TO THE EDITOR: Immune thrombocytopenia (ITP) is an immune-mediated disease characterized by an isolated decrease in platelet count. There are multiple mechanisms underlying this disease, including autoantibodies against platelet membrane proteins and impaired platelet production [1]. In children, ITP is usually acute and the children have a spontaneous recovery, whereas in adults, ITP is chronic and irreversible. First-line therapy for ITP is corticosteroids, which have response rates of 50%–90%; however, a durable response is observed in only 10–30% of patients. For steroid-refractory ITP, there are several treatments, such as splenectomy, danazol, azathioprine, cyclophosphamide, cyclosporine, rituximab, and thrombopoietin (TPO) receptor agonists [2].
Splenectomy induces a durable complete response in approximately 70% of patients with steroid-refractory ITP. However, it is associated with operative risks and postoperative complications such as encapsulated bacterial infections [3]. Rituximab has a good response rate of approximately 60% and a complete response rate of approximately 40%, though the durability over 5 years is only 15–20% [4]. Recently, the TPO receptor agonists, romiplostim and eltrombopag, demonstrated potent activity and had a response rate of more than 80%. However, However, their mechanism of action require relatively long maintenance period [5]. Other third-line therapies demonstrate minimal efficacy and their response duration is often short.
Alemtuzumab, an anti-CD52 humanized monoclonal antibody, has shown strong durable immune suppression and has been used for lymphoproliferative disorders and autoimmune diseases such as multiple sclerosis. One study reported the safety and efficacy of alemtuzumab plus low-dose rituximab in patients with steroid-refractory ITP. The overall response rate was 100% and the median response duration was 46 weeks [6]. Another study reported about a patient with ITP and pure red cell aplasia who showed dramatic elevation of platelet count and hemoglobin levels after alemtuzumab treatment [7]. Since there are few reports assessing the utility of alemtuzumab for treating ITP, this report documents the successful treatment of steroid-refractory ITP with alemtuzumab.
We administered alemtuzumab to two patients (patient 1 and patient 2) who were diagnosed with ITP several years ago. Both patients were female, aged 48 and 50 years, respectively. Both had severe steroid-refractory thrombocytopenia and did not respond to other treatments. Alemtuzumab was administered subcutaneously at a dose of 30 mg on days 1, 3, 5, 29, 31, and 33. We administrated 100 mg hydrocortisone intravenously and 650 mg acetaminophen orally 30 minutes prior to alemtuzumab administration to prevent acute hypersensitivity reactions. Prophylaxis for Pneumocystis jirovecii infection with co-trimoxazole was maintained for 12 weeks after treatment or until the CD4+ T-lymphocyte count increased to ≥200/µL. We performed complete blood counts and cytomegalovirus (CMV) antigen tests weekly and chest radiographies once every 4 weeks. We also tested CD4+, CD8+, and CD4+/CD25+ T-lymphocyte counts before alemtuzumab administration and then repeated every 4 weeks.
A complete response (CR) was defined by a platelet count of ≥100×109/L and a partial response (PR) was defined by a platelet count of ≥50×109/L or a platelet count increment of ≥30×109/L compared to baseline values. Both the platelet count and platelet count increments had to be maintained for 4 weeks. If the platelet recovery was sustained for less than 4 weeks, we classified the case as having a minor response. This study was approved by the Institutional Review Board of Samsung Medical Center, Seoul, Republic of Korea.
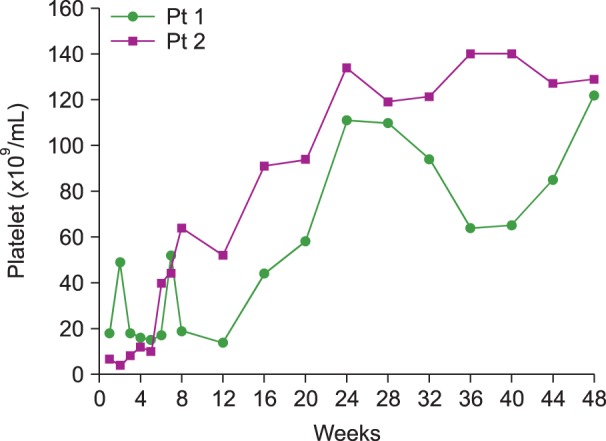
Patient 1 showed no response to treatment including high-dose steroids, splenectomy, danazol, rituximab, tacrolimus, mycophenolate, and azathioprine. Thus, alemtuzumab administration was started in August 2011. Her platelet count increased in the first week, from 18×109/L to 49×109/L, but did not sustain for a long duration. After that, the platelet count fluctuated for several weeks, rapidly increasing after week 16; finally, PR was achieved at week 20 and CR at week 24. At week 5, patient 1 showed 1,156 per 200,000 polymorphonuclear leukocytes in pp65 CMV antigenemia test and elevated liver enzyme levels indicating CMV hepatitis, but it was managed with oral ganciclovir. We did not administer alemtuzumab on day 33 because of the hepatitis. This was the only documented infection or complication after alemtuzumab treatment. The CR was maintained through November 2014. This patient did not come back to the clinic thereafter.
Patient 2 had severe thrombocytopenia with a platelet count of less than 10,000×109/L; the patient required repeated platelet transfusions to control bleeding. Treatment with alemtuzumab began in April 2012. Her platelet count increased gradually at week 5; she achieved CR at week 24 and CR was sustained for 3 years through October 2015 (Fig. 1). There was no definitive infection or complication.
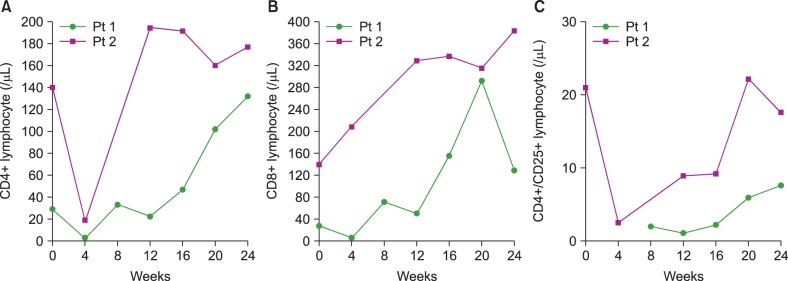
Lymphocyte count had a tendency to decrease after alemtuzumab administration, but it recovered. The CD4+ and CD4+/CD25+ lymphocyte counts were suppressed until week 12; however, their counts increased after week 16 up to the baseline levels. The CD8+ lymphocyte count showed a similar pattern except for much higher increase in those of week 16. This pattern was related to the platelet response pattern from alemtuzumab treatment (Fig. 2).
CD52 is highly expressed on the membranes of most B and T lymphocytes, monocytes, macrophages, eosinophils, natural killer cells, and dendritic cells [89]. Watanabe et al. suggested that CD52 may induce and activate regulatory T-cells [10]. Alemtuzumab, a CD52 monoclonal antibody, causes strong prolonged immunosuppression through complement-mediated and antibody-dependent cellular cytotoxicity (ADCC) that it is used in many immune-related diseases such as multiple sclerosis, chronic lymphocytic leukemia, and graft-versus-host disease [111213]. As the pathogenesis of ITP is heterogeneous, responses to single agents are usually unsuccessful or transient. Alemtuzumab could induce a dramatic immunologic response because it acts on several immunologic effector and regulatory cells. However, the durable response of alemtuzumab in cases of steroid-refractory ITP cannot be explained. Hypothetically, alemtuzumab might have caused a significant immune reconstitution; this is similar to the findings of previous studies that reported substantial changes in T-cell composition in ITP patients with a durable response [1415].
In these cases, alemtuzumab showed excellent and prolonged responses in two patients with refractory ITP. Both patients showed slow initial platelet count increases but demonstrated rapid improvement between weeks 8 and 24. Surprisingly, the response was sustained for 3 years. Although there was a high risk of infection because of the potent immunosuppression by alemtuzumab, no clinically significant infectious complications occurred in either patient, despite their age and previous treatment with multiple immunosuppressants. One patient experienced CMV hepatitis at week 5, but it was well manageable with ganciclovir. CD4+, CD8+, and CD4+/CD25+ lymphocyte counts initially decreased but recovered several months later. In conclusion, we suggest that alemtuzumab could be a viable option for patients with refractory and heavily pre-treated ITP, but this observation requires confirmation with large-scale prospective studies.
References
1. Cines DB, Bussel JB, Liebman HA, Luning Prak ET. The ITP syndrome: pathogenic and clinical diversity. Blood. 2009; 113:6511–6521. PMID: 19395674.

2. Provan D, Stasi R, Newland AC, et al. International consensus report on the investigation and management of primary immune thrombocytopenia. Blood. 2010; 115:168–186. PMID: 19846889.

3. Vianelli N, Galli M, de Vivo A, et al. Efficacy and safety of splenectomy in immune thrombocytopenic purpura: long-term results of 402 cases. Haematologica. 2005; 90:72–77. PMID: 15642672.
4. Arnold DM, Dentali F, Crowther MA, et al. Systematic review: efficacy and safety of rituximab for adults with idiopathic thrombocytopenic purpura. Ann Intern Med. 2007; 146:25–33. PMID: 17200219.

5. Cheng G, Saleh MN, Marcher C, et al. Eltrombopag for management of chronic immune thrombocytopenia (RAISE): a 6-month, randomised, phase 3 study. Lancet. 2011; 377:393–402. PMID: 20739054.

6. Gómez-Almaguer D, Solano-Genesta M, Tarín-Arzaga L, et al. Low-dose rituximab and alemtuzumab combination therapy for patients with steroid-refractory autoimmune cytopenias. Blood. 2010; 116:4783–4785. PMID: 20841509.

7. Pham DC, Guthrie TH, Villas BH, Salazar E. Refractory idiopathic pure red cell aplasia complicated by immune thrombocytopenia successfully treated with subcutaneous alemtuzumab. Am J Hematol. 2008; 83:603.

8. Buggins AG, Mufti GJ, Salisbury J, et al. Peripheral blood but not tissue dendritic cells express CD52 and are depleted by treatment with alemtuzumab. Blood. 2002; 100:1715–1720. PMID: 12176892.

9. Ratzinger G, Reagan JL, Heller G, Busam KJ, Young JW. Differential CD52 expression by distinct myeloid dendritic cell subsets: implications for alemtuzumab activity at the level of antigen presentation in allogeneic graft-host interactions in transplantation. Blood. 2003; 101:1422–1429. PMID: 12393688.

10. Watanabe T, Masuyama J, Sohma Y, et al. CD52 is a novel costimulatory molecule for induction of CD4+ regulatory T cells. Clin Immunol. 2006; 120:247–259. PMID: 16797237.

11. Golay J, Manganini M, Rambaldi A, Introna M. Effect of alemtuzumab on neoplastic B cells. Haematologica. 2004; 89:1476–1483. PMID: 15590398.
12. Zent CS, Chen JB, Kurten RC, Kaushal GP, Lacy HM, Schichman SA. Alemtuzumab (CAMPATH 1H) does not kill chronic lymphocytic leukemia cells in serum free medium. Leuk Res. 2004; 28:495–507. PMID: 15068903.

13. Clynes RA, Towers TL, Presta LG, Ravetch JV. Inhibitory Fc receptors modulate in vivo cytotoxicity against tumor targets. Nat Med. 2000; 6:443–446. PMID: 10742152.
14. Stasi R, Del Poeta G, Stipa E, et al. Response to B-cell depleting therapy with rituximab reverts the abnormalities of T-cell subsets in patients with idiopathic thrombocytopenic purpura. Blood. 2007; 110:2924–2930. PMID: 17548576.
15. Sfikakis PP, Boletis JN, Lionaki S, et al. Remission of proliferative lupus nephritis following B cell depletion therapy is preceded by down-regulation of the T cell costimulatory molecule CD40 ligand: an open-label trial. Arthritis Rheum. 2005; 52:501–513. PMID: 15693003.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download