Abstract
Purpose
To improve our understanding of the pharmacokinetics of triamcinolone acetonide (TA) and TA concentration in ocular tissue, TA concentration was measured in ocular tissue after intravitreal and subtenon injection.
Methods
Fifteen rabbit eyes underwent subtenon TA injection (40 mg/1 ml), and another 15 rabbit eyes underwent intravitreal TA (4 mg/0.1 ml) injection. All eyes were enucleated at 1, 2, 4, 8, and 12 weeks after subtenon and intravitreal injection, and TA concentration in the aqueous and vitreous humor and the retina-choroid were measured by high performance liquid chromatography (HPLC).
Results
In cases of subtenon's TA injection, TA concentrations were 0 ng/ml, 136.7 ng/ml, 178.7 ng/ml, 0 ng/ml, and 0 ng/ml in the aqueous humor; 99.1 ng/ml, 125.7 ng/ml, 726.8 ng/ml, 0 ng/ml, and 0 ng/ml in the vitreous humor; 72.7 ng/ml, 304.6 ng/ml, 459.0 ng/ml, 0 ng/ml, and 0 ng/ml in the retina-choroid after 1, 2, 4, 8, and 12 weeks, respectively. In cases of intravitreal TA injection, the measured values were 0 ng/ml, 0 ng/ml, 0 ng/ml, 654.1 ng/ml, and 0 ng/ml in the aqueous humor; 28152.2 ng/ml, 13646.4 ng/ml, 11388.8 ng/ml, 10297.2 ng/ml, and 183.0 ng/ml in the vitreous humor; and 0 ng/ml, 27.9 ng/ml, 108.3 ng/ml, 0 ng/ml, and 0 ng/ml in the retina-choroid at each time point.
References
1. Ahmed I, Gokhale RD, Shah MV, Patton TF. Physicochemical determinants of drug diffusion across the conjunctiva, sclera and cornea. J Pharm Sci. 1987; 76:583–6.

2. McCartney HJ, Drysdale IO, Gornall AG, Basu PK. An autoradiographic study of the penetration of subconjunctivally injected hydrocortisone into the normal and inflamed rabbit eye. Invest Ophthalmol. 1965; 4:297–302.
3. Bakri SJ, Kaiser PK. Posterior subtenon triamcinolone acetonide for refractory diabetic macular edema. Am J Ophthalmol. 2005; 139:290–4.

4. Helm CJ, Holland GN. The effects of posterior subtenon injection of triamcinolone acetonide in patients with intermediate uveitis. Am J Ophthalmol. 1995; 120:55–64.

5. Geroski DH, Edelhauser HF. Drug delivery for posterior segment eye disease. Invest Ophthalmol Vis Sci. 2000; 41:961–4.
6. Cardillo JA, Melo LA Jr., Costa RA, et al. Comparison of intravitreal versus posterior sub-Tenon's capsule injection of triamcinolone acetonide for diffuse diabetic macular edema. Ophthalmology. 2005; 112:1557–63.

7. Ozdek S, Bahceci UA, Gurelik G, Hasanreisoglu B. Posterior subtenon and intravitreal triamcinolone acetonide for diabetic macular edema. J Diabetes Complications. 2006; 20:246–51.
8. Schindler RH, Chandler D, Thresher R. The clearance of intravitreal triamcinolone acetonide. Am J Ophthalmol. 1982; 93:415–7.

9. Martidis A, Duker JS, Greenberg PB, et al. Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology. 2002; 109:920–7.

10. Greenberg PB, Martidis A, Rogers AH, et al. Intravitreal triamcinolone acetonide for macular oedema due to central retinal vein occlusion. Br J Ophthalmol. 2002; 86:247–8.

11. Jonas JB, Kreissig I, Degenring RF. Intravitreal triamcinolone acetonide for pseudophakic cystoid macular edema. Am J Ophthalmol. 2003; 136:384–6.

12. Antcliff RJ, Spalton DJ, Stanford MR, et al. Intravitreal triamcinolone for uveitic cystoid macular edema: an optical coherence tomography study. Ophthalmology. 2001; 108:765–72.

13. Challa JK, Gillies MC, Penfold PL, et al. Exudative macular degeneration and intravitreal triamcinolone: 18 month follow up. Aust N Z J Ophthalmol. 1998; 26:277–81.

14. Jonas JB, Hayler JK, Sofker A, Panda-Jonas S. Intravitreal injection of crystalline cortisone as adjunctive treatment of proliferative diabetic retinopathy. Am J Ophthalmol. 2001; 131:468–71.

15. Jonas JB, Hayler JK, Sofker A, Panda-Jonas S. Regression of neovascular iris vessels by intravitreal injection of crystalline cortisone. J Glaucoma. 2001; 10:284–7.

16. Degenring RF, Jonas JB. Intravitreal injection of triamcinolone acetonide as treatment for chronic uveitis. Br J Ophthalmol. 2003; 87:361.

17. Bakri SJ, Beer PM. The effect of intravitreal triamcinolone acetonide on intraocular pressure. Ophthalmic Surg Lasers Imaging. 2003; 34:386–90.

18. Gillies MC, Simpson JM, Billson FA, et al. Safety of an intravitreal injection of triamcinolone: results from a randomized clinical trial. Arch Ophthalmol. 2004; 122:336–40.
19. Moshfeghi DM, Kaiser PK, Scott IU, et al. Acute endophthalmitis following intravitreal triamcinolone acetonide injection. Am J Ophthalmol. 2003; 136:791–6.

20. Roth DB, Chieh J, Spirn MJ, et al. Noninfectious endophthalmitis associated with intravitreal triamcinolone injection. Arch Ophthalmol. 2003; 121:1279–82.

21. Jonas JB, Sofker A, Degenring R. Intravitreal triamcinolone acetonide as an additional tool in pars plana vitrectomy for proliferative diabetic retinopathy. Eur J Ophthalmol. 2003; 13:468–73.

22. Beer PM, Bakri SJ, Singh RJ, et al. Intraocular concentration and pharmacokinetics of triamcinolone acetonide after a single intravitreal injection. Ophthalmology. 2003; 110:681–6.

23. Mason JO 3rd, Somaiya MD, Singh RJ. Intravitreal concentration and clearance of triamcinolone acetonide in nonvitrectomized human eyes. Retina. 2004; 24:900–4.

24. Pitkanen L, Ranta VP, Moilanen H, Urtti A. Permeability of retinal pigment epithelium: effects of permeant molecular weight and lipophilicity. Invest Ophthalmol Vis Sci. 2005; 46:641–6.
25. Ambati J, Gragoudas ES, Miller JW, et al. Transscleral delivery of bioactive protein to the choroid and retina. Invest Ophthalmol Vis Sci. 2000; 41:1186–91.
26. Robinson MR, Lee SS, Kim H, et al. A rabbit model for assessing the ocular barriers to the transscleral delivery of triamcinolone acetonide. Exp Eye Res. 2006; 82:479–87.

27. Kim H, Robinson MR, Lizak MJ, et al. Controlled drug release from an ocular implant: an evaluation using dynamic three-dimensional magnetic resonance imaging. Invest Ophthalmol Vis Sci. 2004; 45:2722–31.

28. Kalina PH, Erie JC, Rosenbaum L. Biochemical quantification of triamcinolone in subconjunctival depots. Arch Ophthalmol. 1995; 113:867–9.

29. Tsuji A, Tamai I, Sasaki K. Intraocular penetration kinetics of prednisolone after subconjunctival injection in rabbits. Ophthalmic Res. 1988; 20:31–43.

30. Ranta VP, Urtti A. Transscleral drug delivery to the posterior eye: Prospects of pharmacokinetic modeling. Advanced Drug Delivery Reviews. 2006; 58:1164–81.

31. Chin HS, Park TS, Moon YS, Oh JH. Difference in clearance of intravitreal triamcinolone acetonide between vitrectomized and nonvitrectomized eyes. Retina. 2005; 25:556–60.

32. Wordinger RJ, Clark AF. Effects of glucocorticoids on the trabecular meshwork: towards a better understanding of glaucoma. Prog Retin Eye Res. 1999; 18:629–67.

33. Wingate RJ, Beaumont PE. Intravitreal triamcinolone and elevated intraocular pressure. Aust N Z J Ophthalmol. 1999; 27:431–2.
34. Kang BS, Chung EY, Yun YP, et al. Inhibitory effects of anti-inflammatory drugs on interleukin-6 bioactivity. Biol Pharm Bull. 2001; 24:701–3.

35. Umland SP, Nahrebne DK, Razac S, et al. The inhibitory effects of topically active glucocorticoids on IL-4, IL-5, and interferon-gamma production by cultured primary CD4+ T cells. J Allergy Clin Immunol. 1997; 100:511–9.
36. Sze PY, Iqbal Z. Glucocorticoid action on depolarization- dependent calcium influx in brain synaptosomes. Neuroendocrinology. 1994; 59:457–65.
Figure 1.
The average concentration of triamcinolone acetonide in the vitreous after intravitreal and subtenon injection.
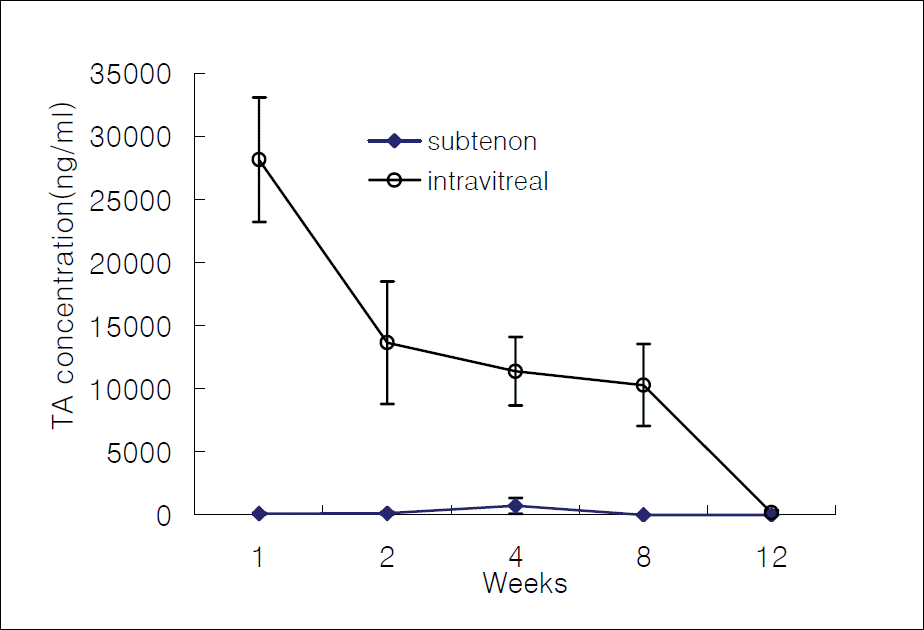
Figure 2.
The average concentration of triamcinolone acetonide in the aqueous humor after intravitreal and subtenon injection.
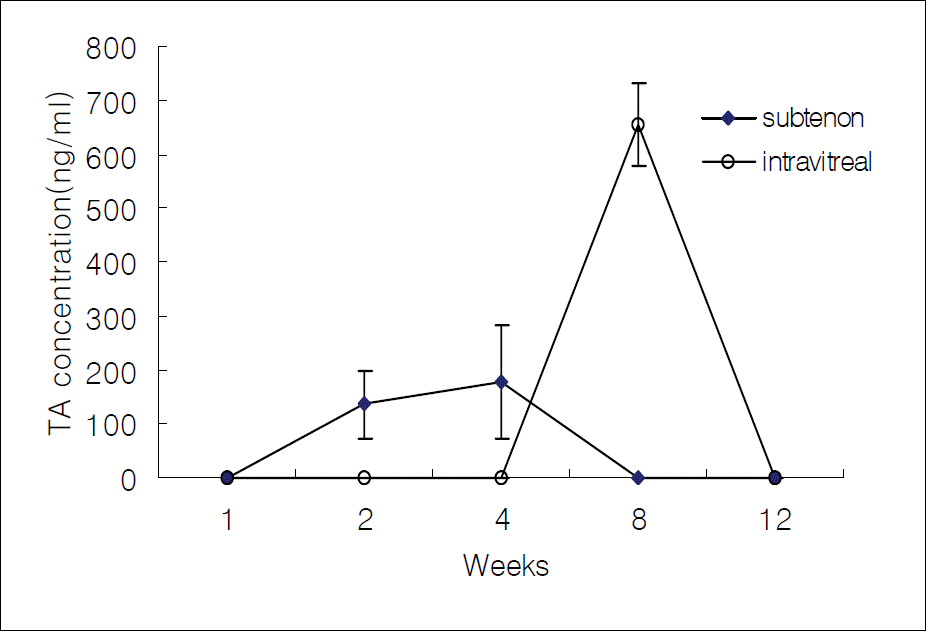
Figure 3.
The average concentration of triamcinolone acetonide in the retina-choroid after intravitreal and subtenon injection.
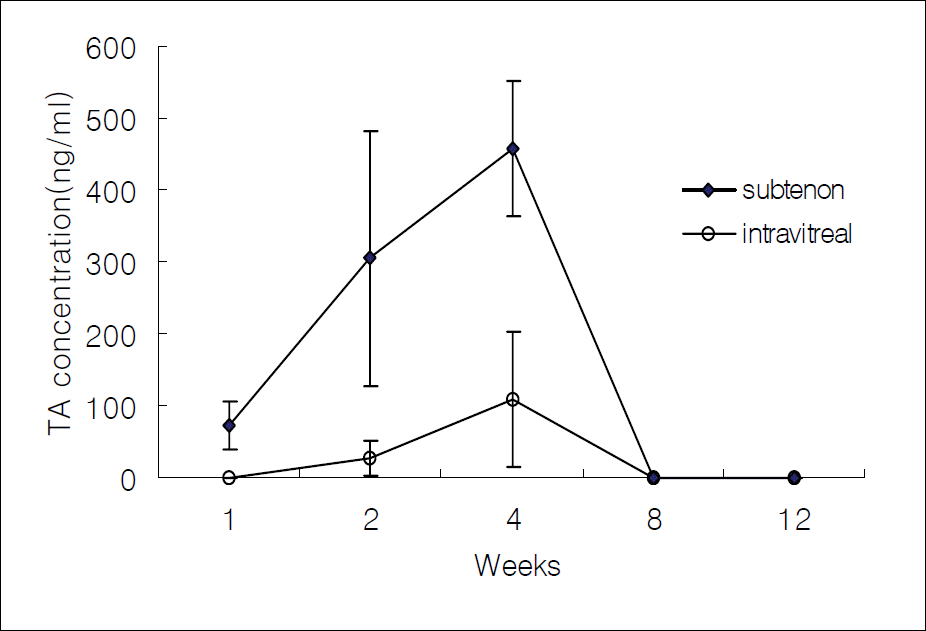
Table 1.
The average concentration of triamcinolone acetonide (TA) in various ocular tissues after subtenon injection
Table 2.
The average concentration of triamcinolone acetonide (TA) in various ocular tissue after intravitreal injection




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download