Abstract
Conventional transcatheter arterial chemoembolization (c-TACE) is a widely used first-line palliative treatment for patients with unresectable hepatocellular carcinoma (HCC). Despite the effectiveness of c-TACE, to date, technique and procedure scheduling has not yet been standardized. Drug-eluting microspheres (DEMs) were therefore introduced to ensure more sustained and tumor-selective drug delivery for permanent embolization. These DEMs can load various drugs and release them in a sustained manner over a prolonged period. This approach ensures the delivery of high concentrations of chemotherapeutic agents to tumors, without increasing systemic concentrations, and promote tumor ischemia and necrosis. This review summarizes the recent advances in the use of DEM-TACE to treat HCC.
Drug-eluting microspheres (DEMs), also called drug-eluting beads (DEBs), are a type of drug delivery system that was introduced in 2006 for the treatment of hepatocellular carcinoma (HCC) (1). Although conventional transcatheter arterial chemoembolization (c-TACE) is considered the first-line palliative treatment for patients with unresectable HCC (2), it has important technical and scheduling drawbacks that have not yet been standardized. Moreover, lipiodol oil used in c-TACE may cause severe pain in some patients. DEM-transcatheter arterial chemoembolization (DEM-TACE) was produced to overcome these drawbacks and ensure sustained and tumor-selective drug delivery while delivering permanent embolization (1). The microspheres can load a variety of drugs through mechanisms such as ion exchange or absorption (134), and when infused directly into target tumors through catheters or microcatheters, can release chemotherapeutic agents in a sustained manner over a prolonged period (13).
Chemotherapeutic agents delivered through this approach are not only cytotoxic to tumor cells but also cause ischemia and tumor necrosis. Moreover, DEM-TACE results in high concentrations of chemotherapeutic agents being delivered to tumors without elevating systemic concentrations. Therefore, the typical adverse effects associated with c-TACE may be reduced by using DEM-TACE. In this review, recent advances in the use of DEM-TACE for the treatment of HCC are summarized.
Many types of microspheres are commercially available (Table 1) (56). The microspheres most commonly used in clinical practice are DC Bead® (BTG, London, UK) and HepaSphere™ (Merit Medical, South Jordan, UT, USA). Other microspheres include Oncozene™ TANDEM® (CeloNova BioSciences, Inc., San Antonio, TX, USA) and LifePearl® (Terumo European Interventional Systems, Leuven, Belgium). All are negatively charged and allow the uploading of different types of chemotherapeutic agents. Doxorubicin is most frequently used in DEM-TACE, and in vitro, more than 99% of a solution can be loaded into any of the previous four microspheretypes within 1 hour (5). However, other microsphere characteristics differ by product, and some can efficiently load doxorubicin concentrations as high as 45 mg/mL, regardless of the bead size (7) (Fig. 1).
Many preclinical in vivo studies have shown the sustained release of pharmacologically active concentrations of doxorubicin into target cancerous tissues over several weeks, with drug diffusion of over 600 µm from the circumference of the beads (89). The local delivery of doxorubicin in HCC explants was compared among patients embolized receiving DEBs with histological modifications. The results revealed that the distribution of the beads in the cancerous and surrounding tissue vasculature could be controlled. Indeed, drug release started within the first few hours of bead infusion and was maintained for at least 1 month, with effective necrosis of the target tissue (10).
It has also been reported that peak serum doxorubicin concentrations are significantly lower after DEB-TACE than after c-TACE (111213). This suggests that the interaction time between the chemotherapeutic agent and tumor cells is increased, while the plasma concentration of the agent is decreased, resulting in more effective treatment and fewer systemic complications (14).
Drug-eluting microspheres are available in different sizes (Table 1). Although large-caliber microspheres (>300 µm) can cause ischemia, they may be associated with premature embolization of the feeding artery before the chemotherapeutic agent can be completely effective (15). Premature arterial embolization may damage the blood vessel, prevent subsequent tumor-directed treatment (16), and cause hypoxia-induced neoangiogenesis (1718). Infusion of 100–300-µm microspheres in an animal model of liver cancer resulted in the delivery of these microspheres intothe tumor or near its margins, justifying their use for precise drug delivery or embolization (19). Although many institutions in Korea suggest using 100–300-µm microspheres in the procedure (20), the choice of microsphere size depends on many factors, including tumor size, feeding artery diameter, personal preference and experience, and the presence or absence of an arteriovenous shunt that increases the risk of pulmonary complications (21).
In general, the indications and contraindications for DEM-TACE are similar to those for c-TACE (Tables 2, 3). Its use depends on the size and vascularity of the tumor, the number of tumors, whether the lesion is infiltrative, and whether the lesion extends to surrounding vessels. Other factors associated with the use of DEM-TACE are the results of liver function tests and possibility of cure. The guidelines of the European Association for the Study of the Liver-European Organisation for Research and Treatment of Cancer recommend TACE as a first-line treatment for asymptomatic patients with preserved liver function who have multinodular lesions but show no evidence of vascular invasion or extrahepatic spread (intermediate stage, or stage B in the Barcelona Clinic Liver Cancer [BCLC] staging system) (222324). TACE is also an appropriate palliative treatment for patients who require curative treatment but are not suitable for surgery or percutaneous ablation (25). Moreover, DEM-TACE is considered a better option than c-TACE for patients with more advanced disease or cardiac failure, and for those expected to have severe post-embolic toxicity (26).
Although liver transplantation is a curative treatment for appropriately selected patients with HCC, a donor liver may not be immediately available, requiring the candidate to wait. By inducing ischemic tumor necrosis and inhibiting tumor progression, both c-TACE and DEM-TACE can extend the period, during which the patient meets the morphologic criteria for liver transplantation, as well as increasing the potential for curative treatment (2728). The reduction of active tumor tissue in such a setting would suggest that patients initially not fulfilling the morphologic criteria may ultimately be shown to benefit from liver transplantation (29).
Drug-eluting microsphere transcatheter arterial chemoembolization is contraindicated inpatients with decompensated baseline liver function, massive arteriovenous shunts, or infiltrative HCC (213031). While portal vein thrombosis was previously considered a contraindication for transarterial therapy (32), it has not been shown to affect survival after DEM-TACE or c-TACE adversely (333435). Other contraindications are related to the chemotherapeutic drugs and angiographic procedures, such as hypersensitivity to contrast media, renal insufficiency, uncorrectable coagulopathy, and peripheral vascular disease preventing arterial access.
The principle of DEM-TACE is to standardize targeted chemotherapeutic agent delivery to ensure a good ischemic effect on tumors and to overcome the drawbacks associated with c-TACE. Although, Lencioni et al. (36) previously described a technique that they proposed as a general guideline for DEM-TACE use (Table 4), there are some approaches that consider each patient and tumor characteristic. DEM agents can be applied to the extrahepatic arteries supplying the tumors (20); however, to the best of our knowledge, no studies have focused on the safety and efficacy of DEM-TACE in this context.
An appropriate planning of DEM-TACE requires hepatic tri-phasic dynamic computed tomography (CT) or magnetic resonance imaging to optimize a microsphere delivery of a chemotherapeutic drug to a lesion. Cone-beam CT is considered a useful imaging tool for determining vascular anatomy and anatomical variants, facilitating better tumor targeting and the avoidance of non-target embolization, as well as predicting the efficacy of DEM-TACE (Fig. 2) (3738). Recent literature has indicated that it should be considered a standard technique, particularly in cases of difficult lesion access, such as when lesions are present in the caudate lobe or when they have extrahepatic collateral arteries (39).
Although a meta-analysis has shown that routine antibiotics may be unnecessary, the judicious use of antibiotics is appropriate for patients at an increased risk of infection (40). Risk factors include old age, larger nodules, bilioenteric anastomosis, biliary stent implantation, compromised hepatic function, hypoalbuminemia, portal vein cancer embolus, and diabetes mellitus (4142). The antibiotics used should cover gram-positive and gramnegative aerobic and anaerobic organisms, according to previous reports (40).
Arterial super-selection at the segmental, subsegmental, and tumor-feeding artery levels was previously considered a favorable prognostic factor for disease-free survival among patients with HCC undergoing c-TACE (43). This was supported by animal studies showing the retention and sustained release of doxorubicin after DC Bead® embolization for up to 90 days after liver embolization (44). Hence, the ischemic effect due to DC Bead® reflux may continue for more than 3 months and be more severe than that produced by c-TACE (44). Recently, a case of multi-organ ischemia was described as a consequence of DC Bead® reflux into the gastroduodenal, right gastric, right gastroepiploic, left gastric, and splenic arteries (45). Therefore, it is preferable for all tumor-feeding arteries to be super-selected to prevent non-target embolization (Fig. 3).
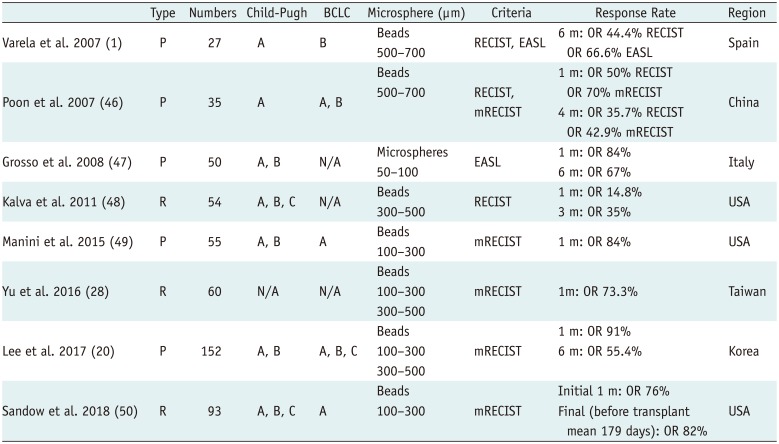
Initial clinical studies confirmed an association between favorable pharmacokinetic profiles of microspheres and good tumor response (Table 5). Objective response rates, defined as the percentages of patients who achieve complete or partial response according to the modified Response Evaluation Criteria in Solid Tumors (RECIST) guidelines, have been found to range from 66.6% to 76.8% (Fig. 4) (14647). Since the initial reports, multiple clinical trials have reinforced the use of DEM-TACE in patients with HCC (20284849). However, clinical results depended on many factors, including cancer stage, number of patients, type and size of microsphere, and follow-up duration.
In a recent Korean prospective multicenter clinical trial using DEB-TACE among 152 patients, 103 (67.8%) had a Child-Pugh class of A5, 114 (75.0%) had a performance status of 0, and 77 (50.7%) had stage A disease according to the BCLC classification (20). One month after treatment with doxorubicin-loaded DC Bead®, the complete and objective response rates were 40.1% and 91.4%, respectively. Among the 121 patients who were analyzed at 6 months, the complete and objective response rates were 43.0% and 55.4%, respectively (20).
Yu et al. (28) conducted a prospective study of the effects of DEB-TACE in 60 patients with HCC who were evaluated before liver transplantation. Following initial DEB-TACE with DC Bead®, 40% achieved complete response and 33.3% achieved partial response according to the modified RECIST guidelines; among these, 15% had stable disease and 11.7% had progressive disease (Fig. 5). No patient experienced major complications or hepatic failure after the procedure. Of the 23 patients who initially did not meet the University of California San Francisco criteria for liver transplantation, 17 (73.9%) were successfully downstaged following DEB-TACE. The high percentage of patients who experienced tumor necrosis with minimal discomfort and without major complications suggests that DEB-TACE may be a safe and effective bridge therapy to liver transplantation in patients with HCC and poor liver function (28).
A retrospective single-center cohort analysis was conducted among 93 consecutive patients between 2011 to 2016 who underwent DEB-TACE using 100–300 µm LC Bead® (BTG) before liver transplantation (50). This showed a favorable long-term treatment response in 87% of patients with histologically low-grade tumors (grades 0–2), including complete and partial response rates of 49% and 38%, respectively. Incontrast, only 33% of patients with histologically high-grade tumors (grades 3–4) responded, with complete and partial response rates of 0% and 33%, respectively (p = 0.001) (50).
Drug-eluting microsphere transcatheter arterial chemoembolization is a safe procedure for patients with HCC, with reports showing low rates of serious complications (1.6–7.2%) (2051). These complications included hepatic insufficiency or infarction, liver abscess, tumor rupture, bile duct injury, cholecystitis, upper gastrointestinal bleeding, pleural effusion, pulmonary embolism, splenic infarction, and spinal embolization. In contrast, minor procedural complications are more frequent, most being associated with the post-embolic syndrome. These complications are observed in 30.2–67.6% of patients and include fever, nausea and vomiting, and abdominal pain (2052). In addition, the incidence of bile duct injury has been reported to be higher in patients with HCC treated with DEMTACE than in those treated with c-TACE (53). This biliary complication may be due to stasis of the microspheres when there is complete embolization of the feeding artery (20).
Although chemotherapeutic drugs are typically accompanied by systemic complications, including fatigue, fever, gastrointestinal symptoms, neurological injury, pulmonary edema, bone marrow suppression, pulmonary edema, and cardiomyopathy, these are rarely observed in patients treated with DEM-TACE (2026). Indeed, it seems that the characteristic pharmacokinetics of DEM-TACE result in much lower systemic concentrations of doxorubicin compared with c-TACE (13567).
Notably, according to recently published studies, the 30-day mortality rates in patients treated with DEM-TACE ranged from 0% to 1.2% (5455). Whereas the median overall survival periods were 43.8–54 months, the 1-, 2-, 3-, 4-, and 5-year survival rates were 88.2–93.6%, 83.8%, 62.0–67.8%, 41.04–54.2%, and 33.9–39.4%, respectively (5456575859).
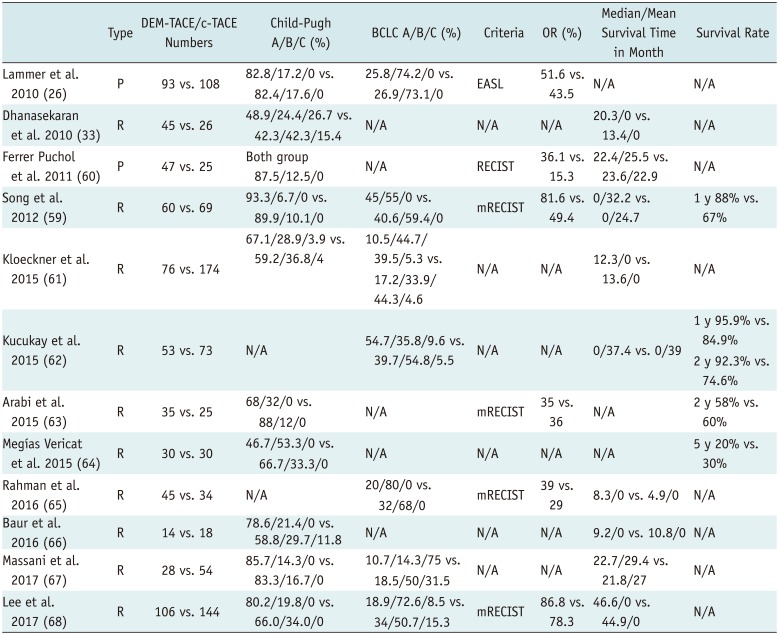
Since DEB-TACE first became commercially available in 2006, many clinical trials have shown its efficacy in patients with HCC and its ability to overcome the drawbacks of c-TACE (Table 6) (263359606162636465666768). DEB-TACE has, therefore, been used in some institutions as a first-line treatment in addition to c-TACE for the treatment of HCC in patients who are ineligible for curative treatment.
In the original research, DEB-TACE produced major tumor necrosis while reducing the side effects of chemotherapy due to the reduced passage of chemotherapeutic agents into the systemic circulation. Although good results were obtained with a mean doxorubicin dose of 128 mg (range, 47–150 mg), the systemic concentrations of doxorubicin were significantly lower in patients treated with DEB-TACE than in those treated with c-TACE (1). Similarly, a prospective single-blind, randomized phase II study of 30 patients with unresectable HCC treated with c-TACE or doxorubicin-eluting HepaSphere™ microspheres showed that the latter had a superior toxicity profile to c-TACE (12). The multicenter, prospective, randomized, single-blind, phase II PRECISION V trial of 212 patients reported objective response rates of 51.6% with DC Bead® and 43.5% with c-TACE (26). However, the overall rates of treatment-emergent complications and serious liver toxicity after embolization were lower among patients treated with DC Bead® than in those treated with c-TACE. These findings indicate that, although the two methods did not significantly differ in efficacy, DEB-TACE was better tolerated and allowed treatment to be repeated in these more vulnerable patients (26).
A retrospective study involving 71 patients reported that DEB-TACE provided a survival advantage over c-TACE, with subgroup analysis showing clear survival benefits in patients with Okuda stage I tumors, Child-Pugh classes A and B disease, and a Cancer of Liver Italian Program score of ≥ 3 (33). However, the safety profiles of the two modalities were similar (33). In another retrospective study involving 129 patients, DEB-TACE with DC Bead® resulted in superior treatment response and delayed tumor progression compared with c-TACE; however, there were no significant differences in treatment-related hepatic toxicities (59).
Liu et al. (69) also showed high safety and efficacy profiles for DEB-TACE compared with c-TACE. In a retrospective study involving 158 patients with HCC, 64 (40.5%) received TACE with lipiodol-doxorubicin and gelatin sponges (Group A), 41 (25.9%) received TACE with lipiodol-doxorubicin and microspheres (Group B), and 53 (33.5%) received TACE with doxorubicin-loaded DEBs (Group C). The study found that more patients in Group C (32.1%) achieved complete response than did those in either Group A (6.3%) or Group B (2.4%) (p < 0.001). Minor complications were also more common in Group A (54.7%) and Group B (34.1%) than in Group C (5.7%) (69).
In contrast to these data, a meta-analysis of seven studies (n = 693 patients) comparing the efficacy of DEB-TACE and c-TACE revealed that tumor response did not differ significantly among methods (70). However, it was conceded that additional large-scale, randomized, controlled studies are required (70). More recent meta-analyses have included multiple randomized controlled trials, prospective studies, and retrospective studies have also shown conflicting results. In a meta-analysis of seven clinical studies (n = 700 patients), Huang et al. (71) showed that tumor response was significantly better, and that the 1- and 2-year survival rates were significantly higher for DEB-TACE than for c-TACE. Xie et al. (72) also performed a meta-analysis of four randomized controlled trials, one uncontrolled prospective study, and one prospective case-control study (n = 652 patients). They reported similar overall survival rates between c-TACE and DEB-TACE, but that the latter was associated with a significantly higher objective response rate and a slightly lower incidence of complications (72). Chen et al. (73) performed a large meta-analysis of 1832 patients and found that DEB-TACE had higher 1-, 2-, and 3-year overall survival rates, and higher 1- and 2-year relapse-free survival rates. Finally, a meta-analysis involving 1449 patients found no significant differences in the 1-, 2-, and 3-year overall survival and complications rates between c-TACE and DEB-TACE (74).
A recent retrospective study included 82 non-surgical patients with HCC who underwent c-TACE or DEB-TACE and performed follow-up for a minimum of 12 months (67). In the DEB-TACE group, they used beads measuring 100–300 µm in diameter that were loaded with 50 mg of doxorubicin. Although the two methods showed similar efficacy, DEB-TACE was better tolerated, with survival mainly affected by preserved liver function (67). Another retrospective study comparing the clinical outcomes of DEB-TACE and c-TACE in 250 consecutive patients showed comparable treatment outcomes (68). Moreover, it not only failed to identify clinical factors that could help when choosing between these treatment modalities but also showed that tumor size was not associated with treatment outcomes in either group. However, because DEB-TACE showed a better safety profile when used to treat large carcinomas, it was recommended that its role should be further examined in patients with HCC at risk of aggravated liver function after treatment with c-TACE (68).
A retrospective comparison of survival outcomes was recently performed in 313 patients with unresectable HCC treated with two different TACE regimens: a c-TACE regimen with three drugs and a DEB-TACE regimen with a single drug (75). Interestingly, it showed no significant difference in survival between the two, though patients who underwent DEB-TACE had shorter hospital stays (75).
Together, these conflicting findings indicate that the comparative efficacy, safety, and survival rates of DEB-TACE and c-TACE remain unclear. However, DEB-TACE has consistently been shown to have superior pharmacokinetic proprieties and lower rates of systemic chemotherapy-associated toxicity, making it the more tolerable option when compared to c-TACE. Based on the current evidence, DEB-TACE may be the superior option when treating patients who are vulnerable or in whom we anticipate deterioration after locoregional chemotherapy.
Smaller caliber microspheres (< 100 µm) may provide better outcomes, with evidence that they show greater distal penetration and more effective embolization than larger microspheres, without influencing patient safety. An animal study of distal embolization showed that occlusion was more distal and that microsphere distribution in the embolized territory was denser when using 30–60-µm rather than 50–100-µm HepaSphere™ microspheres (76). Moreover, the tissue concentration of doxorubicin was higher after embolization with the smaller beads, while the plasma concentration remained very low (76). HepaSphere™ microspheres of dry caliber 30–60-µm are loadable and expand to 120–240 µm in saline and to 96–192 µm after loading with doxorubicin (76). Use of such microspheres in 45 patients who had intermediate stage HCC or who were ineligible for curative treatment produced a local objective response rate of 68.9% in patients who received an intended doxorubicin dosage of 50–100 mg, with an overall complete response rate of 17.8% (Fig. 6) (13). That study also showed very low complication rates, with no deaths or grade 5 complications at 30 days (13). In contrast, the use of larger HepaSphere™ microspheres resulted in lower objective and complete response rates (56% and 12.6%, respectively) (77). Moreover, the complication rates were lower than previously reported for periprocedural mortality (3%) and post-embolic syndrome (18.54%) with other DEMs (47).
An animal study found that beads ranging in diameter from 70 µm to 150 µm are associated with greater tumor coverage, more distal penetration, and higher doxorubicin concentrations in the tumor, without increasing systemic exposure, than is c-TACE (78). An evaluation of the efficacy of 70–150-µm DEBs (M1 DEB) in 45 patients with HCC who were undergoing TACE as either a primary therapy or as a bridge to liver transplantation reported an objective response rate of 77.7% and a median time to best response of 3 months (95% confidence interval: 2–4 months) (Fig. 7) (79). In 13 patients, DEB-TACE served as an effective bridge, downstaging the tumor to make it eligible for liver transplantation or surgery. Pathological examination also revealed more than 90% necrosis in 10 of the 28 nodules. The most common mild complications (grade 1 or 2) were post-embolic syndrome and abdominal pain, each of which occurred in 4.4% of patients. The only serious complication (grade 3) was bleeding from esophageal varices, which was observed in a patient after portal hypertension worsened; however, this patient was treated conservatively and recovered fully within 1 month (79).
A recent retrospective study involving 421 patients with HCC who underwent DEB-TACE using 70–150 µm DC Bead® (M1) showed that the overall objective response rates were 94.5% and 99.5% at 3 and 6 months, respectively (55). These rates were higher than those in a previous multicenter randomized controlled trial (26) using DEB-TACE particles of 300–500 µm and 500–700 µm in diameter, where the objective response rate was 52% at 6 months. No patient experienced treatment-related bleeding, pulmonary complications, or death (55). However, embolization with larger particles has been associated with a 30-day mortality rate of 1.2% (2 of 173 patients died due to procedure-related liver abscess or sepsis) (54) and 10 major complications among 104 patients (3 abscesses and 1 death) (56).
A successful delivery of loaded microspheres to tumors, while minimizing complications, requires super-selection of the feeding artery to avoid damage of non-targeted arteries. Several recently developed techniques have sought to achieve this goal.
In one report, detachable microcoils were used to divert blood flow temporarily in a patient who was undergoing DEB-TACE for a recurrent HCC in segments 8 and 4 after previously undergoing left hepatectomy (80). The recurrent tumor had a small feeding artery arising at an acute angle from the proximal right hepatic artery. Two microcatheters were inserted: the first in the right hepatic artery distal to the opening of the branch vessel supplying the residual tumor and the second in the proximal right hepatic artery, proximal to the ostium of the feeding artery. A 5-mm detachable coil was subsequently loaded through the distal catheter and deployed in the right hepatic artery, followed by the infusion of DC Bead® (80).
Similarly, a double-lumen microballoon catheter with a side hole at the leading end of the balloon was used to infuse chemotherapy during c-TACE in the treatment of HCC, although without super-selective catheterization of the target artery (81). This technique enabled prompt infusion of chemotherapeutic agents into the target artery via distal occlusion of the parent artery, with the distal occlusion ensuring the preservation of a broad area of normal hepatocytes.
Mixing nonionic contrast with loaded beads is essential for direct fluoroscopic monitoring and is typically achieved using drug-eluting radio-opaque beads (8283). LC Bead LUMI™ microspheres are sulfonate-modified polyvinyl alcohol hydrogel micro-beads that measure 70–150 µm in diameter and contain covalently bound iodine (83). Their radio-opacity allows for the real-time assessment of their deposition in the target tumor. Theoretically, the density and distribution of the radio-opaque beads can provide additional information to refine the embolization endpoint and the degree of flow stasis. This allows a better prospective visualization of non-target reflux and small branch feeders. Moreover, contrast retained by DEBs in dense vessels is typically washed out within minutes of the procedures, ensuring a lack of peripheral contrast retention, which has been reported to be a risk factor for treatment failure (84). Performing non-enhanced CT scan immediately to assess contrast retention after embolization may provide important information about treatment (85). The visibility of radio-opaque beads may also provide additional information about the expected effects of treatment, follow-up, and planning for the next session, as required.
Most patients with HCC are diagnosed with intermediate stage tumors, making curative treatment unavailable. Although c-TACE has been considered the treatment of choice for these patients, it has several limitations. DEM-TACE, was therefore, developed to overcome the drawbacks typically associated with c-TACE. Although studies comparing the safety and effectiveness of these methods have failed to show statistically significant superiority with the use of DEM-TACE, this method is considered more tolerable because it eliminates the toxicity associated with systemic chemotherapy. Further improvements in embolization techniques and microsphere types, including a better delineation of the optimal size, are needed to improve the efficacy of DEM-TACE and to widen its applications.
References
1. Varela M, Real MI, Burrel M, Forner A, Sala M, Brunet M, et al. Chemoembolization of hepatocellular carcinoma with drug eluting beads: efficacy and doxorubicin pharmacokinetics. J Hepatol. 2007; 46:474–481. PMID: 17239480.

2. Forner A, Reig ME, de Lope CR, Bruix J. Current strategy for staging and treatment: the BCLC update and future prospects. Semin Liver Dis. 2010; 30:61–74. PMID: 20175034.

3. Sottani C, Poggi G, Quaretti P, Regazzi M, Montagna B, Quaquarini E, et al. Serum pharmacokinetics in patients treated with transarterial chemoembolization (TACE) using two types of epirubicin-loaded microspheres. Anticancer Res. 2012; 32:1769–1774. PMID: 22593459.
4. Jiaqi Y, Hori S, Minamitani K, Hashimoto T, Yoshimura H, Nomura N, et al. [A new embolic material: super absorbent polymer (SAP) microsphere and its embolic effects]. Nihon Igaku Hoshasen Gakkai Zasshi. 1996; 56:19–24. PMID: 8857094.
5. de Baere T, Plotkin S, Yu R, Sutter A, Wu Y, Cruise GM. An in vitro evaluation of four types of drug-eluting microspheres loaded with doxorubicin. J Vasc Interv Radiol. 2016; 27:1425–1431. PMID: 27402527.

6. Pereira PL, Plotkin S, Yu R, Sutter A, Wu Y, Sommer CM, et al. An in-vitro evaluation of three types of drug-eluting microspheres loaded with irinotecan. Anticancer Drugs. 2016; 27:873–878. PMID: 27416270.

7. Lewis AL, Gonzalez MV, Lloyd AW, Hall B, Tang Y, Willis SL, et al. DC bead: in vitro characterization of a drug-delivery device for transarterial chemoembolization. J Vasc Interv Radiol. 2006; 17(2 Pt 1):335–342. PMID: 16517780.

8. Namur J, Wassef M, Millot JM, Lewis AL, Manfait M, Laurent A. Drug-eluting beads for liver embolization: concentration of doxorubicin in tissue and in beads in a pig model. J Vasc Interv Radiol. 2010; 21:259–267. PMID: 20123210.

9. Hong K, Khwaja A, Liapi E, Torbenson MS, Georgiades CS, Geschwind JF. New intra-arterial drug delivery system for the treatment of liver cancer: preclinical assessment in a rabbit model of liver cancer. Clin Cancer Res. 2006; 12:2563–2567. PMID: 16638866.

10. Namur J, Citron SJ, Sellers MT, Dupuis MH, Wassef M, Manfait M, et al. Embolization of hepatocellular carcinoma with drug-eluting beads: doxorubicin tissue concentration and distribution in patient liver explants. J Hepatol. 2011; 55:1332–1338. PMID: 21703190.

11. Varela M, Sala M, Llovet JM, Bruix J. Treatment of hepatocellular carcinoma: is there an optimal strategy? Cancer Treat Rev. 2003; 29:99–104. PMID: 12670452.

12. van Malenstein H, Maleux G, Vandecaveye V, Heye S, Laleman W, van Pelt J, et al. A randomized phase II study of drugeluting beads versus transarterial chemoembolization for unresectable hepatocellular carcinoma. Onkologie. 2011; 34:368–376. PMID: 21734423.

13. Malagari K, Pomoni M, Moschouris H, Kelekis A, Charokopakis A, Bouma E, et al. Chemoembolization of hepatocellular carcinoma with HepaSphere 30-60 μm. Safety and efficacy study. Cardiovasc Intervent Radiol. 2014; 37:165–117. PMID: 24263774.
14. Emerich DF, Snodgrass P, Lafreniere D, Dean RL, Salzberg H, Marsh J, et al. Sustained release chemotherapeutic microspheres provide superior efficacy over systemic therapy and local bolus infusions. Pharm Res. 2002; 19:1052–1060. PMID: 12180539.
15. Lewis AL, Dreher MR, O'Byrne V, Grey D, Caine M, Dunn A, et al. DC BeadM1™: towards an optimal transcatheter hepatic tumour therapy. J Mater Sci Mater Med. 2016; 27:13. PMID: 26676859.

16. Lee S, Kim KM, Lee SJ, Lee KH, Lee DY, Kim MD, et al. Hepatic arterial damage after transarterial chemoembolization for the treatment of hepatocellular carcinoma: comparison of drug-eluting bead and conventional chemoembolization in a retrospective controlled study. Acta Radiol. 2017; 58:131–139. PMID: 27217418.

17. Kobayashi N, Ishii M, Ueno Y, Kisara N, Chida N, Iwasaki T, et al. Co-expression of Bcl-2 protein and vascular endothelial growth factor in hepatocellular carcinomas treated by chemoembolization. Liver. 1999; 19:25–31. PMID: 9928762.

18. Wang B, Xu H, Gao ZQ, Ning HF, Sun YQ, Cao GW. Increased expression of vascular endothelial growth factor in hepatocellular carcinoma after transcatheter arterial chemoembolization. Acta Radiol. 2008; 49:523–529. PMID: 18568538.

19. Lee KH, Liapi E, Vossen JA, Buijs M, Ventura VP, Georgiades C, et al. Distribution of iron oxide-containing Embosphere particles after transcatheter arterial embolization in an animal model of liver cancer: evaluation with MR imaging and implication for therapy. J Vasc Interv Radiol. 2008; 19:1490–1496. PMID: 18755602.

20. Lee M, Chung JW, Lee KH, Won JY, Chun HJ, Lee HC, et al. Korean multicenter registry of transcatheter arterial chemoembolization with drug-eluting embolic agents for nodular hepatocellular carcinomas: six-month outcome analysis. J Vasc Interv Radiol. 2017; 28:502–512. PMID: 27856136.

21. Lee JH, Won JH, Park SI, Won JY, Lee DY, Kang BC. Transcatheter arterial chemoembolization of hepatocellular carcinoma with hepatic arteriovenous shunt after temporary balloon occlusion of hepatic vein. J Vasc Interv Radiol. 2007; 18:377–382. PMID: 17377183.

22. European Association for Study of Liver. European Organisation for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. Eur J Cancer. 2012; 48:599–641. PMID: 22424278.
23. Bruix J, Llovet JM. Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology. 2002; 35:519–524. PMID: 11870363.

24. Kim JH, Shim JH, Lee HC, Sung KB, Ko HK, Ko GY, et al. New intermediate-stage subclassification for patients with hepatocellular carcinoma treated with transarterial chemoembolization. Liver Int. 2017; 37:1861–1868. PMID: 28581250.

25. Han K, Kim JH. Transarterial chemoembolization in hepatocellular carcinoma treatment: Barcelona clinic liver cancer staging system. World J Gastroenterol. 2015; 21:10327–10335. PMID: 26420959.

26. Lammer J, Malagari K, Vogl T, Pilleul F, Denys A, Watkinson A, et al. ; PRECISION V Investigators. Prospective randomized study of doxorubicin-eluting-bead embolization in the treatment of hepatocellular carcinoma: results of the PRECISION V study. Cardiovasc Intervent Radiol. 2010; 33:41–45. PMID: 19908093.
27. Hoóówko W, Wróblewski T, Wojtaszek M, Grąt M, Kobryń K, Ziarkiewicz-Wróblewska B, et al. Transarterial chemoembolization prior to liver transplantation in patients with hepatocellular carcinoma. Ann Transplant. 2015; 20:764–768. PMID: 26712800.

28. Yu CY, Ou HY, Weng CC, Huang TL, Chen TY, Leung-Chit L, et al. Drug-eluting bead transarterial chemoembolization as bridge therapy for hepatocellular carcinoma before living-donor liver transplantation. Transplant Proc. 2016; 48:1045–1048. PMID: 27320552.

29. Clavien PA, Lesurtel M, Bossuyt PM, Gores GJ, Langer B, Perrier A. OLT for HCC Consensus Group. Recommendations for liver transplantation for hepatocellular carcinoma: an international consensus conference report. Lancet Oncol. 2012; 13:e11–e22. PMID: 22047762.

31. Sellers MT, Huggins S, Kegley K, Pollinger HS, Shrestha R, Johnson MW, et al. Multivariate analysis of prognostic factors for survival following doxorubicin-eluting bead transarterial chemoembolization for hepatocellular carcinoma. J Vasc Interv Radiol. 2013; 24:647–654. PMID: 23384831.

32. Lladó L, Virgili J, Figueras J, Valls C, Dominguez J, Rafecas A, et al. A prognostic index of the survival of patients with unresectable hepatocellular carcinoma after transcatheter arterial chemoembolization. Cancer. 2000; 88:50–57. PMID: 10618605.

33. Dhanasekaran R, Kooby DA, Staley CA, Kauh JS, Khanna V, Kim HS. Prognostic factors for survival in patients with unresectable hepatocellular carcinoma undergoing chemoembolization with doxorubicin drug-eluting beads: a preliminary study. HPB (Oxford). 2010; 12:174–118. PMID: 20590884.

34. Fan J, Wu ZQ, Tang ZY, Zhou J, Qiu SJ, Ma ZC, et al. Multimodality treatment in hepatocellular carcinoma patients with tumor thrombi in portal vein. World J Gastroenterol. 2001; 7:28–32. PMID: 11819728.

35. Kim JH, Shim JH, Yoon HK, Ko HK, Kim JW, Gwon DI. Chemoembolization related to good survival for selected patients with hepatocellular carcinoma invading segmental portal vein. Liver Int. 2018; 38:1646–1654. PMID: 29436101.

36. Lencioni R, de Baere T, Burrel M, Caridi JG, Lammer J, Malagari K, et al. Transcatheter treatment of hepatocellular carcinoma with doxorubicin-loaded DC Bead (DEBDOX): technical recommendations. Cardiovasc Intervent Radiol. 2012; 35:980–985. PMID: 22009576.

37. Kim HC. Role of C-arm cone-beam CT in chemoembolization for hepatocellular carcinoma. Korean J Radiol. 2015; 16:114–124. PMID: 25598679.

38. Tacher V, Radaelli A, Lin M, Geschwind JF. How I do it: cone-beam CT during transarterial chemoembolization for liver cancer. Radiology. 2015; 274:320–334. PMID: 25625741.

39. Choi JW, Kim HC, Lee JH, Yu SJ, Cho EJ, Kim MU, et al. Cone beam CT-guided chemoembolization of probable hepatocellular carcinomas smaller than 1 cm in patients at high risk of hepatocellular carcinoma. J Vasc Interv Radiol. 2017; 28:795–803.e1. PMID: 28302348.

40. Wang J, He XD, Zhang YC. Antibiotic prophylaxis in transarterial therapy of hepatocellular carcinoma: a meta-analysis. Can J Gastroenterol. 2012; 26:85–91. PMID: 22312607.

41. Lv WF, Lu D, He YS, Xiao JK, Zhou CZ, Cheng DL. Liver abscess formation following transarterial chemoembolization: clinical features, risk factors, bacteria spectrum, and percutaneous catheter drainage. Medicine (Baltimore). 2016; 95:e350.
42. Woo S, Chung JW, Hur S, Joo SM, Kim HC, Jae HJ, et al. Liver abscess after transarterial chemoembolization in patients with bilioenteric anastomosis: frequency and risk factors. AJR Am J Roentgenol. 2013; 200:1370–1377. PMID: 23701078.

43. Ji SK, Cho YK, Ahn YS, Kim MY, Park YO, Kim JK, et al. Multivariate analysis of the predictors of survival for patients with hepatocellular carcinoma undergoing transarterial chemoembolization: focusing on superselective chemoembolization. Korean J Radiol. 2008; 9:534–540. PMID: 19039270.
44. Lewis AL, Taylor RR, Hall B, Gonzalez MV, Willis SL, Stratford PW. Pharmacokinetic and safety study of doxorubicin-eluting beads in a porcine model of hepatic arterial embolization. J Vasc Interv Radiol. 2006; 17:1335–1343. PMID: 16923981.

45. Kim YW, Kwon JH, Nam SW, Jang JW, Jung HS, Shin YR, et al. Sustained multiple organ ischaemia after transarterial chemoembolization with drug-eluting beads for hepatocellular carcinoma. Exp Ther Med. 2018; 15:1479–1483. PMID: 29434732.

46. Poon RT, Tso WK, Pang RW, Ng KK, Woo R, Tai KS, et al. A phase I/II trial of chemoembolization for hepatocellular carcinoma using a novel intra-arterial drug-eluting bead. Clin Gastroenterol Hepatol. 2007; 5:1100–1108. PMID: 17627902.

47. Grosso M, Vignali C, Quaretti P, Nicolini A, Melchiorre F, Gallarato G, et al. Transarterial chemoembolization for hepatocellular carcinoma with drug-eluting microspheres: preliminary results from an Italian multicentre study. Cardiovasc Intervent Radiol. 2008; 31:1141–1149. PMID: 18696150.

48. Kalva SP, Iqbal SI, Yeddula K, Blaszkowsky LS, Akbar A, Wicky S, et al. Transarterial chemoembolization with doxorubicin-eluting microspheres for inoperable hepatocellular carcinoma. Gastrointest Cancer Res. 2011; 4:2–8. PMID: 21464864.
49. Manini MA, Sangiovanni A, Martinetti L, Viganò D, La Mura V, Aghemo A, et al. Transarterial chemoembolization with drug-eluting beads is effective for the maintenance of the Milan-in status in patients with a small hepatocellular carcinoma. Liver Transpl. 2015; 21:1259–1269. PMID: 26074360.

50. Sandow TA, Arndt SE, Albar AA, DeVun DA, Kirsch DS, Gimenez JM, et al. Assessment of response to transcatheter arterial chemoembolization with doxorubicin-eluting microspheres: tumor biology and hepatocellular carcinoma recurrence in a 5-year transplant cohort. Radiology. 2018; 286:1072–1083. PMID: 29206595.

51. Popovic P, Stabuc B, Jansa R, Garbajs M. Survival of patients with intermediate stage hepatocellular carcinoma treated with superselective transarterial chemoembolization using doxorubicin-loaded DC Bead under cone-beam computed tomography control. Radiol Oncol. 2016; 50:418–426. PMID: 27904450.

52. Prajapati HJ, Dhanasekaran R, El-Rayes BF, Kauh JS, Maithel SK, Chen Z, et al. Safety and efficacy of doxorubicin drug-eluting bead transarterial chemoembolization in patients with advanced hepatocellular carcinoma. J Vasc Interv Radiol. 2013; 24:307–315. PMID: 23375519.

53. Guiu B, Deschamps F, Aho S, Munck F, Dromain C, Boige V, et al. Liver/biliary injuries following chemoembolisation of endocrine tumours and hepatocellular carcinoma: lipiodol vs. drug-eluting beads. J Hepatol. 2012; 56:609–661. PMID: 22027582.

54. Malagari K, Pomoni M, Moschouris H, Bouma E, Koskinas J, Stefaniotou A, et al. Chemoembolization with doxorubicin-eluting beads for unresectable hepatocellular carcinoma: five-year survival analysis. Cardiovasc Intervent Radiol. 2012; 35:1119–1128. PMID: 22614031.

55. Aliberti C, Carandina R, Lonardi S, Dadduzio V, Vitale A, Gringeri E, et al. Transarterial chemoembolization with small drug-eluting beads in patients with hepatocellular carcinoma: experience from a cohort of 421 patients at an Italian center. J Vasc Interv Radiol. 2017; 28:1495–1502. PMID: 28927662.

56. Burrel M, Reig M, Forner A, Barrufet M, de Lope CR, Tremosini S, et al. Survival of patients with hepatocellular carcinoma treated by transarterial chemoembolisation (TACE) using drug eluting beads. Implications for clinical practice and trial design. J Hepatol. 2012; 56:1330–1133. PMID: 22314428.

57. Luz JH, Luz PM, Martin HS, Gouveia HR, Levigard RB, Nogueira FD, et al. DEB TACE for intermediate and advanced HCC - Initial experience in a Brazilian Cancer Center. Cancer Imaging. 2017; 17:5. PMID: 28166821.

58. Kalva SP, Pectasides M, Liu R, Rachamreddy N, Surakanti S, Yeddula K, et al. Safety and effectiveness of chemoembolization with drug-eluting beads for advanced-stage hepatocellular carcinoma. Cardiovasc Intervent Radiol. 2014; 37:381–387. PMID: 23754191.

59. Song MJ, Chun HJ, Song DS, Kim HY, Yoo SH, Park CH, et al. Comparative study between doxorubicin-eluting beads and conventional transarterial chemoembolization for treatment of hepatocellular carcinoma. J Hepatol. 2012; 57:1244–1250. PMID: 22824821.

60. Ferrer Puchol MD, la Parra C, Esteban E, Vaño M, Forment M, Vera A, et al. [Comparison of doxorubicin-eluting bead transarterial chemoembolization (DEB-TACE) with conventional transarterial chemoembolization (TACE) for the treatment of hepatocellular carcinoma]. Radiologia. 2011; 53:246–253. PMID: 21295802.

61. Kloeckner R, Weinmann A, Prinz F, Pinto dos Santos D, Ruckes C, Dueber C, et al. Conventional transarterial chemoembolization versus drug-eluting bead transarterial chemoembolization for the treatment of hepatocellular carcinoma. BMC Cancer. 2015; 15:465. PMID: 26059447.

62. Kucukay F, Badem S, Karan A, Ozdemir M, Okten RS, Ozbulbul NI, et al. A single-center retrospective comparison of doxorubicin-loaded HepaSphere transarterial chemoembolization with conventional transarterial chemoembolization for patients with unresectable hepatocellular carcinoma. J Vasc Interv Radiol. 2015; 26:1622–1629. PMID: 26321015.

63. Arabi M, BenMousa A, Bzeizi K, Garad F, Ahmed I, Al-Otaibi M. Doxorubicin-loaded drug-eluting beads versus conventional transarterial chemoembolization for nonresectable hepatocellular carcinoma. Saudi J Gastroenterol. 2015; 21:175–180. PMID: 26021777.

64. Megías Vericat JE, García Marcos R, López Briz E, Gómez Muñoz F, Ramos Ruiz J, Martínez Rodrigo JJ, et al. Trans-arterial chemoembolization with doxorubicin-eluting particles versus conventional trans-arterial chemoembolization in unresectable hepatocellular carcinoma: a study of effectiveness, safety and costs. Radiologia. 2015; 57:496–504. PMID: 25857250.

65. Rahman FA, Naidu J, Ngiu CS, Yaakob Y, Mohamed Z, Othman H, et al. Conventional versus doxorubicin-eluting beads transarterial chemoembolization for unresectable hepatocellular carcinoma: a tertiary medical centre experience in Malaysia. Asian Pac J Cancer Prev. 2016; 17:4037–4041. PMID: 27644658.
66. Baur J, Ritter CO, Germer CT, Klein I, Kickuth R, Steger U. Transarterial chemoembolization with drug-eluting beads versus conventional transarterial chemoembolization in locally advanced hepatocellular carcinoma. Hepat Med. 2016; 8:69–74. PMID: 27382341.

67. Massani M, Stecca T, Ruffolo C, Bassi N. Should we routinely use DEBTACE for unresectable HCC? cTACE versus DEBTACE: a single-center survival analysis. Updates Surg. 2017; 69:67–73. PMID: 28097502.

68. Lee YK, Jung KS, Kim DY, Choi JY, Kim BK, Kim SU, et al. Conventional versus drug-eluting beads chemoembolization for hepatocellular carcinoma: emphasis on the impact of tumor size. J Gastroenterol Hepatol. 2017; 32:487–496. PMID: 27503585.

69. Liu YS, Ou MC, Tsai YS, Lin XZ, Wang CK, Tsai HM, et al. Transarterial chemoembolization using gelatin sponges or microspheres plus lipiodol-doxorubicin versus doxorubicin-loaded beads for the treatment of hepatocellular carcinoma. Korean J Radiol. 2015; 16:125–132. PMID: 25598680.

70. Gao S, Yang Z, Zheng Z, Yao J, Deng M, Xie H, et al. Doxorubicin-eluting bead versus conventional TACE for unresectable hepatocellular carcinoma: a meta-analysis. Hepatogastroenterology. 2013; 60:813–820. PMID: 23282741.
71. Huang K, Zhou Q, Wang R, Cheng D, Ma Y. Doxorubicin-eluting beads versus conventional transarterial chemoembolization for the treatment of hepatocellular carcinoma. J Gastroenterol Hepatol. 2014; 29:920–925. PMID: 24224722.

72. Xie ZB, Wang XB, Peng YC, Zhu SL, Ma L, Xiang BD, et al. Systematic review comparing the safety and efficacy of conventional and drug-eluting bead transarterial chemoembolization for inoperable hepatocellular carcinoma. Hepatol Res. 2015; 45:190–200. PMID: 25388603.

73. Chen P, Yuan P, Chen B, Sun J, Shen H, Qian Y. Evaluation of drug-eluting beads versus conventional transcatheter arterial chemoembolization in patients with unresectable hepatocellular carcinoma: a systematic review and meta-analysis. Clin Res Hepatol Gastroenterol. 2017; 41:75–85. PMID: 27350573.

74. Facciorusso A, Di Maso M, Muscatiello N. Drug-eluting beads versus conventional chemoembolization for the treatment of unresectable hepatocellular carcinoma: a meta-analysis. Dig Liver Dis. 2016; 48:571–577. PMID: 26965785.

75. Gomes AS, Monteleone PA, Sayre JW, Finn RS, Sadeghi S, Tong MJ, et al. Comparison of triple-drug transcatheter arterial chemoembolization (TACE) with single-drug TACE using doxorubicin-eluting beads: long-term survival in 313 patients. AJR Am J Roentgenol. 2017; 209:722–732. PMID: 28705059.

76. Dinca H, Pelage JP, Baylatry MT, Ghegediban SH, Pascale F, Manfait M, et al. Why do small size doxorubicin-eluting microspheres induce more tissue necrosis than larger ones? A comparative study in healthy pig liver (oral communication 2206-2). In : CIRSE Annual meeting; 2012 September, 15-19; Lisbon, Portugal.
77. Seki A, Hori S, Kobayashi K, Narumiya S. Transcatheter arterial chemoembolization with epirubicin-loaded superabsorbent polymer microspheres for 135 hepatocellular carcinoma patients: single-center experience. Cardiovasc Intervent Radiol. 2011; 34:557–565. PMID: 20821211.

78. Dreher MR, Sharma KV, Woods DL, Reddy G, Tang Y, Pritchard WF, et al. Radiopaque drug-eluting beads for transcatheter embolotherapy: experimental study of drug penetration and coverage in swine. J Vasc Interv Radiol. 2012; 23:257–264.e4. PMID: 22178039.

79. Spreafico C, Cascella T, Facciorusso A, Sposito C, Rodolfo L, Morosi C, et al. Transarterial chemoembolization for hepatocellular carcinoma with a new generation of beads: clinical-radiological outcomes and safety profile. Cardiovasc Intervent Radiol. 2015; 38:129–134. PMID: 24870698.

80. Pua U. “Deploy and retrieve” technique using detachable micro-coil for temporary occlusion during drug-eluting beads chemoembolization. Cardiovasc Intervent Radiol. 2015; 38:1359–1362. PMID: 25672285.

81. Todoroki W, Hirakawa M, Nagao E, Soeda H, Tsuruta S, Honda H. Transarterial chemoembolization for hepatocellular carcinoma using a new double-lumen microballoon catheter with a side hole. J Vasc Interv Radiol. 2014; 25:1485–1486. PMID: 25150909.

82. Levy EB, Krishnasamy VP, Lewis AL, Willis S, Macfarlane C, Anderson V, et al. First human experience with directly image-able iodinated embolization microbeads. Cardiovasc Intervent Radiol. 2016; 39:1177–1186. PMID: 27206503.

83. Aliberti C, Carandina R, Sarti D, Pizzirani E, Ramondo G, Cillo U, et al. Transarterial chemoembolization with DC Bead LUMI™ radiopaque beads for primary liver cancer treatment: preliminary experience. Future Oncol. 2017; 13:2243–2252. PMID: 29063780.

84. Suk Oh J, Jong Chun H, Gil Choi B, Giu Lee H. Transarterial chemoembolization with drug-eluting beads in hepatocellular carcinoma: usefulness of contrast saturation features on cone-beam computed tomography imaging for predicting short-term tumor response. J Vasc Interv Radiol. 2013; 24:483–489. PMID: 23452553.

85. Golowa YS, Cynamon J, Reinus JF, Kinkhabwala M, Abrams M, Jagust M, et al. Value of noncontrast CT immediately after transarterial chemoembolization of hepatocellular carcinoma with drug-eluting beads. J Vasc Interv Radiol. 2012; 23:1031–1035. PMID: 22739645.

Fig. 1
DC Bead® (BTG).
A. Bottle of DC Bead® (100–300 µm) before loading. B. Bottle of DC Bead® (100–300 µm) loaded with doxorubicin.

Fig. 2
Technique of HepaSphere™ (Merit Medical) TACE.
A. Hepatic angiography showing single tumor blush (arrow). B, C. Cone-beam CT angiography precisely delineating single tumor feeder (yellow line). D. HepaSphere™ (30–60 µm) loaded with doxorubicin is slowly injected. CT = computed tomography, TACE = transcatheter arterial chemoembolization
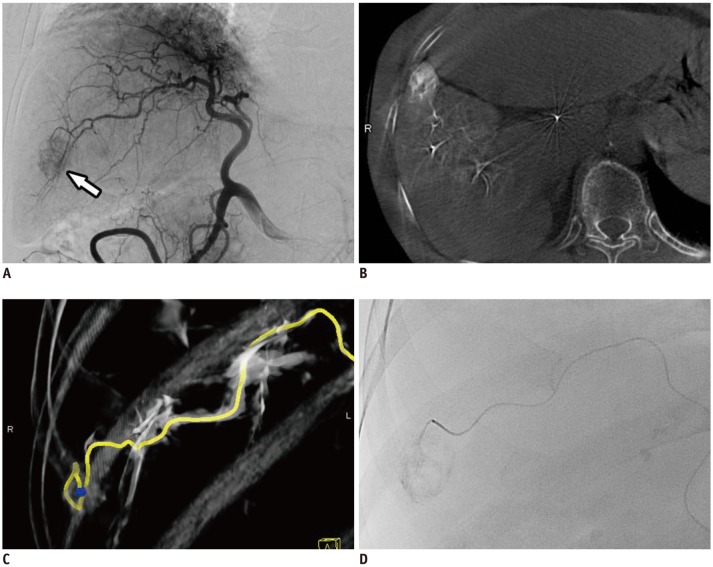
Fig. 3
Technique of DC Bead® (BTG) TACE.
A. Celiac angiography showing single small tumor blush and tumor feeder (arrow). B. After selection of tumor feeder using microcatheter, DC Bead® (100–300 µm) loaded with doxorubicin/nonionic contrast suspension are slowly injected, usually over 1 minute for 1 mL injection.
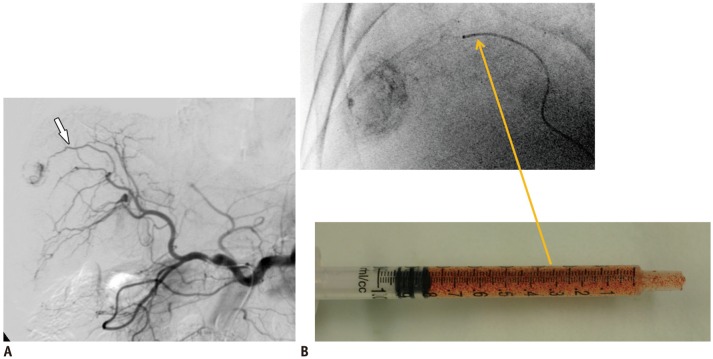
Fig. 4
70-year-old woman with single, intermediate-size HCC (4.3 cm in diameter).
A. Axial contrast-enhanced CT in arterial phase showing 4.3 cm HCC (arrow) in segment 6. B. Hepatic angiography showing single small tumor blush (arrow). C. Axial contrast-enhanced CT image one month after HepaSphere™ (Merit Medical) TACE demonstrating complete response. HCC = hepatocellular carcinoma
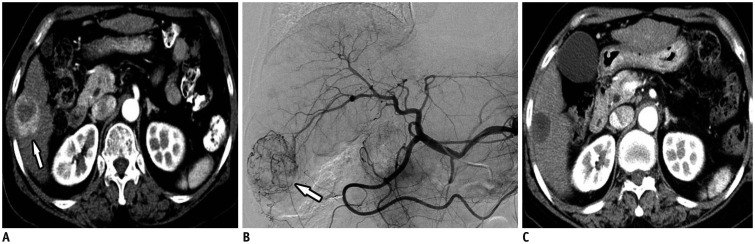
Fig. 5
DC Bead® (BTG) TACE as bridge to liver transplantation.
A. Axial contrast-enhanced CT image in arterial phase showing multiple HCCs (beyond Milan criteria) (arrows) in caudate lobe. B, C. Arteriography obtained at caudate branch (arrowhead) showing tumor blushes (arrows) in caudate lobe. D. Axial contrast-enhanced CT image one month after DC Bead® TACE showing complete response.
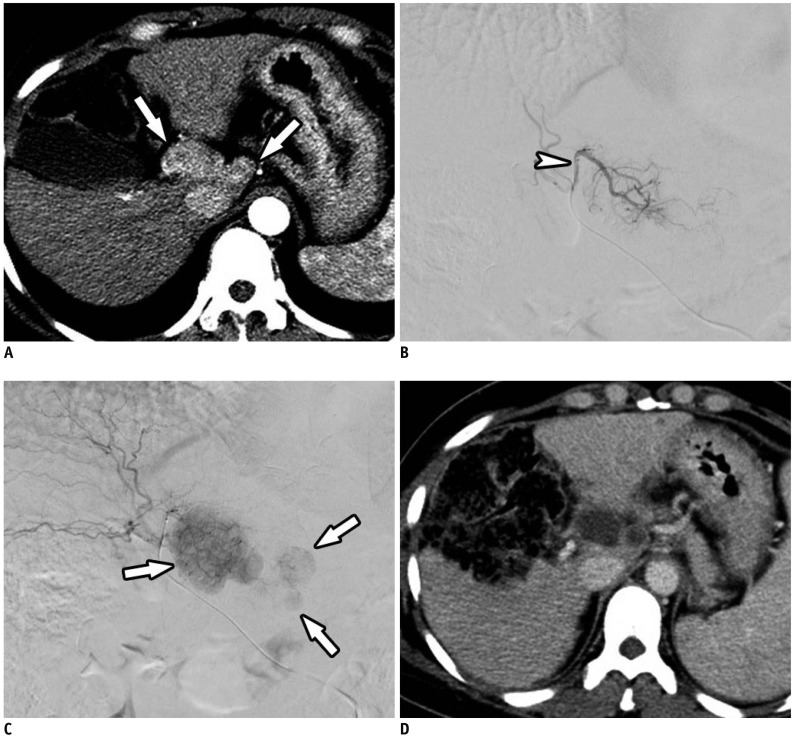
Fig. 6
Use of 30–60-µm HepaSphere™ (Merit Medical) TACE for Multinodular HCCs.
A. Axial contrast-enhanced CT image in arterial phase showing multiple HCCs (arrows) in right lobe of liver. B, C. Axial contrast-enhanced CT images in arterial phase after 1 and 6 months, respectively, showing complete response with no recurrence.
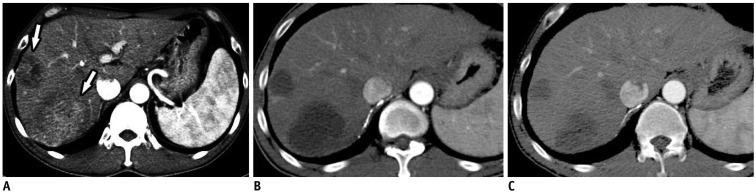
Fig. 7
Use of 70–150-µm DC Bead® (BTG) TACE for Multinodular HCCs.
A, B. Axial contrast-enhanced CT images in arterial phase showing multiple HCCs (arrows) in right lobe of liver. C, D. Axial contrast-enhanced CT images in arterial phase after 1 month showing only partial response.
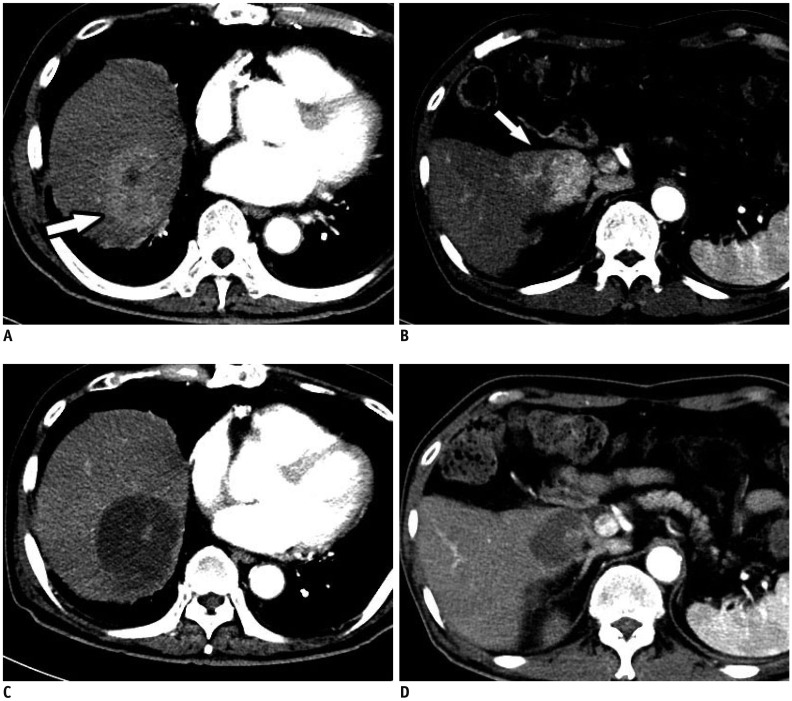
Table 1
Types and Sizes of Microspheres
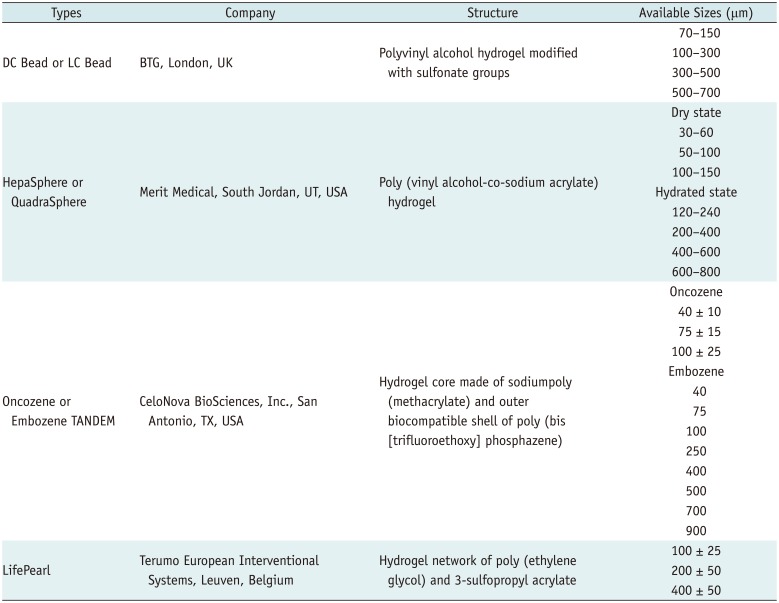
Table 2
Indications for DEM-TACE

Table 3
Contraindications for DEM-TACE
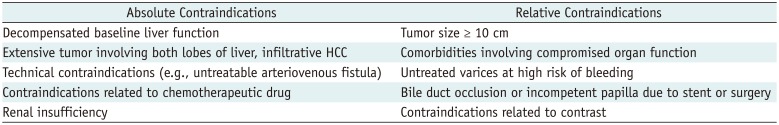
Table 4
Summary of Technique
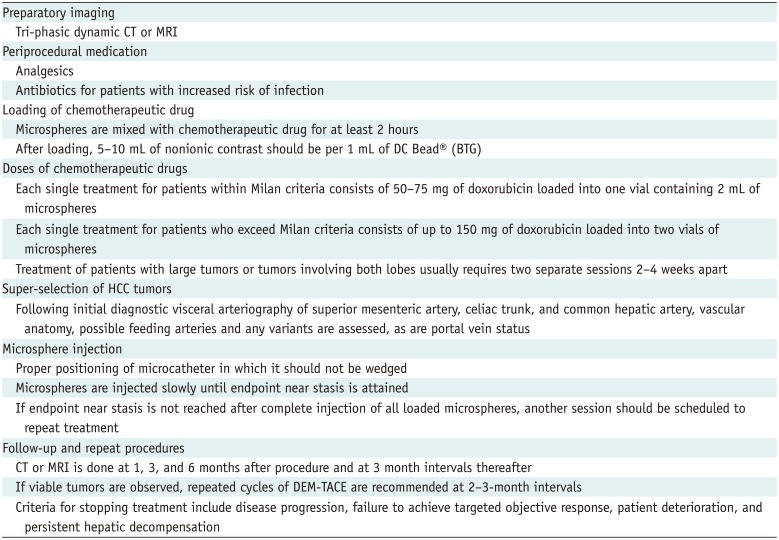
Table 5
Summary of Initial and Recent Clinical Study Showing Response Rate
| Type | Numbers | Child-Pugh | BCLC | Microsphere (µm) | Criteria | Response Rate | Region | |
|---|---|---|---|---|---|---|---|---|
| Varela et al. 2007 (1) | P | 27 | A | B | Beads | RECIST, EASL | 6 m: OR 44.4% RECIST OR 66.6% EASL | Spain |
| 500–700 | ||||||||
| Poon et al. 2007 (46) | P | 35 | A | A, B | Beads | RECIST, mRECIST | 1 m: OR 50% RECIST OR 70% mRECIST | China |
| 500–700 | 4 m: OR 35.7% RECIST OR 42.9% mRECIST | |||||||
| Grosso et al. 2008 (47) | P | 50 | A, B | N/A | Microspheres | EASL | 1 m: OR 84% | Italy |
| 50–100 | ||||||||
| Kalva et al. 2011 (48) | R | 54 | A, B, C | N/A | Beads | RECIST | 1 m: OR 14.8% | USA |
| 300–500 | ||||||||
| Manini et al. 2015 (49) | P | 55 | A, B | A | Beads | mRECIST | 1 m: OR 84% | USA |
| 100–300 | ||||||||
| Yu et al. 2016 (28) | R | 60 | N/A | N/A | Beads | mRECIST | 1m: OR 73.3% | Taiwan |
| 100–300 | ||||||||
| 300–500 | ||||||||
| Lee et al. 2017 (20) | P | 152 | A, B | A, B, C | Beads | mRECIST 300–500 | 1 m: OR 91% | Korea |
| 100–300 | 6 m: OR 55.4% | |||||||
| 300–500 | ||||||||
| Sandow et al. 2018 (50) | R | 93 | A, B, C | A | Beads | mRECIST | Initial 1 m: OR 76% | USA |
| 100–300 | Final (before transplant mean 179 days): OR 82% |
Table 6
Summary of Comparison Study between DEM-TACE vs. c-TACE
| Type | DEM-TACE/c-TACE Numbers | Child-Pugh A/B/C (%) | BCLC A/B/C (%) | Criteria | OR (%) | Median/Mean Survival Time in Month | Survival Rate | |
|---|---|---|---|---|---|---|---|---|
| Lammer et al. 2010 (26) | P | 93 vs. 108 | 82.8/17.2/0 vs. 82.4/17.6/0 | 25.8/74.2/0 vs. 26.9/73.1/0 | EASL | 51.6 vs. 43.5 | N/A | N/A |
| Dhanasekaran et al. 2010 (33) | R | 45 vs. 26 | 48.9/24.4/26.7 vs. 42.3/42.3/15.4 | N/A | N/A | N/A | 20.3/0 vs. 13.4/0 | N/A |
| Ferrer Puchol et al. 2011 (60) | P | 47 vs. 25 | Both group 87.5/12.5/0 | N/A | RECIST | 36.1 vs. 15.3 | 22.4/25.5 vs. 23.6/22.9 | N/A |
| Song et al. 2012 (59) | R | 60 vs. 69 | 93.3/6.7/0 vs. 89.9/10.1/0 | 45/55/0 vs. 40.6/59.4/0 | mRECIST | 81.6 vs. 49.4 | 0/32.2 vs. 0/24.7 | 1 y 88% vs. 67% |
| Kloeckner et al. 2015 (61) | R | 76 vs. 174 | 67.1/28.9/3.9 vs. 59.2/36.8/4 | 10.5/44.7/ 39.5/5.3 vs. 17.2/33.9/ 44.3/4.6 | N/A | N/A | 12.3/0 vs. 13.6/0 | N/A |
| Kucukay et al. 2015 (62) | R | 53 vs. 73 | N/A | 54.7/35.8/9.6 vs. 39.7/54.8/5.5 | N/A | N/A | 0/37.4 vs. 0/39 | 1 y 95.9% vs. 84.9% |
| 2 y 92.3% vs. 74.6% | ||||||||
| Arabi et al. 2015 (63) | R | 35 vs. 25 | 68/32/0 vs. 88/12/0 | N/A | mRECIST | 35 vs. 36 | N/A | 2 y 58% vs. 60% |
| Megías Vericat et al. 2015 (64) | R | 30 vs. 30 | 46.7/53.3/0 vs. 66.7/33.3/0 | N/A | N/A | N/A | N/A | 5 y 20% vs. 30% |
| Rahman et al. 2016 (65) | R | 45 vs. 34 | N/A | 20/80/0 vs. 32/68/0 | mRECIST | 39 vs. 29 | 8.3/0 vs. 4.9/0 | N/A |
| Baur et al. 2016 (66) | R | 14 vs. 18 | 78.6/21.4/0 vs. 58.8/29.7/11.8 | N/A | N/A | N/A | 9.2/0 vs. 10.8/0 | N/A |
| Massani et al. 2017 (67) | R | 28 vs. 54 | 85.7/14.3/0 vs. 83.3/16.7/0 | 10.7/14.3/75 vs. 18.5/50/31.5 | N/A | N/A | 22.7/29.4 vs. 21.8/27 | N/A |
| Lee et al. 2017 (68) | R | 106 vs. 144 | 80.2/19.8/0 vs. 66.0/34.0/0 | 18.9/72.6/8.5 vs. 34/50.7/15.3 | mRECIST | 86.8 vs. 78.3 | 46.6/0 vs. 44.9/0 | N/A |




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download