monitor. We used a lower ALTA dose in this series as a precaution; however, it would be preferable to increase the ALTA dose in the future while monitoring the patients for adverse events.
Although the therapeutic effect was evaluated for a short period of 1 month after ALTA therapy, >95% of the patients in each group showed “improvement” or “cure”. Consistent with this therapeutic effect, the patients were generally satisfied with the results of ALTA sclerotherapy after 1 month. In addition to 19 patients who achieved “cure”, 21 patients who demonstrated “improvement” were “very satisfied” with the treatment. This suggests that improvement of symptoms without complete cure was sufficient for patients to become “very satisfied”. On the other hand, although the treatment failed in only 3 patients, 6 patients were “dissatisfied” with the therapeutic effect. This suggests that some patients found that their symptoms did not improve as much as expected. In those patients who were not satisfied with the outcome, ALTA therapy did not result in disappearance of external hemorrhoids or anal skin tags. Thus, those who were not satisfied with the result may have been disappointed that ALTA therapy was not effective in eliminating external hemorrhoids or skin tags, as expected. In the future, the combination of excision and ALTA therapy may be indicated for patients with internal hemorrhoids who also have external hemorrhoids, anal polyps, or anal skin tags [
Adverse events were observed in approximately 12% of the patients in both the standard and endoscopic ALTA therapy groups. The most frequent adverse event was anal pain; however, anal discomfort is frequent after sclerotherapy and it is unclear whether this should be considered anal pain. Oral analgesics relieved the pain within a few days. However, painless ulceration was noted in some patients. This may have been associated with the 1st step of injection into the submucosal layer at the upper pole of the hemorrhoid. When RBL is performed, the hemorrhoid is ligated and allowed to fall off, leading to superficial ulceration [
The recurrence rate was 8.3% in the endoscopic ALTA therapy group, which did not differ significantly from that in the standard ALTA therapy group (6.1%) or the rates reported previously. This recurrence rate may have been related to the lower ALTA dose used in the endoscopic group. In the future, endoscopic ALTA therapy with a higher dose should be performed while taking care to avoid adverse events. Further follow-up may be necessary to examine the long-term outcomes of such treatment [
This study has several limitations. It was a retrospective, non-randomized, single-center study with a small sample size. Moreover, despite the widespread use of ALTA in Japan, only a limited number of institutions perform endoscopic ALTA sclerotherapy. Therefore, the efficacy and safety of endoscopic ALTA sclerotherapy should be investigated by performing a multicenter study in the future.
For patients who wish to obtain complete cure of hemorrhoids, surgical hemorrhoidectomy is the most effective treatment. However, ALTA therapy is sufficient for those who want to achieve improvement of their symptoms. In particular, endoscopic ALTA therapy does not require perianal anesthesia and may be appropriate for patients without concomitant lesions, such as external hemorrhoids.
There were no significant differences between endoscopic ALTA therapy and standard ALTA therapy in terms of efficacy, adverse events, and recurrence. Therefore, endoscopic ALTA sclerotherapy is an effective and minimally invasive non-surgical treatment option for patients with internal hemorrhoids.
Conceptualization: Yuichi Tomiki, Jun Aoki
Data curation: YT, JA, Shunsuke Motegi, Rina Takahashi, Toshiaki Hagiwara, Masaya Kawai
Investigation: Shun Ishiyama, Kiichi Sugimoto
Resources: Yu Okazawa, Kosuke Mizukoshi, MK
Writing-original draft: YT
Writing-review&editing: YT, Shinya Munakata, Kazuhiro Sakamoto
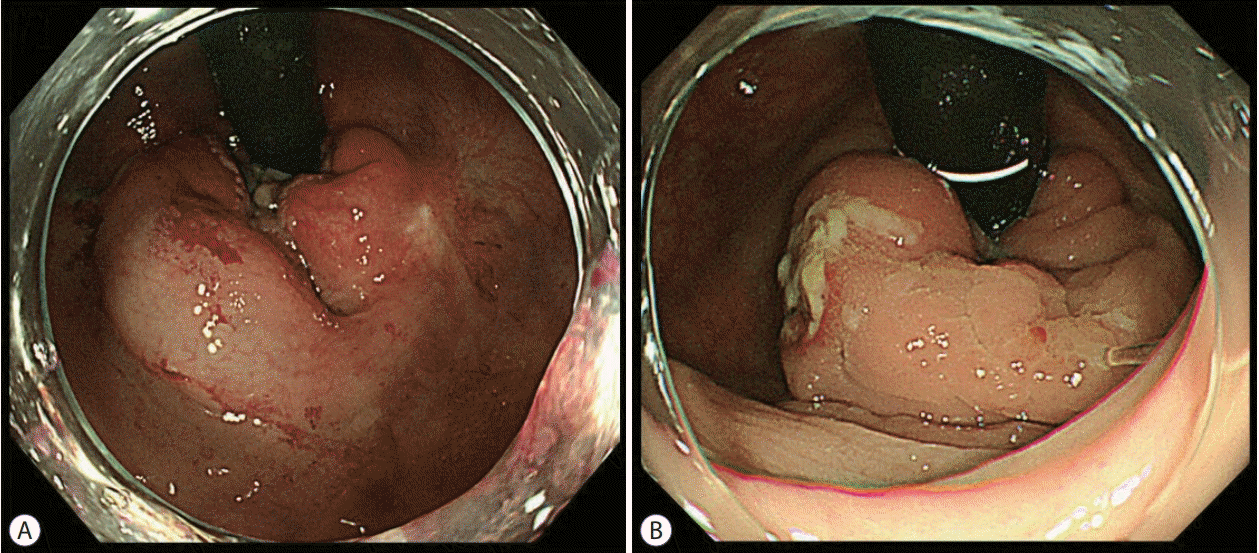
A rectal ulcer detected after endoscopic aluminum potassium sulfate and tannic acid sclerotherapy. (A) Before treatment. (B) Three months after treatment.
Endoscopic aluminum potassium sulfate and tannic acid (ALTA) sclerotherapy for Goligher grade III prolapse. (A) Before treatment. (B) After ALTA injection. (C) After finger massage. (D) At 8 months after treatment.
The Patients’ Characteristics
| Standard ALTA ( |
Endoscopic ALTA ( |
|
|
|---|---|---|---|
| Age | 64.4±13.9 | 61.4±13.1 | 0.58 |
| Sex | |||
| Male | 27 (81.8%) | 31 (64.6%) | 0.09 |
| Female | 6 (18.2%) | 17 (35.4%) | |
| Symptoms | |||
| Bleeding | 11 (33.3%) | 10 (20.9%) | 0.21 |
| Prolapse | 22 (66.7%) | 38 (79.1%) | |
| Grade of hemorrhoids
|
|||
| Ⅱ | 9 (27.3%) | 9 (18.8%) | 0.36 |
| Ⅲ | 24 (72.7%) | 39 (81.2%) | |
| Antithrombotic treatment | 6 (18.2%) | 5 (10.4%) | 0.32 |
ALTA, aluminum potassium sulfate and tannic acid.
Goligher’s classification.
Comparison between Standard and Endoscopic Aluminum Potassium Sulfate and Tannic Acid for the Treatment of Internal Hemorrhoids
| Standard ALTA ( |
Endoscopic ALTA ( |
|
|
|---|---|---|---|
| Injection dose (mL) | 21.9±7.2 | 17.8±3.4 | <0.01 |
| Therapeutic effects | |||
| Cure | 10 (30.3%) | 9 (18.7%) | 0.22 |
| Improvement | 22 (66.7%) | 37 (77.1%) | 0.30 |
| Failure | 1 (3.0%) | 2 (4.2%) | 0.79 |
| Patient satisfaction | |||
| Very satisfied | 20 (60.6%) | 20 (41.7%) | 0.09 |
| Satisfied | 11 (33.3%) | 24 (50.0%) | 0.14 |
| Dissatisfied | 2 (6.1%) | 4 (8.3%) | 0.70 |
| Adverse events (including multiple events) | 4 (12.1%) | 6 (12.5%) | 0.96 |
| Low-grade fever | 1 (3.0%) | 1 (2.1%) | 0.78 |
| Anal pain | 2 (6.1%) | 4 (8.3%) | 0.70 |
| Urinary retention | 1 (3.0%) | 1 (2.1%) | 0.78 |
| Rectal ulcer | 1 (3.0%) | 1 (2.1%) | 0.78 |
| Recurrence | 2 (6.1%) | 4 (8.3%) | 0.70 |
| Within 1 yr | 1 (3.0%) | 3 (6.3%) | 0.51 |
| Follow-up period (days) | 498 (28–1,485) | 676 (28–1,124) |
ALTA, aluminum potassium sulfate and tannic acid.