Abstract
Objectives
Although biofilms have been implicated in poor prognosis after endoscopic sinus surgery (ESS), traditional methods detecting biofilm such as scanning electron microscope and confocal scanning laser microscope were rarely used in the practice. The aims of this study was to determine whether the presence or absence of a biofilm detected by hematoxylin and eosin (H&E) staining followed by light microscopy (LM) that is widely used in daily practice, predicts surgical outcomes after ESS.
Methods
Retrospective analysis of prospectively collected data. Fifty-five consecutive adult patients (>18 years) who underwent ESS for chronic rhinosinusitis with a minimum of 12-months of follow-up were enrolled in this study. Random sinonasal mucosal samples were assessed for biofilm presence using H&E staining with LM. Three independent observers scored whether a biofilm was present or absent based on H&E staining/LM, and the interrater variability was calculated. Pre- and postoperative sinus symptoms and sinonasal mucosal grading were assessed.
Results
Biofilms were present in 28 patients (51%), and the intraclass correlation coefficient according to H&E staining/LM was 0.731. The presence of a biofilm was associated with a higher preoperative Lund-MacKay computed tomography score (22.3 for biofilm-positive patients vs. 18.6 for biofilm-negative patients; P=0.021) and persistent inflammation (mucosal edema and discharge) after ESS (P<0.05).
Chronic rhinosinusitis (CRS) is thought to have a non-bacterial etiology, mainly because of low bacterial culture yield rates. Recently, however, it has become increasingly evident that biofilms are present on the mucosa of CRS patients, but not the mucosa of control subjects [1,2,3]. Furthermore, several studies have shown that CRS patients with a biofilm tend to have poor clinical outcomes after endoscopic sinus surgery (ESS) [3,4,5,6].
Researchers have used various imaging modalities to detect biofilms, including scanning and transmission electron microscopy (SEM and TEM), fluorescent in situ hybridization (FISH), and BacLight LIVE DEAD with confocal scanning laser microscopy (CSLM). These methodologies have some drawbacks. In SEM and TEM, fixation is difficult and it is challenging to differentiate between mucus, clots, and biofilms. CSLM with FISH is time-consuming (>3 hours), and BacLight can only be used for fresh samples. All of these methods are expensive and difficult to apply in a daily clinical setting.
Hematoxylin and eosin (H&E) staining has been shown to be a practical, cheap and easy method for detecting biofilms in CRS patients [7,8]. Although some studies have demonstrated that CRS outcomes are worse in patients confirmed to be biofilm-positive based on other experimental methods, no prior studies have investigated the correlation between biofilms detected by H&E staining and CRS prognosis [3,4,5,6]. If biofilms can be detected reliably by H&E staining and if the presence of a biofilm is correlated with CRS prognosis, ordinary H&E staining could potentially be used to predict outcomes after ESS.
Our aims in the present study were to determine whether H&E staining combined with light microscopy (LM) examination can be used to detect biofilms in patients with CRS and to determine whether the presence or absence of a biofilm can predict surgical outcomes after ESS.
We first conducted a pilot study to prospectively evaluate the feasibility of detecting biofilms with H&E staining. Then, we determined the reproducibility of detecting the presence of a biofilm with H&E staining and investigated if there was a correlation between surgical outcomes and the presence of a biofilm. The Institutional Review Board of Samsung Medical Center approved this study (IRB No. 2011-05-023-003).
We obtained fresh tissue samples from five patients with CRS during ESS for analysis by BacLight staining (FilmTracer Live/Dead Biofilm Viability Kit; Invitrogen, Molecular Probes, Carlsbad, CA, USA) and CSLM (LSM700; Carl Zeiss, Jena, Germany). Specimens from another six patients were collected during ESS for analysis by FISH with EUB338 (5'-Cy3-GCTGCCTCC-CGTAGGAGT-3') 4,6-diamidino-2-phenylindole (DAPI; Bioneer, Daejeon, Korea), which is bacterial-specific probe. The FISH and BacLight protocols were performed as described previously [9,10].
The mucosal specimens of the 11 CRS patients were fixed in 4% formalin and embedded in paraffin blocks using routine procedures, followed by H&E staining. H&E-stained sections were analyzed by LM and specimens prepared by FISH and BacLight were analyzed by CSLM.
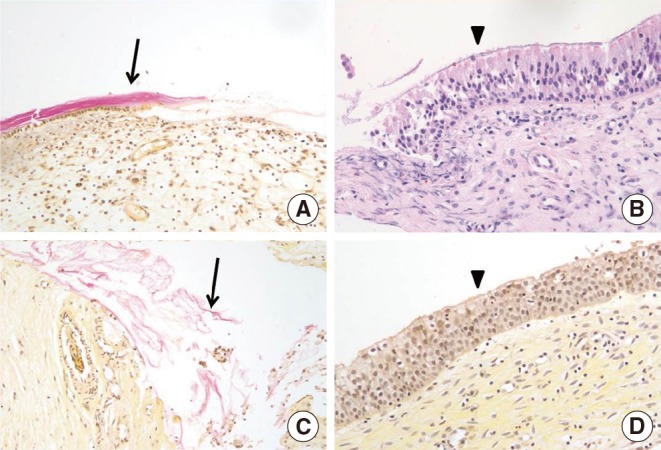
The presence of a biofilm in H&E-stained slides was diagnosed based on the following criteria [7]: (1) irregularly shaped groupings of small basophilic material one third of the size of the surrounding epithelial or inflammatory cells (Fig. 1A); (2) presence of a biofilm over the epithelial lining, not in or under the epithelial lining (Fig. 1A); (3) biofilm tightly adherent to the surface epithelium or pulled away slightly (Fig. 1B); or (4) a dense extracellular polysaccharide substance (EPS) material with embedded basophillic substance coating the epithelial surface (Fig. 1C).
Concordance between the presence or absence of a biofilm determined using H&E staining with LM versus the other standard modalities (FISH or BacLight with CSLM) was evaluated.
We performed retrospective analysis of prospectively collected data from a tertiary rhinological setting. The medical records and H&E-stained pathology slides of adult patients (≥19 years old) who underwent ESS for medically recalcitrant CRS between January 2009 and May 2010 were evaluated. All patients were operated on by a single senior surgeon (HJD) and followed-up for a minimum of 12 months.
Preoperative clinical data, including demographic information, previous sinus surgery history, asthma history, allergy confirmed by skin prick test, smoking status, and preoperative Lund-MacKay computed tomography scores were recorded. Preoperative nasal symptom scores including nasal obstruction, purulent rhinorrhea, postnasal drip sense, hyposmia, and facial fullness evaluated by 7-point Likert scales were also collected. Total symptom scores ranged between 5 and 35. The presence or absence of nasal symptoms and nasal endoscopic scores were evaluated 12 months after surgery. If patients reported any symptoms, however mild, they were classified as having ongoing persistent symptoms. Endoscopic scores were assigned by an investigator (SDH) who remained blinded to the biofilm status of the patients. Discharge (none, mild, or severe) and edema (none, mild, polypoid, or polyposis) were analyzed. Biofilm analysis was performed by an ear, nose and throat (ENT) doctor (SDH) and two pathologists (SYH and HYP) who were blinded to the preoperative and postoperative clinical data. Mucosa specimens from the ethmoid cavity classified as biofilm-positive by two or three observers were defined as biofilm-positive samples.
Statistical analyses were performed using IBM SPSS ver. 19 (IBM Co., Armonk, NY, USA). For all statistical tests, P≤0.05 was considered significant. The significance of differences between the characteristics of biofilm-positive and biofilm-negative patients were analyzed using a chi-square or Fisher exact test for dichotomous data or the Mann-Whitney U-test for two-way independent samples. Interrater variability was calculated using an intraclass correlation coefficient.
Of the 11 specimens stained with H&E, a biofilm was detected in seven according to the criteria described in the methods. All biofilms detected by H&E staining were also detected by FISH (n=3) and BacLight (n=4). All samples classified as biofilm-negative by H&E staining were also classified as biofilm-negative by FISH or BacLight.
The biofilm status of 55 patients with CRS was determined by H&E staining and LM. The patient sample consisted of 35 males and 20 females, with a mean age of 44 years (range, 19 to 73 years). Biofilms were detected in 28 of the 55 patients (50.9%). The interrater variability of the three independent observers was 0.731.
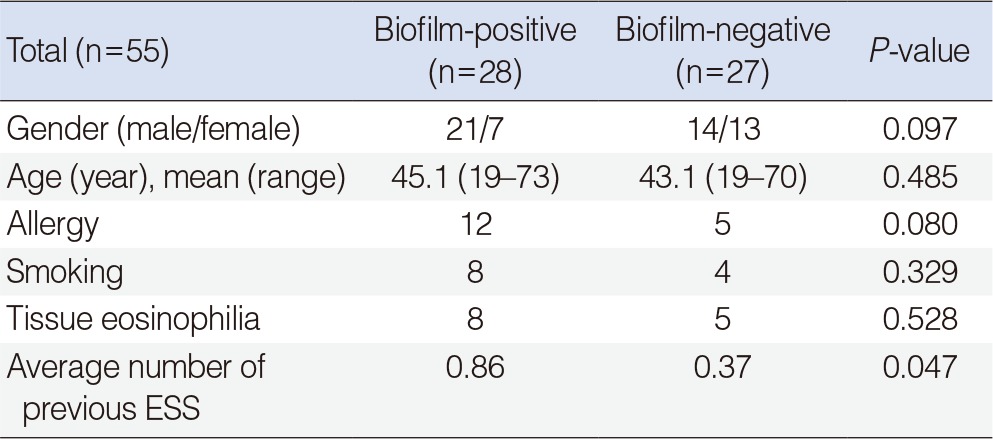
There were no significant differences in gender, age, allergy, smoking status, or tissue eosinophilia (>10/high power field) between the biofilm-positive and biofilm-negative patients. Patients with a biofilm had a more frequent history of previous sinus surgery than biofilm-negative patients (Table 1).
Biofilm-positive patients had a higher preoperative symptom score (mean, 22.3±7.57) than the biofilm-negative group (mean, 18.6±7.55), but this difference was not statistically significant (Fig. 2A). The biofilm-positive group had a significantly higher Lund-McKay score (mean, 16.1±4.91) than the biofilm-negative group (mean, 12.5±5.6) (Fig. 2B).
The presence of ongoing persistent symptoms and postoperative endoscopic sinus scores were both used as postoperative outcome measures. Nineteen of 28 patients with a biofilm (67.9%) had ongoing persistent symptoms, whereas just nine of 27 patients without a biofilm (33.3%) had ongoing symptoms (χ2 test, P=0.021) (Fig. 3).
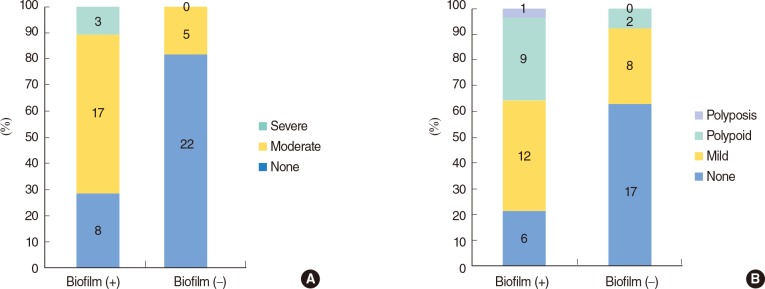
Biofilm-positive patients had significantly worse postoperative endoscopic scores, including discharge and edema scores, than biofilm-negative patients (Fisher exact test, P<0.05) (Fig. 4).
The detection of biofilms by H&E staining showed good, but not perfect agreement among the three raters (κ=0.731). This value is lower than the previously published interrater variability of BacLight/CSLM (0.8274) [3] and FISH/CSLM (0.9148) [11]. However, a previous study [11] reported that five of 20 patients (25%) had discrepant BacLight and FISH results, which are now known to be the most sensitive and specific tests [12] for detecting biofilms. In other words, there are no perfect modalities for detecting biofilms.
Two previous studies demonstrated that biofilm detection by H&E staining is reliable in CRS patients [7,8]. However, the presence of a biofilm may be missed by H&E staining, because this staining is just used to investigate tissue layers and their microscopic architecture.
Another issue with H&E staining is that a EPS lining is similar to a mucous lining. This was the most discordant point among the three investigators when diagnosing biofilms based on H&E staining. We performed mucicarmine staining, which specifically stains mucin, to confirm that the 'EPS' layer was not a mucus layer, and we were able to distinguish the EPS layer from the mucinous layer in some patients (Fig. 5). H&E staining combined with Gram staining has been proposed as a robust and reliable method for detection of bacterial biofilms [4]. Therefore, in cases where observers cannot easily distinguish whether a biofilm is present based on H&E staining, mucicarmine and/or Gram staining may be helpful.
In contrast to a previous study [3] that reported that H&E staining correctly identified the presence or absence of a biofilm in 100% of cases, we found that there was variation among the observers, with good (κ=0.731) rather than excellent agreement among observers. However, considering the cost-effectiveness of H&E staining, we consider it a reliable method for detecting biofilms.
There is increasing evidence that biofilms play a crucial role in several chronic inflammatory diseases [4,6,13,14]. Detection of a biofilm in patients who underwent ESS for CRS can help predict the clinical course of CRS and inform adjunctive treatment strategies. Although specific treatments are not available for targeting biofilms, it is very important to know if a biofilm is present, because it is strongly associated with treatment failure and persistent symptoms. In the current study, the presence or absence of biofilm determined just by H&E staining was correlated with preoperative disease severity and postoperative ongoing mucosal inflammation. H&E staining, which is a practical, cheap, and easy method for detecting biofilms, could potentially be widely used to predict worse postoperative outcomes.
It was the main limitation of this study that the presence or absence of a biofilm was not confirmed in all H&E specimens by standard methods such as FISH or BacLight with CSLM. However, this was not the primary goal of our study, and a previous study [7] confirmed a good correspondence between H&E results and FISH or BacLight results. We just want to show that the findings inferred from H&E staining with LM could help to predict outcome of ESS, practically and cost-effectively.
In conclusion, there was good agreement among raters regarding the presence or absence of biofilms in H&E-stained paraffin-embedded specimens. This is the first study to report that biofilms as just detected by H&E staining may play a role in refractory CRS after ESS. Routine H&E staining of surgical specimens and determination of a biofilm by pathology laboratories might be practical and cost-effective modalities for predicting worse outcome of ESS for CRS.
ACKNOWLEDGMENTS
This study was supported by a grant from the Health and Medical Technology R&D Program, Korea (Grant No. A110267). We are thank you for assistance of Ha Young Park.
References
1. Sanclement JA, Webster P, Thomas J, Ramadan HH. Bacterial biofilms in surgical specimens of patients with chronic rhinosinusitis. Laryngoscope. 2005; 4. 115(4):578–582. PMID: 15805862.

2. Foreman A, Psaltis AJ, Tan LW, Wormald PJ. Characterization of bacterial and fungal biofilms in chronic rhinosinusitis. Am J Rhinol Allergy. 2009; Nov-Dec. 23(6):556–561. PMID: 19958600.

3. Singhal D, Psaltis AJ, Foreman A, Wormald PJ. The impact of biofilms on outcomes after endoscopic sinus surgery. Am J Rhinol Allergy. 2010; May-Jun. 24(3):169–174. PMID: 20537281.

4. Zhang Z, Han D, Zhang S, Han Y, Dai W, Fan E, et al. Biofilms and mucosal healing in postsurgical patients with chronic rhinosinusitis. Am J Rhinol Allergy. 2009; Sep-Oct. 23(5):506–511. PMID: 19807984.

5. Bendouah Z, Barbeau J, Hamad WA, Desrosiers M. Biofilm formation by Staphylococcus aureus and Pseudomonas aeruginosa is associated with an unfavorable evolution after surgery for chronic sinusitis and nasal polyposis. Otolaryngol Head Neck Surg. 2006; 6. 134(6):991–996. PMID: 16730544.

6. Psaltis AJ, Weitzel EK, Ha KR, Wormald PJ. The effect of bacterial biofilms on post-sinus surgical outcomes. Am J Rhinol. 2008; Jan-Feb. 22(1):1–6. PMID: 18284851.

7. Hochstim CJ, Choi JY, Lowe D, Masood R, Rice DH. Biofilm detection with hematoxylin-eosin staining. Arch Otolaryngol Head Neck Surg. 2010; 5. 136(5):453–456. PMID: 20479374.

8. Toth L, Csomor P, Sziklai I, Karosi T. Biofilm detection in chronic rhinosinusitis by combined application of hematoxylin-eosin and gram staining. Eur Arch Otorhinolaryngol. 2011; 10. 268(10):1455–1462. PMID: 21547386.

9. Vasquez N, Mangin I, Lepage P, Seksik P, Duong JP, Blum S, et al. Patchy distribution of mucosal lesions in ileal Crohn's disease is not linked to differences in the dominant mucosa-associated bacteria: a study using fluorescence in situ hybridization and temporal temperature gradient gel electrophoresis. Inflamm Bowel Dis. 2007; 6. 13(6):684–692. PMID: 17206669.
10. Psaltis AJ, Ha KR, Beule AG, Tan LW, Wormald PJ. Confocal scanning laser microscopy evidence of biofilms in patients with chronic rhinosinusitis. Laryngoscope. 2007; 7. 117(7):1302–1306. PMID: 17603329.

11. Foreman A, Singhal D, Psaltis AJ, Wormald PJ. Targeted imaging modality selection for bacterial biofilms in chronic rhinosinusitis. Laryngoscope. 2010; 2. 120(2):427–431. PMID: 19950368.

12. Chen HH, Liu X, Ni C, Lu YP, Xiong GY, Lu YY, et al. Bacterial biofilms in chronic rhinosinusitis and their relationship with inflammation severity. Auris Nasus Larynx. 2012; 4. 39(2):169–174. PMID: 21641137.

13. Suh JD, Ramakrishnan V, Palmer JN. Biofilms. Otolaryngol Clin North Am. 2010; 6. 43(3):521–530. PMID: 20525507.

14. Hochstim CJ, Masood R, Rice DH. Biofilm and persistent inflammation in endoscopic sinus surgery. Otolaryngol Head Neck Surg. 2010; 11. 143(5):697–698. PMID: 20974342.

Fig. 1
Appearance of biofilms on H&E-stained sections (×100) from patients with chronic rhinosinusitis. (A) Irregularly-shaped groupings of small basophilic material one third of the size of the surrounding epithelial or inflammatory cells, present over the epithelial lining, not in or under the epithelial lining. (B) Tightly adherent to the surface epithelium or pulled away slightly. (C) Dense extracellular polysaccharide substance material with embedded basophillic substance coating the epithelial surface.
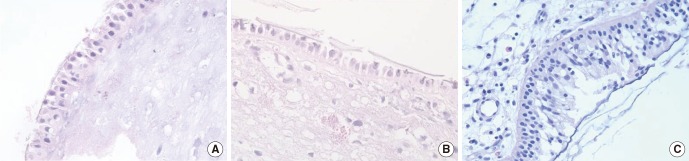
Fig. 2
Comparison of (A) preoperative symptom scores (Mann-Whitney test, P=0.075) and (B) Lund-MacKay computed tomography scores (Mann-Whitney test, P=0.021) between biofilm-positive (+) and biofilm-negative (-) patients.
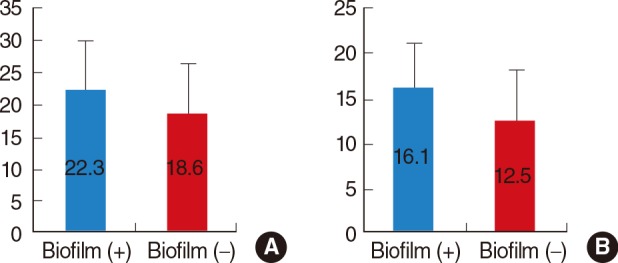
Fig. 3
Comparison of postoperative ongoing persistent symptoms between biofilm-positive (+) and biofilm-negative (-) patients.
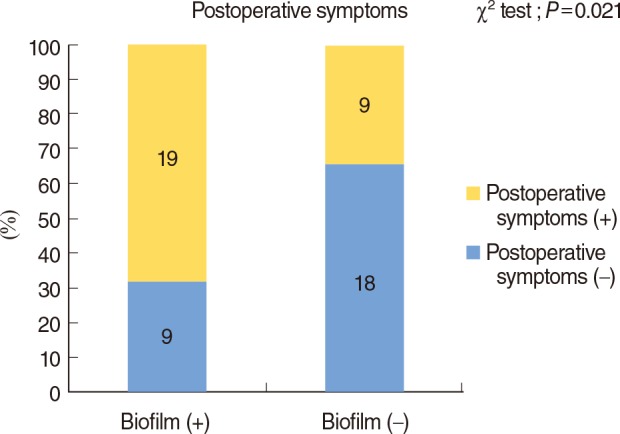
Fig. 4
Comparison of postoperative endoscopic scores including (A) discharge (Fisher exact test, P<0.001) and (B) edema (Fisher exact test, P=0.005) between biofilm-positive (+) and biofilm-negative (-) patients.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download