Abstract
Infection of the central nervous system with Nocardia sp. usually manifests as supratentorial abscesses. Supratentorial and cerebellar abscesses from infection with Nocardia sp. following immunosuppression with long-term corticosteroids for idiopathic thrombocytopenia (ITP) have not been reported. An 83 years-old, human immunodeficiency virus (HIV)-negative, polymorbid male with ITP for which he required corticosteroids since age 53 years developed tiredness, dyspnoea, hemoptysis, abdominal pain, and progressive gait disturbance. Imaging studies of the lung revealed an enhancing tumour in the right upper lobe with central and peripheral necrosis, multiple irregularly contoured hyperdensities over both lungs, and right-sided pleural effusions. Sputum culture grew Nocardia sp. Neurological diagnostic work-up revealed dysarthria, dysphagia, ptosis, hypoacusis, tremor, dysdiadochokinesia, proximal weakness of the lower limbs, diffuse wasting, and stocking-type sensory disturbances. The neurological deficits were attributed to an abscess in the upper cerebellar vermis, myopathy from corticosteroids, and polyneuropathy. Meropenem for 37 days and trimethoprime-sulfamethoxazole for 3 months resulted in a reduction of the pulmonary, but not the cerebral lesions. Therefore, sultamicillin was begun, but without success. Long-term therapy with corticosteroids for ITP may induce not only steroid myopathy but also immune-incompetence with the development of pulmonary and cerebral nocardiosis. Cerebral nocardiosis may not sufficiently respond to long-term antibiotic therapy why switching to alternative antibiotics or surgery may be necessary.
Central nervous system (CNS) affection taking the form of supratentorial, cerebellar, or spinal cord abscesses from hematogenic dissemination of a pulmonary infection with Nocardia sp. is a rare finding,1,2 and the induction of nocardiosis due to long-term therapy with corticosteroids for idiopathic thrombocytopenia (ITP) is more rarely found.3-5 Two of these cases developed supratentorial abscesses. However, both cerebellar and supratentorial abscesses from Nocardia sp. in a patient under corticosteroids for ITP, have not been reported.
The patient is an 83 years-old, human immunodeficiency virus (HIV)-negative, Caucasian male, with a history of recurrent nasal bleedings since childhood, recurrent gingival bleedings, frequent cutaneous hematoma after minor traumata, which were attributed to ITP diagnosed at age 52 years, splenectomy at age 52 years for the ITP, coronary heart disease resulting in two myocardial infarctions at age 52 years, corticosteroids for ITP since age 53 years, steroid-induced diabetes mellitus since age 53 years, requiring insulin since age 78 years, arterial hypertension since years, hyperlipidemia since age 53 years, percutaneous transluminal coronary angioplasty at age 62 years, aortocoronary bypass grafting at ages 64 and 75 years, atrial fibrillation since age 68 years, cataract surgery at ages 71 and 74 years, bilateral carotid artery stenosis requiring stent implantation at age 72 years, a 6-fold stent placing for coronary stenoses at age 75 years, bilateral hypoacusis since age 78 years requiring hearing devices bilaterally and sensori-motor polyneuropathy since age 78 years. Since age 82 years, he also noted a mild cognitive decline and non-specific visual impairment and started to use a stick for walking and a walker since two weeks prior to admission. His family history was positive for diabetes mellitus and short stature in his mother. From age 68 years until admission, he was taking phenprocoumon.
He was admitted at age 83 years because of chronic tiredness, dyspnea, hemoptysis, and abdominal pain. Blood chemical findings showed leucocytosis with a maximum value of 38.8/nL (n, 4.0-9.0/nL) on hospital day (hd) 3, thrombocytopenia with a minimum value of 4/nL (n, 150-450/nL) on hd 15 and hd 18, renal insufficiency, and elevated C-reactive protein with a maximum value of 34.7 mg/dL (n, ≤ 0.6 mg/dL) at hd 3 (Table 1). There were also elevated glutamate-oxalate transaminase, glutamate pyruvate transaminase, and lactate dehydrogenase (Table 1). Creatine-kinase was normal throughout hospitalization. There was slight hypocalcemia (Table 1) and elevation of ferritin to 3817 ng/mL (n, 30-400 ng/mL). Thyroid function parameters were indicative of hyperthyroidism. The HbA1c was 7.4% (n, ≤ 6%). X-ray of the lung revealed multiple reticulonodular infiltrates up to one cm in diameter over both lungs with predominance in the left lower lobe, a pleural effusion in the horizontal interlobar fissure, and an encapsulated effusion in the right-sided diagonal interlobar fissure. X-ray of the lung five weeks earlier had been normal. A CT-scan of the thorax showed an 11×7 cm, enhancing mass lesion in the right upper lobe with central and peripheral necrosis and multiple irregularly contoured hyperdensities over both lungs (Fig. 1). Sputum cultures grew Nocardia sp.
Clinical neurologic examination revealed slight dysarthria, dysphagia, bilateral ptosis, bilateral hypoacusis, postural tremor, left-sided dysdiadochokinesia, weakness for hip extension bilaterally (M5-), reduced tendon reflexes on the upper limbs and absent tendon reflexes on the lower limbs, diffuse wasting, stocking-type sensory disturbances bilaterally, and ataxic stance. He was able to walk with a walker. MRI of the cerebrum showed a hyperintense, enhancing lesion of 4 mm in diameter in the cranial part of the cerebellar vermis (Fig. 2). Additionally, there were small hyperintense lesions in the occipital deep white matter and subcortically bilaterally (Fig. 2). Lumbar puncture was recommended, but contraindicated due to ITP. No manifestations of nocardiosis other than in the lung and cerebrum were detected.
For ITP, the patient received prednisolone 50 mg/d until hd 9. For the general infection, he initially received amoxicillin between hd 2 and hd 4. From hd 4 to hd 7, he received caspofungin and between hd 5 and hd 7 piperazillin. Meropenem was started on hd 8 and maintained until hd 45. He had never developed fever during the entire hospitalization. After dismissal, the patient was switched to a long-term therapy with trimethoprim-sulfamethoxazol. Shortly after admission, oral anticoagulation was stopped because of severe thrombocytopenia of 4,000/nL (Table 1). Since hd 23, corticosteroids had to be restarted because of deteriorating ITP (Table 1). His therapy at dismissal included danazol, furosemide, pantoprazole, atorvastatin, metoprolol, enalapril, amiodarone, prednisolone, folic acid, and insulin in addition to the antibiotic. At the three month follow-up, dimension of the pulmonary lesions had regressed. On the contrary, the cerebral lesions had progressed despite trimethoprim-sulfamethoxazol, promting a switch to sultamicillin and then to meropenem. Unexpectedly, the patient died from acute myocardial infarction shortly thereafter. Autopsy excluded a pulmonary malignancy as the underlying cause of nocardiosis and confirmed that antibiotic therapy was not effective in eradicating the cerebral infection, although cerebral tissue cultures were negative for Nocardia sp.
Nocardiosis is an opportunistic local infection with consecutive hematogenic spread in half of the cases from the Gram-positive, filamentous, eosinophilic organism Nocardia sp. The most frequently found species is N. farcinica,6,7 followed by N. asteroides,8-10 N. paucivorans,11 N. otitidiscaviarum,12 N. brasiliensis,13 N. cyriacigeorgica,14,15 or N. transvalensis.14,16,17 Nocardiosis is an uncommon disease, but its frequency has increased due to the high number of immunosuppressive treatments, improved diagnostic facilities, and increasing survival times of patients with malignancy.18 Patients become infected by inhalation, direct traumatic cutaneous inoculation, or by eating contaminated food.19 The most common primary site of nocardiosis is the respiratory tract, being the origin of hematogenic dissemination with a high incidence to the skin, subcutaneous tissue, or CNS.6,19,20 Extra-pulmonary manifestations of nocardiosis include CNS abscesses or meningitis,6 chorioiditis,6 intraocular vasculitis,21 or renal abscesses.22 Most patients have an underlying chronic diseases or suffer from endogenous or drug-induced immunosuppression with for instance TNF-alpha blockers, but Nocardia can also infect healthy subjects.23 Culture or biopsy followed by identification of the infectious species by PCR is recommended because of the specific therapeutic strategies associated with each species and their different sensitivity to antibiotics.6 Because of the high relapse rates, treatment is recommended to be continued for 12 months.24
CNS nocardiosis usually manifests as cerebral abscesses or rarely as meningitis or chorioiditis. The abscesses usually develop supratentorially but cerebellar abscesses have been also reported (Table 2).2,25-27 Though the preference of Nocardia sp. for the CNS is well known, CNS abscesses from Nocardia sp. are rare and account for only 1-2% of brain abscesses.1,28 In almost half of the patients with pulmonary nocardiosis, dissemination is associated with brain abscesses.10 Early detection and treatment is important because the mortality is three times higher than that of other bacterial cerebral abscesses.28 Since the literature about CNS nocardiosis takes the form of anecdotal reports, small case series, or retrospective studies (Table 2), an optimal treatment approach has not yet been established. Abscesses with Nocardia sp. represent a diagnostic and therapeutic challenge, since they are associated with high mortality rates, particularly in immunocompromised patients. Cerebral abscesses are managed either conservatively with stereotactic aspiration, or by open craniotomy and enucleation.29 Surgical treatment is indicated if the lesions are large and readily accessible or in case of progression of the lesions within two weeks after initiation of the antibiotic treatment. There is growing evidence that the size of the cerebral lesion and clinical and immune status of the patient are relevant to surgical decision making. In spite of the existing controversy with regard to the surgical management of these lesions, an early diagnosis through stereotactic aspiration and the beginning of an antibiotic therapy are essential for a favorable outcome.19 In most cases of cerebral nocardiosis the overall prognosis is favorable upon adequate antibiotic or surgical therapy.30 However, there are several cases with poor outcome (Table 2).18,25,31
The patient presented herein is interesting because of severe side-effects to long-term corticosteroid treatment for ITP, including pulmonary and cerebral nocardiosis, induction of diabetes shortly after initiating corticosteroids at age 53 years, cataract, and possibly steroid myopathy. Whether corticosteroids could be responsible also for arterial hypertension, dermal atrophy, or dermal bleedings remains speculative. Since arterial hypertension had occurred already prior to the administration of steroids, a causal relationship is rather unlikely. It also remains elusive if myopathy was due to the side-effects of corticosteroids alone or due to other causes. The combination of thrombocytopenia, diabetes, and hypoacusis has been reported together with the thiamine-responsive megaloblastic anemia syndrome,32 but anemia was not a dominant feature in the present patient. The combination of diabetes, hyperlipidemia, arterial hypertension, atrial fibrillation, thrombocytopenia, sensorineural hearing loss, the renal cyst, myopathy, and cataract could also be attributed to a systemic metabolic defect as the underlying pathology. Unfortunately, however, further diagnostic work-up into this direction was limited due to the ITP. The cause of the markedly elevated ferritin levels remains elusive, but could be explained with the chronic infection. Pulmonary malignancy was definitively excluded at autopsy. The slightly reduced calcium levels were attributed to hypoalbuminemia. That the antibiotic therapy definitively eradicated the infection with Nocardia sp. remains speculative since negative culture results cannot be reliably interpreted as evidence that the infection was eradicated. Whether the patient was resistent to the applied antibiotics and would have profited from linezolid, as has recently been reported,33 is unknown.
The present case shows that long-term therapy with corticosteroids for ITP may induce immune-incompetence, resulting in the development of pulmonary and cerebral nocardiosis. Cerebral nocardiosis may not sufficiently respond to long-term antibiotic therapy and may necessitate switching to alternative antibiotics or surgery. Long-term corticosteroid therapy may induce secondary steroid myopathy or enhance primary metabolic myopathy.
Figures and Tables
Fig. 1
CT-scan of the thorax showing an enhancing mass lesion in the right upper lobe with central and peripheral necrosis and multiple irregularly contoured hyperdensities over both lungs.
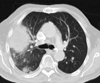
Fig. 2
T1-weighted MRI images of the brain show a slightly enhancing mass lesion in the cerebellar vermis (A) and a hyperintense lesion in the right occipital lobe (B).
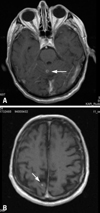
Table 1
Hematological and Blood Chemical Values of the Described Patient during Hospitalisation for Nocardiosis

RL, reference limit; hd, hospital day; Leuko, leukocyte count; Ery, erythrocyte count; Thrombo, thrombocyte count; BUN, blood urea nitrogen; Krea, creatinine; CRP, C-reactive protein; GOT, glutamate-oxalate-transaminase; GPT, glutamate-pyruvate-transaminase; LDH, lactate-dehydrogenase; CPK, creatine-phosphokinase; nd, not done.
References
1. Fleetwood IG, Embil JM, Ross IB. Nocardia asteroides cerebral abscess in immunocompetent hosts: report of three cases and review of surgical recommendations. Surg Neurol. 2000. 53:605–610.

2. Oerlemans WG, Jansen EN, Prevo RL, Eijsvogel MM. Primary cerebellar nocardiosis and alveolar proteinosis. Acta Neurol Scand. 1998. 97:138–141.
3. Ando T, Usa T, Ide A, Abe Y, Sera N, Tominaga T, et al. Pulmonary nocardiosis associated with idiopathic thrombocytopenic purpura. Intern Med. 2001. 40:246–249.

4. Lai CC, Lee LN, Teng LJ, Wu MS, Tsai JC, Hsueh PR. Disseminated Nocardia farcinica infection in a uraemia patient with idiopathic thrombocytopenia purpura receiving steroid therapy. J Med Microbiol. 2005. 54:1107–1110.

5. Wakhlu A, Agarwal V, Dabadghao S, Prasad KN, Nityanand S. Nocardiosis in patients of chronic idiopathic thrombocytopenic purpura on steroids. J Assoc Physicians India. 2004. 52:591–593.
6. Schriever S, Mistry-Burchardi N, Grabein B, Näbauer M, Siebold C, Hoops JP, et al. [Nocardia farcinica: life-threatening chorioiditis under systemic Immunosuppression]. Klin Monbl Augenheilkd. 2002. 219:164–167.
7. Yildiz O, Alp E, Tokgoz B, Tucer B, Aygen B, Sumerkan B, et al. Nocardiosis in a teaching hospital in the Central Anatolia region of Turkey: treatment and outcome. Clin Microbiol Infect. 2005. 11:495–499.

8. Borchers M, von der Mülbe B, Teikemeier F, Theegarten D. [Pulmonary nocardiasis with abscesses spreading to cerebrum, cerebellum and orbits]. Dtsch Med Wochenschr. 2006. 131:1085–1088.

9. Murray RJ, Himmelreich U, Gomes L, Ingham NJ, Sorrell TC. Cerebral nocardiosis characterized by magnetic resonance spectroscopy in vivo. Clin Infect Dis. 2002. 34:849–852.

10. Nakamura S, Mihara T, Hitotsumatsu T, Soejima Y, Izumikawa K, Seki M, et al. [Case report of disseminated nocardiosis complicated in an elderly person with pneumoconiosis]. Kansenshogaku Zasshi. 2006. 80:721–725.

11. Khan SN, Sanche SE, Robinson CA, Pirouzmand F. N. paucivorans infection presenting as a brain abscess. Can J Neurol Sci. 2006. 33:426–427.
12. Tatti KM, Shieh WJ, Phillips S, Augenbraun M, Rao C, Zaki SR. Molecular diagnosis of Nocardia farcinica from a cerebral abscess. Hum Pathol. 2006. 37:1117–1121.

13. Konovalov AN, Khaitov RM, Shimanskií VN, Aleksandrova IA, Pinegin BV, Serkov SV, et al. [Multiple cerebral abscess caused by Nocardia brasilientis]. Zh Vopr Neirokhir Im N N Burdenko. 2005. 1:39–43.
14. Alp E, Yildiz O, Aygen B, Sumerkan B, Sari I, Koc K, et al. Disseminated nocardiosis due to unusual species: two case reports. Scand J Infect Dis. 2006. 38:545–548.
15. Barnaud G, Deschamps C, Manceron V, Mortier E, Laurent F, Bert F, et al. Brain abscess caused by Nocardia cyriacigeorgica in a patient with human immunodeficiency virus infection. J Clin Microbiol. 2005. 43:4895–4897.
16. Mukhopadhyay C, Sahu RN, Ayyagari A. Nocardial brain abscesses in a HIV positive patient misinterpreted as tubercular brain abscesses. Nepal Med Coll J. 2005. 7:74–76.
17. Yorke RF, Rouah E. Nocardiosis with brain abscess due to an unusual species, Nocardia transvalensis. Arch Pathol Lab Med. 2003. 127:224–226.
18. Oshiro S, Ohnishi H, Ohta M, Tsuchimochi H. Intraventricular rupture of Nocardia brain abscess--case report. Neurol Med Chir (Tokyo). 2003. 43:360–363.
19. Aboal C, Salamano R, Braselli A, Mansilla M, Galaret M, Pedreira W. [Recurrent brain abscess caused by Nocardia asteroides in a patient with primary polycythaemia]. Arq Neuropsiquiatr. 2006. 64:526–529.
20. Peters BR, Saubolle MA, Costantino JM. Disseminated and cerebral infection due to Nocardia farcinica: diagnosis by blood culture and cure with antibiotics alone. Clin Infect Dis. 1996. 23:1165–1167.
21. Rafiei N, Tabandeh H, Bhatti MT, Monshizadeh R. Retinal fibrovascular proliferation associated with Nocardia subretinal abscess. Eur J Ophthalmol. 2006. 16:641–643.

22. Elmaci I, Senday D, Silav G, Ekenel F, Balak N, Ayan E, et al. Nocardial cerebral abscess associated with mycetoma, pneumonia and membranoproliferative glomerulonephritis. J Clin Microbiol. 2007. 45:2072–2074.
23. Sonesson A, Oqvist B, Hagstam P, Björkman-Burtscher IM, Miörner H, Petersson AC. An immunosuppressed patient with systemic vasculitis suffering from cerebral abscesses due to Nocardia farcinica identified by 16S rRNA gene universal PCR. Nephrol Dial Transplant. 2004. 19:2896–2900.
24. Sabuncuoglu H, Cibali Açikgo ZZ, Caydere M, Ustün H, Semih Keskil I. Nocardia farcinica brain abscess: a case report and review of the literature. Neurocirugia (Astur). 2004. 15:600–603.
25. Durmaz R, Atasoy MA, Durmaz G, Adapinar B, Arslanta A, Aydinli A, et al. Multiple nocardial abscesses of cerebrum, cerebellum and spinal cord, causing quadriplegia. Clin Neurol Neurosurg. 2001. 103:59–62.
26. Eisenblätter M, Disko U, Stoltenburg-Didinger G, Scherbl H, Scherübl KP, Roth A, et al. Isolation of Nocardia paucivorans from the cerebrospinal fluid of a patient with relapse of cerebral nocardiosis. J Clin Microbiol. 2002. 40:3532–3534.
27. Kayacan SM, Türkmen A, Aliş H, Imer M, Barlas O, Ecder ST, et al. Successful therapy combined with surgery for severe posttransplant nocardiosis. J Nephrol. 2001. 14:304–306.
28. Shin JH, Lee HK. Nocardial brain abscess in a renal transplant recipient. Clin Imaging. 2003. 27:321–324.
29. Valarezo J, Cohen JE, Valarezo L, Spektor S, Shoshan Y, Rosenthal G, et al. Nocardial cerebral abscess: report of three cases and review of the current neurosurgical management. Neurol Res. 2003. 25:27–30.

30. Soto-Hernández JL, Moreno-Andrade T, Góngora-Rivera F, Ramírez-Crescencio MA. Nocardia abscess during treatment of brain toxoplasmosis in a patient with aids, utility of proton MR spectroscopy and diffusion-weighted imaging in diagnosis. Clin Neurol Neurosurg. 2006. 108:493–498.

31. Roquer J, Pou A, Herraiz J, Campodarve I, Sequeira T, Vilató J, et al. Primary cerebral abscess due to nocardia presenting as 'ghost tumor'. Clinical and pathological study. Eur Neurol. 1990. 30:254–257.
32. Diaz GA, Banikazemi M, Oishi K, Desnick RJ, Gelb BD. Mutations in a new gene encoding a thiamine transporter cause thiamine-responsive megaloblastic anaemia syndrome. Nat Genet. 1999. 22:309–312.

33. Jodlowski TZ, Melnychuk I, Conry J. Linezolid for the treatment of Nocardia spp. infections. Ann Pharmacother. 2007. 41:1694–1699.

34. Hashimoto M, Johkura K, Ichikawa T, Shinonaga M. Brain abscess caused by Nocardia nova. J Clin Neurosci. 2008. 15:87–89.

35. Kennedy KJ, Chung KH, Bowden FJ, Mews PJ, Pik JH, Fuller JW, et al. A cluster of nocardial brain abscesses. Surg Neurol. 2007. 68:43–49.

36. Oztürk S, Tufan F, Alişir S, Görçin S, Güven D, Cağatay A, et al. A case of isolated Nocardia asteroides brain abscess in a kidney transplant recipient. Transplant Proc. 2006. 38:3121–3124.

37. Dahan K, El Kabbaj D, Venditto M, Pastural M, Delahousse M. Intracranial Nocardia recurrence during fluorinated quinolones therapy. Transpl Infect Dis. 2006. 8:161–165.

38. Ghalib MB, Hassan IS. Prolonged unconsciousness in a patient with End-stage Renal Disease. Saudi J Kidney Dis Transpl. 2006. 17:273–277.
39. Kilincer C, Hamamcioglu MK, Simsek O, Hicdonmez T, Aydoslu B, Tansel O, et al. Nocardial brain abscess: review of clinical management. J Clin Neurosci. 2006. 13:481–485.

40. Ozaras R, Mert A, Hakko E, Tabak F, Ozturk R. Pulmonary, cerebral, and soft tissue nocardiosis. Lancet Infect Dis. 2004. 4:222.

41. Pieroth L, Winterkorn JM, Schubert H, Millar WS, Kazim M. Concurrent sino-orbital aspergillosis and cerebral nocardiosis. J Neuroophthalmol. 2004. 24:135–137.

42. Oshiro S, Ohnishi H, Ohta M, Tsuchimochi H. Intraventricular rupture of Nocardia brain abscess--case report. Neurol Med Chir (Tokyo). 2003. 43:360–363.

43. Börm W, Gleixner M. Nocardia brain abscess misinterpreted as cerebral infarction. J Clin Neurosci. 2003. 10:130–132.

44. Montoya JP, Carpenter JL, Holmes GP, Hurley DL, Winn R. Disseminated Nocardia transvalensis infection with osteomyelitis and multiple brain abscesses. Scand J Infect Dis. 2003. 35:189–196.

45. Mooraki A, Jenabi A, Bastani B. Resolution of pulmonary and cerebral nocardiosis in renal transplant patient despite continued immunosuppression: a case report. Transplant Proc. 2003. 35:2694–2695.
46. Murray RJ, Himmelreich U, Gomes L, Ingham NJ, Sorrell TC. Cerebral nocardiosis characterized by magnetic resonance spectroscopy in vivo. Clin Infect Dis. 2002. 34:849–852.

47. Malincarne L, Marroni M, Farina C, Camanni G, Valente M, Belfiori B, et al. Primary brain abscess with Nocardia farcinica in an immunocompetent patient. Clin Neurol Neurosurg. 2002. 104:132–135.

48. Lee GY, Daniel RT, Brophy BP, Reilly PL. Surgical treatment of nocardial brain abscesses. Neurosurgery. 2002. 51:668–671.

49. Weber L, Yium J, Hawkins S. Intracranial Nocardia dissemination during minocycline therapy. Transpl Infect Dis. 2002. 4:108–112.
50. Acar T, Arshad M. Nocardia asteroides cerebral abscess in a renal transplant recipient: short report. Acta Chir Belg. 2002. 102:470–471.

51. Vialle R, Aghakhani N, Otayza F, Monteanu V, Safi H, Tadie M. [Nocardia farcinica brain abscess: clinical and specific radiological findings and management. Report of two cases in immunononcompromised patients]. Neurochirurgie. 2002. 48:516–521.
52. Marlowe M, Ali-Ahmad D, Cherrick I, Higgins MJ, Kiska DL, Domachowske JB. Central nervous system nocardiosis in an immunocompetent child. Pediatr Infect Dis J. 2000. 19:661–662.

53. Hartmann A, Halvorsen CE, Jenssen T, Bjørneklett A, Brekke IB, Bakke SJ, et al. Intracerebral abscess caused by Nocardia otitidiscaviarum in a renal transplant patient--cured by evacuation plus antibiotic therapy. Nephron. 2000. 86:79–83.

54. Prieto de Paula JM, Folgueral Pérez M, Galán Ramos J, Cosín Ochaíta J. [Persistent neutrophilic meningitis and brain abscesses in a male patient with pulmonary sarcoidosis and corticosteroid therapy. Meningitis and cerebral abscesses due to Nocardia sp. Pulmonary sarcoidosis, steroid treatment]. Rev Clin Esp. 2000. 200:489–490.
55. Palomares M, Martinez T, Pastor J, Osuna A, Bravo JA, Alvarez G, et al. Cerebral abscess caused by Nocardia asteroides in renal transplant recipient. Nephrol Dial Transplant. 1999. 14:2950–2952.

56. Cosnett JE, Moodley M, Bill PL, Bullock R. Operculum syndrome from brain abscess in a left-hander. J Neurol Neurosurg Psychiatry. 1988. 51:307–308.

57. Mogilner A, Jallo GI, Zagzag D, Kelly PJ. Nocardia abscess of the choroid plexus: clinical and pathological case report. Neurosurgery. 1998. 43:949–952.

58. Orsolon P, Bagni B, Talmassons G, Guerra UP. Evaluation of a patient with a brain abscess caused by nocardia asteroides infection with Ga-67 and Tc-99m HMPAO leukocytes. Clin Nucl Med. 1997. 22:407–408.

59. Fuzier R, Vironneau M, Noves F, Cathala B, Trémoulet M. [Cerebral abscess caused by Nocardia]. Ann Fr Anesth Reanim. 1997. 16:1049–1050.
60. Machado CM, Macedo MC, Castelli JB, Ostronoff M, Silva AC, Zambon E, et al. Clinical features and successful recovery from disseminated nocardiosis after BMT. Bone Marrow Transplant. 1997. 19:81–82.

61. Pyhtinen J, Pääkkö E, Jartti P. Cerebral abscess with multiple rims of MRI. Neuroradiology. 1997. 39:857–859.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download